Platinum tetrafluoride
| |
| Names | |
|---|---|
| IUPAC name
Platinum(IV) fluoride
| |
| Other names
Platinum tetrafluoride
Platinous fluoride | |
| Identifiers | |
3D model (
JSmol ) |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| F4Pt | |
| Molar mass | 271.078[1] |
| Appearance | red-orange solid[1] |
| Density | 7.08 g/cm3 (calc.)[2] |
| Melting point | 600 °C (1,112 °F; 873 K)[1] |
| +455.0·10−6 cm3/mol | |
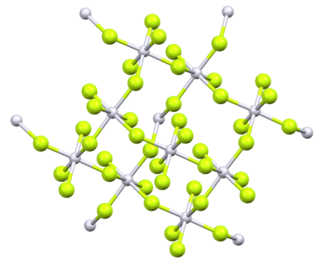
| Structure | |
Orthorhombic, oF40
| |
| Fdd2, No. 43[2] | |
a = 0.9284 nm, b = 0.959 nm, c = 0.5712 nm
| |
| Related compounds | |
Other anions
|
Platinum(IV) bromide Platinum(IV) chloride |
Related compounds
|
Platinum(VI) fluoride
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Platinum tetrafluoride is the
Preparation
The compound was first reported by
Properties
Platinum tetrafluoride vapour at 298.15 K consists of individual molecules. The enthalpy of sublimation is 210 kJmol−1.[5] Original analysis of powdered PtF4 suggested a tetrahedral molecular geometry, but later analysis by several methods identified it as octahedral, with four of the six fluorines on each platinum bridging to adjacent platinum centres.[6]
Reactions
A solution of platinum tetrafluoride in water is coloured reddish brown, but it rapidly decomposes, releasing heat and forming an orange coloured
Platinum tetrafluoride can form adducts with selenium tetrafluoride and bromine trifluoride.[7] Volatile crystalline adducts are also formed in combination with BF3, PF3, BCl3, and PCl3.[7]
Related compounds
The fluoroplatinates are salts containing the PtF62− ion. Fluoroplatinic acid H2PtF6 forms yellow crystals that absorb water from the air. Ammonium, sodium, magnesium, calcium, strontium, and rare earth including lanthanum fluoropalatinate salts are soluble in water.[7] Potassium, rubidium, caesium, and barium salts are insoluble in water.[7]
References
- ^ ISBN 1-4398-5511-0.
- ^ ]
- ^ Moissan, H. "Platinum tetrafluoride". Comptes Rendus Hebdomadaires des Séances de l'Académie des Sciences. 109: 807–9.
- .
- .
- ISBN 9780080578767.
- ^ a b c d e f Derek Harry Lohmann (October 1961). The fluorides of platinum and related compounds (Thesis). University of British Columbia.
