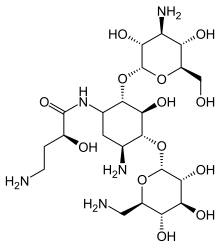
Amikacin
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Amikin, Amiglyde-V, Arikayce, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682661 |
| License data | |
| Pregnancy category |
|
inhalation | |
| Drug class | Aminoglycoside |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | >90%[8] |
| Protein binding | 0–11% |
| Metabolism | Mostly unmetabolized |
| Elimination half-life | 2–3 hours |
| Excretion | Kidney |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Amikacin is an
Amikacin, like other
Amikacin was patented in 1971, and came into commercial use in 1976.
Medical uses
Amikacin is most often used for treating severe infections with
It is often used in the following situations:[9]
- Bronchiectasis[17]
- Bone and joint infections
- Granulocytopenia, when combined with ticarcillin, in people with cancer[18]
- Intra-abdominal infections (such as peritonitis) as an adjunct to other medicines, like clindamycin, metronidazole, piperacillin/tazobactam, or ampicillin/sulbactam
- Meningitis:
- for meningitis by E. coli, as an adjunct to imipenem
- for meningitis caused by Pseudomonas, as an adjunct to meropenem
- for meningitis caused by Acinetobacter, as an adjunct to imipenem or colistin
- for neonatal meningitis caused by Streptococcus agalactiae or Listeria monocytogenes, as an adjunct to ampicillin
- for neonatal meningitis caused by Gram negative bacteria such as E. coli, as adjunct to a 3rd-generation cephalosporin
- Mycobacterial infections, including as a second-line agent for active M. chelonae, and M. fortuitum.
- Rhodococcus equi, which causes an infection resembling tuberculosis
- Respiratory tract infections, including as an adjunct to beta-lactams or carbapenem for hospital-acquired pneumonia
- Sepsis, including that in neonates,[14] as an adjunct to beta-lactams or carbapenem
- Skin and suture-site infections[14]
- Urinary tract infections that are caused by bacteria resistant to less toxic drugs (often by Enterobacteriaceae or P. aeruginosa)
Amikacin may be combined with a beta-lactam antibiotic for empiric therapy for people with neutropenia and fever.[9]
Available forms
A liposome inhalation suspension is also available and approved to treat Mycobacterium avium complex (MAC) in the United States,[20][5] and in the European Union.[6]
Amikacin liposome inhalation suspension is the first drug approved under the US limited population pathway for antibacterial and antifungal drugs (LPAD pathway).[20] It also was approved under the accelerated approval pathway.[20] The US Food and Drug Administration (FDA) granted the application for amikacin liposome inhalation suspension fast track, breakthrough therapy, priority review, and qualified infectious disease product (QIDP) designations.[20] The FDA granted approval of Arikayce to Insmed, Inc.[20]
The safety and efficacy of amikacin liposome inhalation suspension, an inhaled treatment taken through a nebulizer, was demonstrated in a randomized, controlled clinical trial where patients were assigned to one of two treatment groups.[20] One group of patients received amikacin liposome inhalation suspension plus a background multi-drug antibacterial regimen, while the other treatment group received a background multi-drug antibacterial regimen alone.[20] By the sixth month of treatment, 29 percent of patients treated with amikacin liposome inhalation suspension had no growth of mycobacteria in their sputum cultures for three consecutive months compared to 9 percent of patients who were not treated with amikacin liposome inhalation suspension.[20]
Special populations
Amikacin should be used in smaller doses in the elderly, who often have age-related decreases in kidney function, and children, whose kidneys are not fully developed yet. It is considered
In general, amikacin should be avoided in infants.[21] Infants also tend to have a larger volume of distribution due to their higher concentration of extracellular fluid, where aminoglycosides reside.[8]
