Nereistoxin
 Natural product parent of the series (nereistoxin)
| |
| Names | |
|---|---|
| Preferred IUPAC name
N,N-Dimethyl-1,2-dithiolan-4-amine | |
| Other names
NTX
| |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEBI |
|
| ChemSpider | |
ECHA InfoCard
|
100.121.136 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H11NS2 | |
| Molar mass | 149.27 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nereistoxin is a natural product identified in 1962 as the toxic organic compound N,N-dimethyl-1,2-dithiolan-4-amine. It had first been isolated in 1934 from the marine annelid Lumbriconereis heteropoda and acts by blocking the nicotinic acetylcholine receptor.[1] Researchers at Takeda in Japan investigated it as a possible insecticide. They subsequently developed a number of derivatives that were commercialised,[2][3] including those with the ISO common names[4] bensultap,[5] cartap,[6] thiocyclam[7] and thiosultap.[8][9]
Structures and synthesis
-
cartap
-
bensultap
-
thiosultap
-
thiocyclam
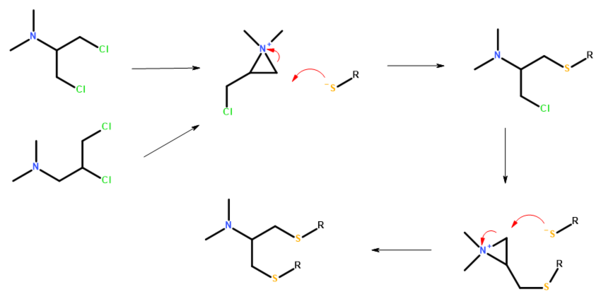
Bensultap (R=SO2Ph) was made by the reaction of the sodium salt of benzenethiolsulfonate (PhSO2SNa) with N,N-dimethyl 1,3-dichloro-2-propylamine or N,N-dimethyl 2,3-dichloropropylamine in ethanol.[9]
Bensultap can be converted to nereistoxin by treatment with alkali.[9]
History
Japanese fishermen used the
Mechanism of action
Nereistoxin has chemical similarity to
Usage
None of the insecticidal analogues of nereistoxin became major products in agriculture and their use was mainly limited to Japanese and Chinese cultivation of rice, where their control of pests such as the rice stem borer Chilo suppressalis was significant.[9] They were not licensed for use in Europe or the USA. The limited success of this group of chemicals was partly due to other compounds having similar modes of action but higher potency and mammalian safety becoming available.[15]
References
- .
- ISBN 978-0-85404-499-3.
- S2CID 87599872.
- ^ "Compendium of Pesticide Common Names". BCPC.
- ^ Pesticide Properties Database. "Bensultap". University of Hertfordshire.
- ^ Pesticide Properties Database. "Cartap". University of Hertfordshire.
- ^ Pesticide Properties Database. "Thiocyclam". University of Hertfordshire.
- ^ Pesticide Properties Database. "Thiosultap". University of Hertfordshire.
- ^ .
- PMID 4384262.
- PMID 14709019.
- ^ PMID 23317040.
- .
- ISBN 978-0-85404-559-4.
- PMID 23934864.
Further reading
- Godfrey, C. R. A. (17 November 1994). Agrochemicals from Natural Products. ISBN 0824795539.
External links
- Cartap in the Pesticide Properties DataBase (PPDB)
- Bensultap in the Pesticide Properties DataBase (PPDB)
- Thiosultap in the Pesticide Properties DataBase (PPDB)
- Thiocyclam in the Pesticide Properties DataBase (PPDB)