Nicotinic acetylcholine receptor
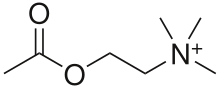

Nicotinic acetylcholine receptors, or nAChRs, are
The nicotinic receptors are considered
As
Since nicotinic receptors help transmit outgoing signals for the sympathetic and parasympathetic systems, nicotinic receptor antagonists such as hexamethonium interfere with the transmission of these signals. Thus, for example, nicotinic receptor antagonists interfere with the baroreflex[9] that normally corrects changes in blood pressure by sympathetic and parasympathetic stimulation of the heart.
Structure
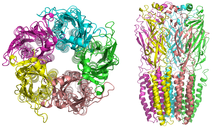
Nicotinic receptors, with a molecular mass of 290
In vertebrates, nicotinic receptors are broadly classified into two subtypes based on their primary sites of expression: muscle-type nicotinic receptors and neuronal-type nicotinic receptors. In the muscle-type receptors, found at the neuromuscular junction, receptors are either the embryonic form, composed of α1, β1, γ, and δ subunits in a 2:1:1:1 ratio ((α1)2β1γδ), or the adult form composed of α1, β1, δ, and ε subunits in a 2:1:1:1 ratio ((α1)2β1δε).
A number of electron microscopy and x-ray crystallography studies have provided very high resolution structural information for muscle and neuronal nAChRs and their binding domains.[10][14][15][16]
Binding
As with all ligand-gated ion channels, opening of the nAChR channel pore requires the binding of a chemical messenger. Several different terms are used to refer to the molecules that bind receptors, such as
In muscle-type nAChRs, the acetylcholine binding sites are located at the α and either ε or δ subunits interface. In neuronal nAChRs, the binding site is located at the interface of an α and a β subunit or between two α subunits in the case of α7 receptors. The binding site is located in the extracellular domain near the
Channel opening
Nicotinic AChRs may exist in different interconvertible conformational states. Binding of an agonist stabilizes the open and desensitized states. In normal physiological conditions, the receptor needs exactly two molecules of ACh to open.[19] Opening of the channel allows positively charged ions to move across it; in particular, sodium enters the cell and potassium exits. The net flow of positively charged ions is inward.
The nAChR is a non-selective cation channel, meaning that several different positively charged ions can cross through.
Many neuronal nAChRs can affect the release of other neurotransmitters.[5] The channel usually opens rapidly and tends to remain open until the agonist diffuses away, which usually takes about 1 millisecond.[4] AChRs can spontaneously open with no ligands bound or can spontaneously close with ligands bound, and mutations in the channel can shift the likelihood of either event.[23][18] Therefore, ACh binding changes the probability of pore opening, which increases as more ACh binds.
The nAChR is unable to bind ACh when bound to any of the
Effects
The activation of receptors by nicotine modifies the state of
Regulation
Desensitization
Ligand-bound desensitization of receptors was first characterized by Katz and Thesleff in the nicotinic acetylcholine receptor.[26]
Prolonged or repeated exposure to a stimulus often results in decreased responsiveness of that receptor toward a stimulus, termed desensitization. nAChR function can be modulated by phosphorylation[27] by the activation of second messenger-dependent protein kinases. PKA[26] and PKC,[28] as well as tyrosine kinases,[29] have been shown to phosphorylate the nAChR resulting in its desensitization. It has been reported that, after prolonged receptor exposure to the agonist, the agonist itself causes an agonist-induced conformational change in the receptor, resulting in receptor desensitization.[30]
Desensitized receptors can revert to a prolonged open state when an agonist is bound in the presence of a positive allosteric modulator, for example PNU-120,596.[31] Also, there is evidence that indicates specific chaperone molecules have regulatory effects on these receptors.[32]
Roles
The subunits of the nicotinic receptors belong to a multigene family (16 members in humans) and the assembly of combinations of subunits results in a large number of different receptors (for more information see the Ligand-Gated Ion Channel database). These receptors, with highly variable
Subunits
17 vertebrate nAChR subunits have been identified, which are divided into muscle-type and neuronal-type subunits. Although an α8 subunit/gene is present in avian species such as the chicken, it is not present in human or mammalian species.[35]
The nAChR subunits have been divided into 4 subfamilies (I–IV) based on similarities in protein sequence.[36] In addition, subfamily III has been further divided into 3 types.
