Rhodium pentafluoride
| |

| |
| Names | |
|---|---|
| IUPAC name
Rhodium(V) fluoride
| |
| Identifiers | |
3D model (
JSmol ) |
|
PubChem CID
|
|
| |
| |
| Properties | |
| F5Rh | |
| Molar mass | 197.89751 g·mol−1 |
| Appearance | Red solid |
| Density | 3.95 g cm3 |
| Structure | |
| Monoclinic | |
| P21/a | |
a = 12.338, b = 9.9173, c = 5.5173 α = 90°, β = 100.42°°, γ = 90°
| |
Lattice volume (V)
|
663.85 |
Formula units (Z)
|
8 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Rhodium pentafluoride is an inorganic compound with the formula Rh4F20. It is a red solid. It is prepared by fluorination of rhodium trifluoride at 400 °C.[1]
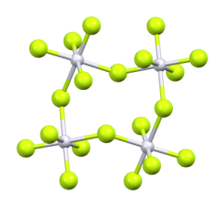
According to X-ray crystallography, the Rh centers are octahedral. The structure is very similar to that of the related
iridium pentafluoride. All are tetrameric, meaning that they have the molecular structure [MF5]4. The M-F distances for the bridging fluoride ligands are typically about 0.2 Å longer than the Rh-F distances for the nonbridging fluoride ligands. In the case of rhodium pentafluoride, these distances average 1.999(4) and 1.808(8) Å.[2]
The Rh-F-Rh angles average 135°, which leads to a ruffled structure. In contrast, the M-F-M centers are linear in the pentafluorides of niobium, tantalum, molybdenum, and tungsten.
References
- doi:10.1039/c19650000306.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - doi:10.1021/ic50129a029.)
{{cite journal}}: CS1 maint: multiple names: authors list (link
