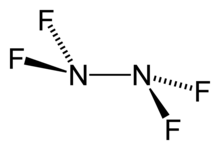
Tetrafluorohydrazine
| |
| |
| Names | |
|---|---|
| IUPAC name
1,1,2,2-tetrafluorohydrazine
| |
| Other names
Tetrafluorohydrazine, perfluorohydrazine, UN 1955
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.030.091 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| N2F4 | |
| Molar mass | 104.008 g·mol−1 |
| Appearance | Colourless gas |
| Melting point | −164.5 °C (−264.1 °F; 108.6 K)[1] |
| Boiling point | −73 °C (−99 °F; 200 K)[1] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Explosion |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
10 mL/kg (rat, intraperitoneal)[2] |
LC50 (median concentration)
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetrafluorohydrazine or perfluorohydrazine,
Tetrafluorohydrazine is manufactured from nitrogen trifluoride using an iron catalyst or iron(II) fluoride.
Properties
Tetrafluorohydrazine is in equilibrium with its radical monomer nitrogen difluoride.[3]
- N2F4 ⇌ 2 •NF2
At room temperature N2F4 is mostly associated with only 0.7% in the form of NF2 at 5mm Hg pressure. When the temperature rises to 225 °C, it mostly dissociates with 99% in the form of NF2.[4]
The energy needed to break the N−N bond in N2F4 is 20.8 kcal/mol, with an entropy change of 38.6 eu.[4] For comparison, the dissociation energy of the N−N bond is 14.6 kcal/mol (61 kJ/mol) in N2O4, 10.2 kcal/mol (43 kJ/mol) in N2O2, and 60 kcal/mol (250 kJ/mol) in N2H4. The enthalpy of formation of N2F4 (ΔfH°) is 34.421 kJ/mol.[5]
Uses
Tetrafluorohydrazine is used in some
Safety
Tetrafluorohydrazine is a highly hazardous chemical that explodes in the presence of organic materials.[2]
It is a toxic chemical which irritates skin, eyes and lungs. It is a
Tetrafluorohydrazine explodes or ignites on contact with
There is a fatal case in which during opening of valves to check the pressure, the cylinder exploded, killing one man and injuring another.[2]
References
- ^ ISBN 978-0-08-037941-8.
- ^ a b c d e f g h "Tetrafluorohydrazine". pubchem.ncbi.nlm.nih.gov. National Library of Medicine. Retrieved 26 March 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ISBN 978-3-662-06341-5. Retrieved 29 August 2015.
- ^ . molecule dimensions and angles
- ^ "Nitrogen difluoride NF2(g)". www.chem.msu.su.
- ^ Tetrafluorohydrazine at DTIC.mil archived March 12, 2007