| Neuronal-type | Muscle-type | ||||
| I | II | III | IV | ||
|---|---|---|---|---|---|
| α9, α10 | α7, α8 | 1 | 2 | 3 | α1, β1, δ, γ, ε |
| α2, α3, α4, α6 | β2, β4 | α5
| |||
- α genes: CHRNA1 (muscle), CHRNA2 (neuronal), CHRNA3, CHRNA4, CHRNA5, CHRNA6, CHRNA7, CHRNA8, CHRNA9, CHRNA10
- β genes: CHRNB1 (muscle), CHRNB2 (neuronal), CHRNB3, CHRNB4
- Other genes: CHRND (delta), CHRNE (epsilon), CHRNG (gamma)
Neuronal nAChRs are transmembrane proteins that form pentameric structures assembled from a family of subunits composed of α2–α10 and β2–β4.[37] These subunits were discovered from the mid-1980s through the early 1990s, when cDNAs for multiple nAChR subunits were cloned from rat and chicken brains, leading to the identification of eleven different genes (twelve in chickens) that code for neuronal nAChR subunits; The subunit genes identified were named α2–α10 (α8 only found in chickens) and β2–β4.[38] It has also been discovered that various subunit combinations could form functional nAChRs that could be activated by acetylcholine and nicotine, and the different combinations of subunits generate subtypes of nAChRs with diverse functional and pharmacological properties.[39] When expressed alone, α7, α8, α9, and α10 are able to form functional receptors, but other α subunits require the presence of β subunits to form functional receptors.[37] In mammals, nAchR subunits have been found to be encoded by 17 genes, and of these, nine genes encoding α-subunits and three encoding β-subunits are expressed in the brain. β2 subunit-containing nAChRs (β2nAChRs) and α7nAChRs are widely expressed in the brain, whereas other nAChR subunits have more restricted expression.[40] The pentameric assembly of nAChRs is subjected to the subunits that are produced in various cell types such as in the human lung where epithelial and muscular pentamers largely differ.[41]
CHRNA5/A3/B4
An important nAchR gene cluster (CHRNA5/A3/B4) contains the genes encoding for the
CHRNA6/CHRNB3
CHRNB3 and CHRNA6 are also grouped in a gene cluster, located on 8p11.[42] Multiple studies have shown that SNPS in the CHRNB3–CHRNA6 have been linked to nicotine dependence and smoking behavior, such as two SNPs in CHRNB3, rs6474413 and rs10958726.[42] Genetic variation in this region also displays influence susceptibility to use drugs of abuse, including cocaine and alcohol consumption.[44] Nicotinic receptors containing α6 or β3 subunits expressed in brain regions, especially in the ventral tegmental area and substantia nigra, are important for drug behaviors due to their role in dopamine release.[45] Genetic variation in these genes can alter sensitivity to drugs of abuse in numerous ways, including changing the amino acid structure of the protein or cause alterations in transcriptional and translational regulation.[44]
CHRNA4/CHRNB2
Other well studied nAChR genes include the CHRNA4 and CHRNB2, which have been associated as Autosomal Dominant Nocturnal Frontal Lobe Epilepsy (ADNFLE) genes.[42][46] Both of these nAChR subunits are present in the brain and the occurrence of mutations in these two subunits cause a generalized type of epilepsy. Examples include the CHRNA4 insertion mutation 776ins3 that is associated with nocturnal seizures and psychiatric disorders, and the CHRNB2 mutation I312M that seems to cause not only epilepsy but also very specific cognitive deficits, such as deficits in learning and memory.[46][47] There is naturally occurring genetic variation between these two genes and analysis of single nucleotide polymorphisms (SNPs) and other gene modifications show a higher variation in the CHRNA4 gene than in the CHRNB2 gene, implying that nAChR β2, the protein encoded by CHRNB2, associates with more subunits than α4. CHRNA2 has also been reported as a third candidate for nocturnal frontal lobe seizures.[42][46]
CHRNA7