The elderly tend to have amikacin stay longer in their system; while the average clearance of amikacin in a 20-year-old is 6 L/hr, it is 3 L/hr in an 80-year-old.[22]
Clearance is even higher in people with cystic fibrosis.[23]
In people with muscular disorders such as myasthenia gravis or Parkinson's disease, amikacin's paralytic effect on neuromuscular junctions can worsen muscle weakness.[9]
Adverse effects
Side-effects of amikacin are similar to those of other aminoglycosides. Kidney damage and ototoxicity (which can lead to hearing loss) are the most important effects, occurring in 1–10% of users.[17] The nephro- and ototoxicity are thought to be due to aminoglycosides' tendency to accumulate in the kidneys and inner ear.[8]
Amikacin can cause neurotoxicity if used at a higher dose or for longer than recommended. The resulting effects of neurotoxicity include
Amikacin causes nephrotoxicity (damage to the kidneys), by acting on the
Amikacin can cause neuromuscular blockade (including acute muscular paralysis) and respiratory paralysis (including apnea).[9]
Rare side effects (occurring in fewer than 1% of users) include allergic reactions,
The amikacin liposome inhalation suspension prescribing information includes a boxed warning regarding the increased risk of respiratory conditions including hypersensitivity pneumonitis (inflamed lungs), bronchospasm (tightening of the airway), exacerbation of underlying lung disease and hemoptysis (spitting up blood) that have led to hospitalizations in some cases.[20][5] Other common side effects in patients taking amikacin liposome inhalation suspension are dysphonia (difficulty speaking), cough, ototoxicity (damaged hearing), upper airway irritation, musculoskeletal pain, fatigue, diarrhea and nausea.[20][5]
Contraindications
Amikacin should be avoided in those who are sensitive to any aminoglycoside, as they are cross-allergenic (that is, an allergy to one aminoglycoside also confers hypersensitivity to other aminoglycosides). It should also be avoided in those sensitive to sulfite (seen more among people with asthma),[14] since most amikacin usually comes with sodium metabisulfite, which can cause an allergic reaction.[9]
In general, amikacin should not be used with or just before/after another drug that can cause neurotoxicity, ototoxicity, or nephrotoxicity. Such drugs include other aminoglycosides; the antiviral
Amikacin should not be used with
Interactions
Amikacin can be inactivated by other beta-lactams, though not to the extent as other aminoglycosides, and is still often used with
The effect of amikacin is increased when used with drugs derived from the
Potent diuretics not only cause ototoxicity themselves, but they can also increase the concentration of amikacin in the serum and tissue, making the ototoxicity even more likely.
Amikacin can decrease the effect certain vaccines, such as the live BCG vaccine (used for tuberculosis), the cholera vaccine, and the live typhoid vaccine by acting as a pharmacological antagonist.[17]
Pharmacology
Mechanism of action
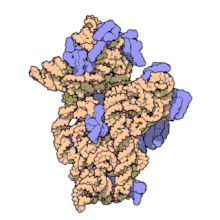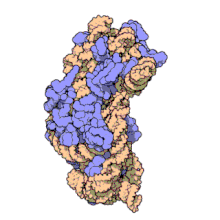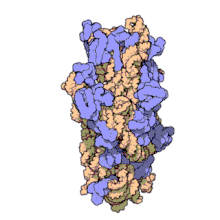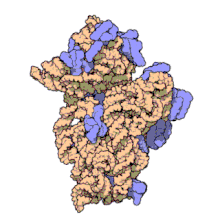
Amikacin irreversibly binds to
At normal doses, amikacin-sensitive bacteria respond within 24–48 hours.[14]
Resistance
Amikacin evades attacks by all antibiotic-inactivating enzymes that are responsible for
Bacteria that are resistant to streptomycin and capreomycin are still susceptible to amikacin; bacteria that are resistant to kanamycin have varying susceptibility to amikacin. Resistance to amikacin also confers resistance to kanamycin and capreomycin.[31]
Resistance to amikacin and kanamycin in Mycobacterium, the causative agent of tuberculosis, is due to a mutation in the rrs gene, which codes for the 16S rRNA. Mutations such as these reduce the binding affinity of amikacin to the bacteria's ribosome.
