Roussin's red salt

| |
| |
| Names | |
|---|---|
| IUPAC name
potassium tetranitrosyl-di-μ-sulfidodiiron(Fe–Fe)(2–)
| |
| Other names
Ferrate(2-), tetranitrosyldi-mu-thioxodi-, (Fe-Fe), dipotassium
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Fe2N4K2O4S2 | |
| Molar mass | 374.04 g/mol |
| Appearance | Dark red crystals |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Roussin's red salt is the
Structure and bonding
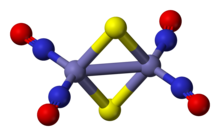
Roussin's red salt anion is an edge-shared bitetrahedron, wherein a pair Fe(NO)2 units are bridged by a pair of
Synthesis
The French chemist Z. Roussin
- Fe2I2(NO)4 + 2Li2S → Li2Fe2S2(NO)4 + 2LiI
To obtain the "esters", the salt is alkylated:
- Li2Fe2S2(NO)4 + 2 RX → Fe2(SR)2(NO)4 + 2 LiX
Esters can also be easily be prepared from the reaction of Fe2I2(NO)4 with the thiol.
Occurrence and potential applications
It is found in nature as its "esters" with the formula Fe2(SR)2(NO)4, where "R" is any alkyl group.[3] In addition Roussin's red salt is discussed in the fields of microbiology and food science due to its mutagenic properties.[8]
The ester derivative are being investigated as nitric oxide donors in biology and medicine, due to the relatively low toxicity and good stability of Roussin's red salt.[9]Photodissociation of the compound induces the release of NO, thereby sensitizing target cells to exposure to radiation.[8]
See also
References
- .
- ^ Roussin, M. L. (1858). "Recherches sur les nitrosulfures doubles de fer (nouvelle classe de sels)". Ann. Chim. Phys. 52: 285–303.
- ^ .
- doi:10.1039/B409519G.
- .
- .
- .
- ^ ISBN 0-7506-3365-4.
- S2CID 215408785.