Several studies have reported an association between CHRNA7 and endophenotypes of psychiatric disorders and nicotine dependence, contributing to the significant clinical relevance of α7 and research being done on it.[46] CHRNA7 was one of the first genes that had been considered to be involved with schizophrenia. Studies identified several CHRNA7 promoter polymorphisms that reduce the genes transcriptional activity to be associated with schizophrenia, which is consistent with the finding of reduced levels of a7 nAChRs in the brain of schizophrenic patients.[46] Both nAChRs subtypes, α4β2 and α7, have been found to be significantly reduced in post-mortem studies of individuals with schizophrenia.[48] Additionally, smoking rates are significantly higher in those with schizophrenia, implying that smoking nicotine may be a form of self-medicating.[49]
Notable variations
Nicotinic receptors are pentamers of these subunits; i.e., each receptor contains five subunits. Thus, there is immense potential of variation of these subunits, some of which are more commonly found than others. The most broadly expressed subtypes include (α1)2β1δε (adult muscle-type), (α3)2(β4)3 (ganglion-type), (α4)2(β2)3 (CNS-type) and (α7)5 (another CNS-type).[50] A comparison follows:
| Receptor-type | Location | Effect; functions | Nicotinic agonists |
Nicotinic antagonists |
|---|---|---|---|---|
Muscle-type: (α1)2β1δε[50] or (α1)2β1δγ |
Neuromuscular junction | EPSP, mainly by increased Na+ and K+ permeability |
|
|
Ganglion-type : (α3)2(β4)3 |
autonomic ganglia |
EPSP, mainly by increased Na+ and K+ permeability |
| |
| Heteromeric CNS-type: (α4)2(β2)3 |
Brain |
| ||
| Further CNS-type: (α3)2(β4)3 |
Brain | presynaptic excitation |
| |
| Homomeric CNS-type: (α7)5 |
Brain | presynaptic excitation,[50] mainly by increased Na+, K+ and Ca2+ permeability. Major subtype involved in some of the cognitive effects of nicotine.[55] Moreover, activation of (α7)5 could improve neurovascular coupling response in neurodegenerative disease[56] and neurogenesis in ischemic stroke.[57] Also involved in the pro-angiogenic effects of nicotine and accelerate the progression of chronic kidney disease in smokers.[58][59][60]
|
|
See also
- Muscarinic agonist
- Muscarinic antagonist
- TDBzcholine
- Myasthenia gravis
- Congenital myasthenic syndrome
- Adrenergic
- Cholinergic
References
- PMID 24849809.
- ISBN 978-4-431-68011-6.
- ^ ISBN 978-0-87893-697-7.
- ^ a b c d e f Siegel GJ, Agranoff BW, Fisher SK, Albers RW, Uhler MD (1999). "Basic Neurochemistry: Molecular, Cellular and Medical Aspects". GABA Receptor Physiology and Pharmacology (6th ed.). American Society for Neurochemistry. Retrieved 2008-10-01.
- ^ PMID 11532443.
- PMID 17073660.
- PMID 30969605.
- S2CID 9441100.
- PMID 206690.
- ^ PMID 15701510.
- PMID 15023997.
- S2CID 19114228.
- ^ hdl:2434/978688.
- S2CID 4415937.
- S2CID 30096256.
- PMID 27698419.
- ISBN 978-0-12-660303-3.
- ^ S2CID 19008547.
- OCLC 38067558.[page needed]
- S2CID 8684707.
- PMID 15644873.
- S2CID 4356336.
- PMID 28134524.
- PMID 2580101.
- PMID 18327915.
- ^ PMID 1331363.
- PMID 6302672.
- PMID 3038884.
- S2CID 4239105.
- PMID 712829.
- PMID 15858066.
- S2CID 31929001.
- PMID 23040810.
- S2CID 42215860.
- S2CID 39839166.
- S2CID 2040912.
- ^ PMID 20685379.
- PMID 21704022.
- S2CID 39839166.
- S2CID 661315.
- PMID 33050277.
- ^ PMID 19564872.
- PMID 16594641.
- ^ PMID 27085880.
- PMID 17825262.
- ^ PMID 18691557.
- S2CID 29811931.
- PMID 10989262.
- S2CID 41306366.
- ^ ISBN 978-0-443-07145-4.[page needed]
- ^ a b Neurosci.pharm - MBC 3320 Acetylcholine Archived 2007-12-27 at the Wayback Machine
- PMID 28607947.
- PMID 23933294.
- ^ "Nicotine: Biological activity". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Retrieved 7 February 2016.
Kis as follows; α2β4=9900nM [5], α3β2=14nM [1], α3β4=187nM [1], α4β2=1nM [4,6]. Due to the heterogeneity of nACh channels we have not tagged a primary drug target for nicotine, although the α4β2 is reported to be the predominant high affinity subtype in the brain which mediates nicotine addiction [2-3].
- PMID 22300026.
- PMID 26647792.
- PMID 28551702.
- PMID 22796717.
- PMID 23892062.
- S2CID 14562170.
External links
 Media related to Nicotinic acetylcholine receptors at Wikimedia Commons
Media related to Nicotinic acetylcholine receptors at Wikimedia Commons- Calculated spatial position of Nicotinic acetylcholine receptor in the lipid bilayer
