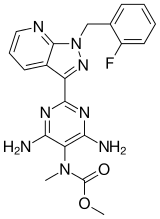
Riociguat
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Adempas |
| Other names | BAY 63-2521 |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 94% |
| Protein binding | 95% |
| Metabolism | CYP1A1, CYP3A4, CYP2C8, CYP2J2 |
| Metabolites | N-desmethylriociguat (active), glucuronide (inactive) |
| Elimination half-life | 12 h (patients); 7 h (healthy people)[3] |
| Excretion | 33–45% via kidney, 48–59% via bile duct |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Riociguat, sold under the brand name Adempas, is a
It is available as a
Contraindications
Riociguat can cause
The substance is also contraindicated in pulmonary hypertension in combination with idiopathic interstitial pneumonia (PH-IIP). A clinical trial testing riociguat for this purpose was prematurely terminated because it increased severe adverse effects and mortality in patients with pulmonary hypertension caused by idiopathic interstitial pneumonia when compared to placebo.[6]
Adverse effects
Serious adverse effects in clinical trials included bleeding. Hypotension (low blood pressure), headache, and
Interactions
Chemistry and mechanism of action
In healthy individuals
Pharmacology
Riociguat at concentration between 0.1 and 100
History
Discovery
The first
Several clinical trials have been undertaken to investigate and evaluate diverse aspects of riociguat, and some of them are still ongoing.[16]
Phase I clinical trials
One of the first studies was designed to test the safety profile, pharmacokinetics and pharmacodynamics of single oral doses of riociguat (0.25–5 mg). 58 healthy male subjects were given riociguat orally (oral solution or immediate-release tablet) in a randomised, placebo-controlled trial. Doses of riociguat were increased stepwise, and riociguat was well tolerated up to 2.5 mg.[12]
Phase II clinical trials
A
An open-label, non-controlled phase II trial of riociguat in 75 adult patients (42 with CTEPH and 33 with PAH, all in World Health Organization (WHO) functional class II or III) evaluated the safety and tolerability, and the effects on hemodynamics, exercise capacity and functional class. Riociguat was given three times daily for 12 weeks. Doses were titrated at 2-week intervals from 1.0 mg three times daily to a maximum of 2.5 mg three times daily. Riociguat had a favourable safety profile, and also significantly improved exercise capacity and hemodynamic parameters such as pulmonary vascular resistance, cardiac output and pulmonary arterial pressure compared to baseline values.[17]
In addition, a phase II study of riociguat is underway in patients with other forms of PH such as associated with interstitial lung disease (PH-ILD).[18]
Phase III clinical trials
The phase III trials on riociguat are multi-center studies. The study program included large randomized, double-blind, placebo-controlled pivotal trial phase (CHEST-1 and PATENT-1), and open-label extensions of these studies (CHEST-2 and PATENT-2). Details of these studies are reported on ClinicalTrials.gov, a register of studies maintained by the National Institutes of Health (NIH).[16]
CHEST
The Chronic Thromboembolic Pulmonary Hypertension sGC-Stimulator Trial (CHEST) was a randomized, placebo-controlled trial aimed to analyse the efficacy and safety of riociguat in CTEPH patients.[19] After a 16-week riociguat treatment the patient's exercise capacity were evaluated by measuring the change in the six-minute walk test (6-MWT).[20] Patients having completed CHEST-1 were invited to enter the extension trial, CHEST-2. The first interim analysis of CHEST-2 showed that riociguat was well tolerated, with a good long-term safety profile in patients with CTEPH.[21]
PATENT
The Pulmonary Arterial Hypertension sGC-Stimulator Trial (PATENT) was a randomized, placebo-controlled trial that investigated the efficacy and safety of riociguat in PAH patients. After a 12-week treatment the patient's exercise capacity was evaluated by measuring the change in the 6-MWT.[22] Patients having completed PATENT-1 were invited to enter the extension trial, PATENT-2.[23] The first interim analysis of PATENT-2 showed that at one year, long-term riociguat was well tolerated in patients with PAH and showed sustained benefits in 6MWD and WHO FC. The safety profile of riociguat in PATENT-2 was similar to that observed in PATENT-1.
