Spectinomycin
 | |
| Clinical data | |
|---|---|
| Trade names | Trobicin |
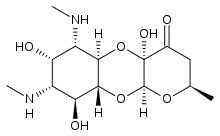
| Other names | (2R,4aR,5aR,6S,7S,8R,9S,9aR,10aS)-4a,7,9-trihydroxy-2-methyl-6,8-bis(methylamino)decahydro-4H-pyrano[2,3-b][1,4]benzodioxin-4-one , SPT/SPE/SC/SP[1] |
| AHFS/Drugs.com | Monograph |
| Routes of administration | IM |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| Melting point | 184 to 194 °C (363 to 381 °F) |
| |
| |
| | |
Spectinomycin, sold under the tradename Trobicin among others, is an
Common side effects include pain at the area of injection, rash, nausea, fever, and trouble sleeping.
Spectinomycin was discovered in 1961.
Medical uses
It is given by intramuscular injection to treat gonorrhea, especially in patients who are allergic to penicillins.
This antibiotic is no longer available in the United States for human use, but is still available for veterinary use.
Side effects
Side effects include
.Mechanism of action
Spectinomycin binds to the
Biosynthetic mechanism

Biosynthesis of spectinomycin begins similar to all aminoglycosides, with the formation of an
While all this happens, an alternate pathway is occurring creating the sugar functional group from a similar starting product. In this pathway,
Nature
It is in
History
Spectinomycin was discovered 1961.[3] A disruption in the supply occurred in 2001.[7]
References
- ^ "Antibiotic abbreviations list". Retrieved 22 June 2023.
- ^ a b c d e f g h i "Spectinomycin Hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 2015-09-24. Retrieved Sep 6, 2015.
- ^ ISBN 9781439882405. Archivedfrom the original on 2015-10-03.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- PMID 17371823.
- ISBN 9780470741689.
- PMID 11575327.
External links
- "Spectinomycin". Drug Information Portal. U.S. National Library of Medicine.