Pharmacokinetics
Amikacin is not absorbed orally and thus must be administered parenterally. It reaches peak serum concentrations in 0.5–2 hours when administered intramuscularly. Less than 11% of the amikacin actually binds to plasma proteins. It is distributed into the
While the half-life of amikacin is normally two hours, it is 50 hours in those with end-stage renal disease.[16]
The majority (95%) of amikacin from an intramuscular or intravenous dose is secreted unchanged via
Chemistry
Amikacin is derived from kanamycin A:[33][34]

Veterinary uses
While amikacin is only FDA-approved for use in dogs and for intrauterine infection in horses, it is one of the most common aminoglycosides used in veterinary medicine,
In dogs and cats, amikacin is commonly used as a topical antibiotic for
In horses, amikacin is FDA-approved for uterine infections (such as
Side effects in animals include nephrotoxicity, ototoxicity, and allergic reactions at IM injection sites. Cats tend to be more sensitive to the vestibular damage caused by ototoxicity. Less frequent side effects include neuromuscular blockade, facial edema, and peripheral neuropathy.[8][35]
The half-life in most animals is one to two hours.[43]
Treating overdoses of amikacin requires kidney dialysis or peritoneal dialysis, which reduce serum concentrations of amikacin, and/or penicillins, some of which can form complexes with amikacin that deactivate it.[8]
References
- ^ a b "Amikacin Use During Pregnancy". Drugs.com. 2 December 2019. Retrieved 13 March 2020.
- FDA. Retrieved 22 October 2023.
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ "Amikacin 250 mg/ml Injection - Summary of Product Characteristics (SmPC)". (emc). 16 September 2015. Retrieved 13 March 2020.
- ^ a b c d "Arikayce- amikacin suspension". DailyMed. 30 September 2018. Retrieved 13 March 2020.
- ^ a b "Arikayce liposomal EPAR". European Medicines Agency. 21 July 2020. Retrieved 4 March 2023.
- ^ "Arikayce liposomal Product information". Union Register of medicinal products. Retrieved 3 March 2023.
- ^ ISBN 978-0-470-95964-0.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z "Amikacin Sulfate". The American Society of Health-System Pharmacists. Archived from the original on 20 December 2016. Retrieved 8 December 2016.
- ISBN 9789241547659.
- ISBN 9783527607495. Archivedfrom the original on 20 December 2016.
- ISBN 9780191039621. Archivedfrom the original on 24 November 2015.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ a b c d e f g h i j k l "Amikacin sulfate injection, solution". DailyMed. 10 April 2019. Retrieved 13 March 2020.
- ISBN 978-0-19-975971-2. Archivedfrom the original on 10 September 2017.
- ^ S2CID 30373734.
- ^ a b c d e f g "amikacin (Rx)". Medscape. WebMD. Archived from the original on 9 August 2017. Retrieved 9 August 2017.
- ^ ISBN 978-0-444-53716-4.
- ISBN 978-0-12-411492-0.
- ^ a b c d e f g h i j "FDA approves a new antibacterial drug to treat a serious lung disease using a novel pathway to spur innovation". U.S. Food and Drug Administration (FDA) (Press release). Retrieved 12 November 2018.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ ISBN 978-1-4377-0282-8. Archivedfrom the original on 10 September 2017.
- ISBN 978-0-12-803348-7.
- ISBN 978-1-4918-0000-3. Archivedfrom the original on 10 September 2017.
- ^ ISBN 978-1-4557-7377-0. Archivedfrom the original on 10 September 2017.
- ^ ISSN 0378-6080.
- ISBN 978-0-321-91855-0.
- ^ "Amikacin". DrugBank. 2 August 2017. Archived from the original on 16 August 2017. Retrieved 10 August 2017.
- ^ a b Mudd E (7 August 2017). "O Aminoglycosides". Pharmacological Sciences. Archived from the original on 16 August 2017. Retrieved 14 August 2017.
- ^ S2CID 38981498.
- PMID 28365471.
- PMID 20797644.
- PMID 24216518.
- from the original on 16 August 2017.
- ISSN 0099-5428.
- ^ a b c d Forney B. "Amikacin for Veterinary Use". Wedgewood Pharmacy. Archived from the original on 16 August 2017. Retrieved 9 August 2017.
- ISBN 978-1-118-68590-7. Archivedfrom the original on 10 September 2017.
- ISBN 978-0-323-24293-6. Archivedfrom the original on 10 September 2017.
- ISBN 978-0-323-24196-0. Archivedfrom the original on 10 September 2017.
- ISBN 978-1-118-71416-4.
- ^ "Amiglyde-V- amikacin sulfate injection". DailyMed. U.S. National Library of Medicine. 9 March 2017. Archived from the original on 16 August 2017. Retrieved 8 August 2017.
- ISSN 0737-0806.
- ISBN 978-0-323-29170-5.
- ISBN 978-0-323-24485-5. Archivedfrom the original on 10 September 2017.