Other studies
Effect of riociguat on bone metabolism
This randomized, double blind, placebo controlled Phase I study investigated the effect of riociguat, administered as 2.5 mg immediate-release (IR)-tablets twice daily over 14 days, on the bone metabolism.[24] Effects on bone formation had been seen in growing, juvenile and adolescent rats. In juvenile rats, the changes consisted of thickening of trabecular bone and hyperostosis and remodeling of metaphyseal and diaphyseal bone, whereas in adolescent rats an overall increase of bone mass was observed. On the other hand, no such effects were observed in adult rats.
Interaction study with sildenafil
This study investigated safety, tolerability, pharmacokinetics and the impact on pulmonary and systemic haemodynamics of single doses of 0.5 and 1 mg of riociguat in patients with PAH and stable treatment of sildenafil (20 mg thrice daily) in a non-randomized uncontrolled trial.[25] The study showed potentially unfavorable safety signals with sildenafil plus riociguat and no evidence of a positive benefit/risk ratio. Therefore, the concomitant use of riociguat with phosphodiesterase-5 inhibitors is contraindicated.
See also
- Cinaciguat, a sGC activator (not sGC stimulator).
- PDE5 inhibitors act downstream in the nitric oxide signalling pathway, reducing cyclic GMP degradation.
- Endothelin receptor antagonist, another class of drugs used in PAH
References
- FDA. Retrieved 22 October 2023.
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2014". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ a b c d Riociguat FDA Professional Drug Information
- ^ "Background Riociguat". Bayer HealthCare. Archived from the original on 18 July 2011. Retrieved 15 December 2009.
- ^ "2022 First Generic Drug Approvals". U.S. Food and Drug Administration (FDA). 3 March 2023. Archived from the original on 30 June 2023. Retrieved 30 June 2023.
- ^ "Adempas not for use in patients with pulmonary hypertension caused by idiopathic interstitial pneumonia". European Medicines Agency. 24 June 2016.
- PMID 7540722.
- ^ PMID 16955067.
- ^ PMID 19129292.
- PMID 19089334.
- PMID 18550612.
- ^ S2CID 206433961.
- PMID 650406.
- S2CID 4402074.
- PMID 19263460.
- ^ a b ClinicalTrials.gov: Riociguat
- ^ "ATS International conference". American Thoracic Society. 2009. Archived from the original on 30 December 2009.
- ^ Clinical trial number NCT00694850 for "Impact of Multiple Doses of BAY 63-2521 on Safety, Tolerability, Pharmacokinetics and Pharmacodynamics in Patients With Interstitial Lung Disease (ILD) Associated Pulmonary Hypertension" at ClinicalTrials.gov
- PMID 23883377.
- ^ Clinical trial number NCT00855465 for "A Study to Evaluate Efficacy and Safety of Oral BAY63-2521 in Patients With CTEPH" at ClinicalTrials.gov
- ^ Clinical trial number NCT00910429 for "BAY63-2521 - Long-term Extension Study in Patients With Chronic Thromboembolic Pulmonary Hypertension" at ClinicalTrials.gov
- ^ Clinical trial number NCT00810693 for "A Study to Evaluate Efficacy and Safety of Oral BAY63-2521 in Patients With Pulmonary Arterial Hypertension (PAH)" at ClinicalTrials.gov
- ^ Clinical trial number NCT00863681 for "BAY63-2521:Long-term Extension Study in Patients With Pulmonary Arterial Hypertension" at ClinicalTrials.gov
- ^ Clinical trial number NCT00855660 for "Effect of Riociguat on Bone Metabolism" at ClinicalTrials.gov
- ^ Clinical trial number NCT00680654 for "Interaction Study in Patients With Pulmonary Hypertension and Stable Treatment of Sildenafil 20 mg TID" at ClinicalTrials.gov
External links
- "Riociguat". Drug Information Portal. U.S. National Library of Medicine.
