Nitrosamine
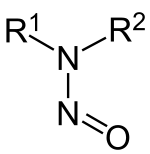
Nitrosamines (or more formally N-nitrosamines) are
Chemistry
The organic chemistry of nitrosamines is well developed with regard to their syntheses, their structures, and their reactions.
- HONO + R2NH → R2N-NO + H2O
The nitrous acid usually arises from protonation of a
With regards to structure, the C2N2O core of nitrosamines is planar, as established by
Nitrosamines are not directly carcinogenic. Metabolic activation is required to convert them to the
History and occurrence
In 1956, two British scientists, John Barnes and Peter Magee, reported that a simple member of the large class of N-nitrosamines,
Tobacco exposure
A common way ordinary consumers are exposed to nitrosamines is through tobacco use and cigarette smoke.
Dietary exposure
For example, during the 1970s, certain Norwegian farm animals began exhibiting elevated levels of liver cancer. These animals had been fed herring meal preserved with sodium nitrite. The sodium nitrite had reacted with dimethylamine in the fish and produced dimethylnitrosamine.[22]
The effects of nitroso compounds vary dramatically across the gastrointestinal tract, and with diet. Nitroso compounds present in stool do not induce nitrosamine formation, because stool has neutral
The formation process is inhibited when amine concentration is low (e.g. a low-protein diet or no fermented food). The process may also be inhibited in the case of high
Medication impurities
There have been recalls for various medications due to the presence of nitrosamine impurities. There have been recalls for angiotensin II receptor blockers, ranitidine, valsartan, duloxetine, and others.
The US Food and Drug Administration published guidance about the control of nitrosamine impurities in medicines.[32][33] Health Canada published guidance about nitrosamine impurities in medications[34] and a list of established acceptable intake limits of nitrosamine impurities in medications.[35]
Examples
| Substance name | CAS number
|
Synonyms | Molecular formula | Physical appearance | Carcinogenity category |
|---|---|---|---|---|---|
| N-Nitrosonornicotine | 16543-55-8 | NNN | C9H11N3O | Light yellow low-melting solid | |
4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone[36]
|
64091-91-4 | NNK, 4′-(nitrosomethylamino)-1-(3-pyridyl)-1-butanone | C10H15N3O2 | Light yellow oil | |
| N-Nitrosodimethylamine | 62-75-9 | Dimethylnitrosamine, N,N-dimethylnitrosamine, NDMA, DMN | C2H6N2O | Yellow liquid | EPA-B2; IARC-2A; TLV -A3
|
| N-Nitrosodiethylamine | 55-18-5 | Diethylnitrosamide, diethylnitrosamine, N,N-diethylnitrosamine, N-ethyl-N-nitrosoethanamine, diethylnitrosamine, DANA, DENA, DEN, NDEA | C4H10N2O | Yellow liquid | EPA-B2; IARC-2A |
| 4-(Methylnitrosamino)-1-(3-pyridyl)-1-butanol | 76014-81-8 | NNAL | |||
| N-Nitrosoanabasine | 37620-20-5 | NAB | C10H13N3O | Yellow Oil | IARC-3 |
| N-Nitrosoanatabine | 71267-22-6 | NAT | C10H11N3O | Clear yellow-to-orange oil | IARC-3 |
See also
- UDMH, are also carcinogenic.
- Possible health hazards of pickled vegetables
- Tobacco-specific nitrosamines
Additional reading
- Altkofer, Werner; Braune, Stefan; Ellendt, Kathi; Kettl-Grömminger, Margit; Steiner, Gabriele (2005). "Migration of nitrosamines from rubber products - are balloons and condoms harmful to the human health?". Molecular Nutrition & Food Research. 49 (3): 235–238. PMID 15672455.
- Proctor, Robert N. (2012). Golden Holocaust: Origins of the Cigarette Catastrophe and the Case for Abolition. Berkeley: University of California Press. OCLC 784884555.
References
- ^ California Water Boards, State Water Resources Control Board, "Nitrosamines", 09 December 2024
- PMID 33474939.
- ^ World Health Organization, Medical product alert, Note on Nitrosamine impurities, "Update on Nitrosamine impurities", 20 November 2019
- PMID 2272285.
- PMID 16865769.
- ^ PMID 2017213.
- ISBN 0-8412-0503-5.
- ^ Vogel, A. I. (1962). Practical Organic Chemistry (3rd ed.). Impression. p. 1074.
- ISBN 978-0-471-72091-1
- ^ Williams 1988, p. 142.
- ISBN 0-521-26796-X.
- ISBN 978-0-471-72091-1
- .
- ^ PMID 9625726.
- ISBN 978-0-12-407798-0.
- ^ Gregory N. Connolly; Howard Saxner (August 21, 2001). "Informational Update Research on Tobacco Specific Nitrosamines (TSNAs) in Oral Snuff and a Request to Tobacco Manufacturers to Voluntarily Set Tolerance Limits For TSNAs in Oral Snuff".
{{cite journal}}: Cite journal requires|journal=(help) - PMID 22062097.
- PMID 17052997.
- S2CID 4951579.
- S2CID 13274953.
- PMID 16865769.
- ISBN 978-1-4614-1586-2.
- PMID 7285009.
- PMID 17761300.
- ^ PMID 17785370.
- .
- S2CID 26275960.
- S2CID 196379002.
- S2CID 41045030. Retrieved 2015-06-06.
Evidence now exists that ascorbic acid is a limiting factor in nitrosation reactions in people.
- PMID 20026204.
- .
- ^ "Control of Nitrosamine Impurities in Human Drugs". U.S. Food and Drug Administration (FDA). 24 February 2021. Archived from the original on September 1, 2020.
- ^ "Control of Nitrosamine Impurities in Human Drugs - Guidance for Industry". Archived from the original on 2020-09-02.
- ^ "Nitrosamine impurities in medications: Guidance". Health Canada. 4 April 2022. Retrieved 29 March 2024.
- ^ "Nitrosamine impurities in medications: Established acceptable intake limits". Health Canada. 15 March 2024. Retrieved 29 March 2024.
- ^ Hecht, Steven S.; Borukhova, Anna; Carmella, Steven G. "Tobacco specific nitrosamines" Chapter 7; of "Nicotine safety and toxicity" Society for Research on Nicotine and Tobacco; 1998 - 203 pages
