Chugaev elimination
Source: Wikipedia, the free encyclopedia.
| Chugaev elimination | |
|---|---|
| Named after | Lev Chugaev |
| Reaction type | Elimination reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000538 |
The Chugaev elimination is a chemical reaction that involves the elimination of water from
Lev Aleksandrovich Chugaev
(1873-1922), who first reported the reaction sequence in 1899.

In the first step, a xanthate salt is formed out of the alkoxide and carbon disulfide (CS2). With the addition of iodomethane, the alkoxide is transformed into a methyl xanthate.

At about 200 °C, the alkene is formed by an intramolecular elimination. In a 6-membered cyclic transition state the hydrogen atom is removed from the carbon atom β to the xanthate oxygen in a syn-elimination. The side product decomposes to carbonyl sulfide (OCS) and methanethiol.
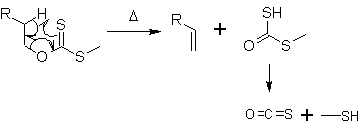
The Chugaev elimination is similar in mechanism to other thermal elimination reactions such as the
Cope elimination and ester pyrolysis
. Xanthates typically undergo elimination from 120 to 200 °C, while esters typically require 400 to 500 °C and amine oxides routinely react between 80 and 160 °C.
In the development of
See also
References
- .
- Latscha, Hans P. (2002). Chemie-Basiswissen. Berlin: Springer.
- .

