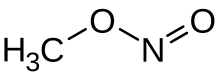
Methyl nitrite
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Methyl nitrite | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.009.882 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
SMILES
| |
| Properties | |
| CH3NO2 | |
| Molar mass | 61.040 g·mol−1 |
| Appearance | Yellow gas[1] |
| Density | 0.991 g/cm3[1] |
| Melting point | −16 °C (3 °F; 257 K)[1] |
| Boiling point | −12 °C (10 °F; 261 K)[1] |
| Thermochemistry[2] | |
Std enthalpy of (ΔfH⦵298)formation |
−66.1 kJ/mol |
| Hazards | |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Methyl nitrite is an
Structure
At room temperature, methyl nitrite exists as a mixture of cis and trans
 |

|
| cis-methyl nitrite | trans-methyl nitrite |
Synthesis
Methyl nitrite can be prepared by the reaction of
The figure shows the two gas-phase structures of methyl nitrite, as determined by IR and microwave spectroscopy.
Methyl nitrite free of nitromethane can be made by reacting iodomethane with nitrogen dioxide:
- 2 CH3I + 2 NO2 → 2 CH3ONO + I2
Properties and uses
Methyl nitrite is a precursor and intermediate, e.g. during production of phenylpropanolamine.[6]
Methyl nitrite is also present in aged cigarette smoke. Here it is presumably formed from nitrogen dioxide (itself formed by oxidation of nitric oxide) and methanol.[7]
Environmental impact
As one product of the combustion of
Safety
Methyl nitrite is a toxic asphyxiating gas, a potent cyanotic agent. Exposure may result in methemoglobinemia.[6]
Methyl nitrite is an oxidizing agent and a heat-sensitive explosive; its sensitivity increases in presence of metal oxides. With inorganic bases it forms explosive salts. It forms explosive mixtures with air. It is used as a rocket propellant, a monopropellant.[9] It explodes more violently than ethyl nitrite. Lower alkyl nitrites may decompose and burst the container even when stored under refrigeration.[10]
See also
References
- ^ a b c d Haynes, p. 3.382
- ^ Haynes, p. 5.20
- .
- ^ OCLC 236055357.
- ISBN 978-0-618-39941-3.
- ^ a b METHYL NITRITE – National Library of Medicine HSDB Database. Toxnet.nlm.nih.gov. Retrieved on 2019-03-10.
- ISBN 9781466515529.
- ISSN 0007-0335.
- ^ Methyl Nitrite. Cameochemicals.noaa.gov. Retrieved on 2019-03-10.
- ISBN 9781483162508.
Cited sources
- Haynes, William M., ed. (2011). ISBN 978-1439855119.

