Praseodymium compounds
Praseodymium compounds are compounds formed by the lanthanide metal praseodymium (Pr). In these compounds, praseodymium generally exhibits the +3 oxidation state, such as PrCl3, Pr(NO3)3 and Pr(CH3COO)3. However, compounds with praseodymium in the +2 and +4 oxidation states, and unlike other lanthanides, the +5 oxidation state, are also known.
Halides

Praseodymium metal reacts with all the stable halogens to form green trihalides:[1]
- 2 Pr (s) + 3 F2 (g) → 2 PrF3 (s)
- 2 Pr (s) + 3 Cl2 (g) → 2 PrCl3 (s)
- 2 Pr (s) + 3 Br2 (g) → 2 PrBr3 (s)
- 2 Pr (s) + 3 I2 (g) → 2 PrI3 (s)
Praseodymium(III) bromide is the only stable bromide of praseodymium. It adopts the UCl3 crystal structure.[7] The praseodymium ions are 9-coordinate and adopt a tricapped trigonal prismatic geometry.[8] The praseodymium–bromine bond lengths are 3.05 Å and 3.13 Å.[9] Praseodymium(III) iodide can be prepared by heating praseodymium and iodine in an inert atmosphere produces praseodymium(III) iodide,[10] or by heating praseodymium with mercury(II) iodide.[11] It forms orthorhombic crystals which are hygroscopic.[10] It crystallizes in the PuBr3 type[11][12] with space group Cmcm (No. 63) with a = 4.3281(6) Å, b = 14.003(6) Å and c = 9.988(3) Å.[13]
The
Oxides
Praseodymium can form many different oxides, although the only oxides that are stable at room temperature are Pr2O3, Pr6O11 and PrO2. Praseodymium(III) oxide is a green powder that forms hexagonal crystals,[14] and crystallizes in the manganese(III) oxide or bixbyite structure.[15] Praseodymium(IV) oxide can be produced by boiling Pr6O11 in water or acetic acid:[16]
- Pr6O11 + 3 H2O → 4 PrO2 + 2 Pr(OH)3
Praseodymium(III,IV) oxide is the most stable form of the praseodymium oxides at ambient temperature and pressure.[17] It is soluble in water[18] and has a cubic fluorite structure.[19] It can be prepared via solid-state methods such as thermolysis, molten salt method, calcination or precipitation.[19][17][20]
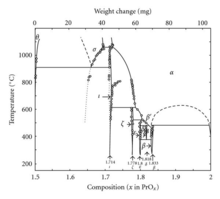
In addition to Pr6O11, praseodymium also forms a system of oxides at different phases:[21]
| Value | Formula | Phase | x in PrOx | Average oxidation state of Pr |
|---|---|---|---|---|
| 4 | Pr2O3 | θ | 1.5 | 3 |
| 7 | Pr7O12 | ι | 1.714 | 3.428 |
| 9 | Pr9O16 | ζ | 1.778 | 3.556 |
| 10 | Pr5O9 | ε | 1.8 | 3.6 |
| 11 | Pr11O20 | δ | 1.818 | 3.636 |
| 12 | Pr6O11 | β | 1.833 | 3.667 |
| ∞ | PrO2 | 2 | 4 |
Organopraseodymium compounds
Organopraseodymium compounds are compounds with a
- Organopraseodymium compounds are very air- and water-sensitive and pyrophoric.
- Chemistry in the 0 oxidation state is far more limited. In fact, their electropositive nature makes their organometallic compounds more likely to be ionic.
- Organopraseodymium compounds form no stable .
σ-Bonded complexes
Metal-carbon σ bonds are found in alkyls of praeodymium such as [PrMe6]3− and Pr[CH(SiMe3)2]3.
π-Bonded complexes
Cyclopentadienyl complexes, are known for praseodymium. It can be produced by the following reaction scheme:
- 3 Na[Cp] + PrCl3 → Pr[Cp]3 + 3 NaCl
These compounds are of limited use and academic interest.[25]
Applications
Praseodymium(III) nitride is used in high-end electric and semiconductor products, and as a raw material to produce phosphor. Also it is used as a magnetic material and sputtering target material.[26] Many praseodymium compounds, such as praseodymium(III) oxalate, are used to colour some glasses and enamels. If mixed with certain other materials, praseodymium(III) oxalate paints glass intense yellow.[27]
Pictures of praseodymium compounds
-
Praseodymium(III) acetate (Pr(CH3COO)3)
-
Praseodymium(III) oxide (Pr2O3)
-
Praseodymium(III) chloride (PrCl3)
-
Praseodymium(III) nitrate (Pr(NO3)3)
-
Praseodymium(III) sulfate octahydrate (Pr2(SO4)3·8H2O)
-
Praseodymium(III) hydroxide (Pr(OH)3)
-
Praseodymium(III) phosphate (PrPO4)
References
- ^ "Chemical reactions of Praseodymium". Webelements. Retrieved 9 July 2016.
- doi:10.1016/j.matlet.2006.04.124. Retrieved 2019-03-26.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ J. Cybinska, J. Sokolnicki, J. Legendziewicz, G. Meyer, Journal of Alloys and Compounds, 341, 115–123 (2002).
- ^ L. F. Druding, J. D. Corbett, "Lower Oxidation States of the Lanthanides. Neodymium(II) Chloride and Iodide", J. Am. Chem. Soc. 83, 2462 (1961); J. D. Corbett, Rev. Chim. Minerale 10, 239 (1973),
- ^ a b F. T. Edelmann, P. Poremba, in: Synthetic Methods of Organometallic and Inorganic Chemistry, (W. A. Herrmann, ed.), Vol. 6, Georg Thieme Verlag, Stuttgart, 1997.
- ^ J. Cybinska, J. Sokolnicki, J. Legendziewicz, G. Meyer, Journal of Alloys and Compounds, 341, 115–123 (2002).
- ISBN 978-0-19-965763-6.
- ^ a b c Greenwood and Earnshaw, p. 1240–2
- doi:10.1063/1.338741.
- ^ ISBN 978-1-4987-5429-3.
- ^ .
- ISBN 978-0-19-965763-6.
- ISBN 0-8493-0594-2
- ISBN 978-3-540-20481-7, retrieved 2009-03-18
- ^ Chinese: 《无机化学丛书》.第七卷 钪 稀土元素. 科学出版社. 1.3.4 氧化态+4的化合物. P193~195
- ^ .
- ^ "Praseodymium Oxide (Pr6O11)". www.reade.com. Retrieved 2018-03-15.
- ^ .
- ^ a b Shamshi Hassan, M., Shaheer Akhtar, M., Shim, KB. et al. Morphological and Electrochemical Properties of Crystalline Praseodymium Oxide Nanorods. Nanoscale Res Lett 5, 735 (2010). https://doi.org/10.1007/s11671-010-9547-8
- ISBN 978-0-08-037941-8.
- ISBN 978-0-08-037941-8.
- ISBN 978-0-08-037941-8.
- S2CID 212564931.
- ISBN 978-0-470-14547-0. Retrieved 28 July 2013.
- ^ "Praseodymium Nitride (PrN) Powder". Stanford Advanced Materials. Retrieved 18 June 2021.
- ^ "Praseodymium Oxalate 99%-99.999% from Metall Rare Earth Limited". metall.com.cn. Retrieved 17 June 2021.
- ^ Osten HJ, Liu JP, P Gaworzewski, E Bugiel, Zaumseil P: IEDM Technical Digest 653. 2000.







