Roussin's black salt
| |
| Names | |
|---|---|
| Other names
potassium heptanitrosyltri-μ3-thiotetraferrate
| |
| Identifiers | |
| |
3D model (
JSmol ) |
|
PubChem CID
|
|
| |
| |
| Properties | |
| Fe4KN7O7S3 | |
| Molar mass | 568.70 g·mol−1 |
| Appearance | Black solid |
| Melting point | 198 to 200 °C (388 to 392 °F; 471 to 473 K) |
| Related compounds | |
Related
|
Roussin's Red Salt
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Roussin's black salt is a
the red salt also bearing his name
.
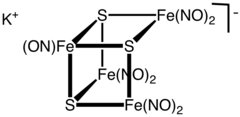
Structure
The cluster anion has the geometry of an incomplete cubane-type cluster with C3v symmetry. The dark colour of the complex is attributed to a number of charge-transfer interactions.[3]

Synthesis
Roussin’s black salt is produced by the reaction of nitrous acid, potassium hydroxide, potassium sulfide, and iron(II) sulfate in aqueous solution.[4] It can also be formed by the conversion of Roussin's red salt in mildly acidic conditions. This reaction is reversible and Roussin’s red salt is reformed upon alkalization of the reaction solution.
Uses
Roussin’s black salt is a nitric oxide donor.[5] Also, Roussin’s Black Salt exhibits antibacterial activity in some food processing applications.[6]
See also
References
- ISBN 978-0-08-037941-8.
- .
- .
- .
- PMID 15050534.
- ISBN 978-0-08-037941-8.
