Tin(IV) fluoride
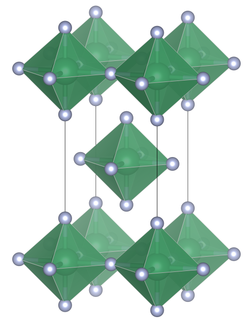 Unit cell of tin(IV) fluoride
| |
| Names | |
|---|---|
| IUPAC name
tin(IV) fluoride
| |
| Other names
stannic fluoride, tin tetrafluoride
| |
| Identifiers | |
3D model (
JSmol ) |
|
ECHA InfoCard
|
100.029.105 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| SnF4 | |
| Molar mass | 194.704 g/mol |
| Appearance | white solid |
| Melting point | above 700 °C (sublimes) |
| Structure | |
Tetragonal, tI10
| |
| I4/mmm, No. 139 | |
| Related compounds | |
Other anions
|
Tin(IV) chloride Tin(IV) bromide Tin(IV) iodide |
Other cations
|
Tin tetrafluoride
Lead tetrafluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tin(IV) fluoride is a chemical compound of tin and fluorine with the chemical formula SnF4 and is a white solid with a melting point above 700 °C.[1]
SnF4 can be prepared by the reaction of tin metal with fluorine gas:[2]
- Sn + 2F2 → SnF4
However, a passivating metal fluoride layer will be created and the surface will eventually become unreactive. An alternative synthesis is the reaction of SnCl4 with anhydrous hydrogen fluoride:[1]
- SnCl4 + 4HF → SnF4 + 4HCl
With alkali metal fluorides (e.g. KF) hexafluorostannates are produced (e.g.K2SnF6), which contain the octahedral SnF62− anion. SnF4 behaves as a
Lewis acid and adducts L2·SnF4 and L·SnF4 have been produced.[2]
Structure
Unlike the other tin
trans to one another.[3] The melting point of SnF4 is much higher (700 °C) than the other tin(IV) halides which are relatively low melting, (SnCl4, −33.3 °C; SnBr4, 31 °C; SnI4, 144 °C).[1] The structure can also be contrasted with the tetrafluorides of the lighter members of group 14, (CF4, SiF4 and GeF4) which in the solid state form molecular crystals.[2]
See also
- Stannous fluoride
References
- ^ ISBN 0-7506-3365-4.
- ^ ISBN 0-12-352651-5.
- ISBN 978-0130399137
