Aluminium nitride

| |
| |
| Names | |
|---|---|
| Other names
AlN
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.041.931 |
| EC Number |
|
| 13611 | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| AlN | |
| Molar mass | 40.989 g/mol[1] |
| Appearance | white to pale-yellow solid |
| Density | 3.255 g/cm3[1] |
| Melting point | 2,500 °C (4,530 °F; 2,770 K)[6] |
| hydrolyses (powder), insoluble (monocrystalline) | |
| Solubility | insoluble, subject of hydrolysis in water solutions of bases and acids [2] |
| Band gap | 6.015 eV[3][4] (direct) |
| Electron mobility | ~300 cm2/(V·s) |
Thermal conductivity
|
321 W/(m·K)[5] |
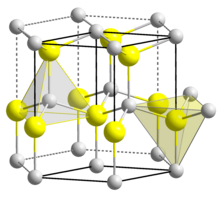
| Structure[7] | |
| Wurtzite | |
| C6v4-P63mc, No. 186, hP4 | |
a = 0.31117 nm, c = 0.49788 nm
| |
Formula units (Z)
|
2 |
| Tetrahedral | |
| Thermochemistry[8] | |
Heat capacity (C)
|
30.1 J/(mol·K) |
Std molar
entropy (S⦵298) |
20.2 J/(mol·K) |
Std enthalpy of (ΔfH⦵298)formation |
−318.0 kJ/mol |
Gibbs free energy (ΔfG⦵)
|
−287.0 kJ/mol |
| Hazards | |
| GHS labelling: | |
  
| |
| Warning | |
| H315, H319, H335, H373, H411 | |
| P260, P261, P264, P271, P280, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P332+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Aluminium nitride (
History and physical properties
AlN was first synthesized in 1862 by F. Briegleb and A. Geuther.[9][10]
AlN, in the pure (undoped) state has an
The material exists primarily in the hexagonal
One of the unique intrinsic properties of wurtzite AlN is its spontaneous polarization. The origin of spontaneous polarization is the strong ionic character of chemical bonds in wurtzite AlN due to the large difference in electronegativity between aluminium and nitrogen atoms. Furthermore, the non-centrosymmetric wurtzite crystal structure gives rise to a net polarization along the c-axis. Compared with other III-nitride materials, AlN has a larger spontaneous polarization due to the higher nonideality of its crystal structure (Psp: AlN 0.081 C/m2 > InN 0.032 C/m2 > GaN 0.029 C/m2).[13] Moreover, the piezoelectric nature of AlN gives rise to internal piezoelectric polarization charges under strain. These polarization effects can be utilized to induce a high density of free carriers at III-nitride semiconductor heterostructure interfaces completely dispensing with the need of intentional doping. Owing to the broken inversion symmetry along the polar direction, AlN thin film can be grown on either metal-polar or nitrogen-polar faces. Their bulk and surface properties depend significantly on this choice. The polarization effect is currently under investigation for both polarities.
Critical spontaneous and piezoelectric polarization constants for AlN are listed in the table below:[13][14]
| e31
(C/m2) |
e33
(C/m2) |
c13
(GPa) |
c33
(GPa) |
a0
(Å) |
c0
(Å) | |
| AlN | -0.60 | 1.46 | 108 | 373 | 3.112 | 4.982 |
AlN has high
AlN is one of the few materials that have both a wide and direct bandgap (almost twice that of SiC and GaN) and large thermal conductivity.[15] This is due to its small atomic mass, strong interatomic bonds, and simple crystal structure.[16] This property makes AlN attractive for application in high speed and high power communication networks. Many devices handle and manipulate large amounts of energy in small volumes and at high speeds, so due to the electrically insulating nature and high thermal conductivity of AlN, it becomes a potential material for high-power power electronics. Among group III-nitride materials, AlN has higher thermal conductivity compared to gallium nitride (GaN). Therefore, AlN is more advantageous than GaN in terms of heat dissipation in many power and radio frequency electronic devices.
Thermal expansivity is another critical property for high temperature applications. The calculated thermal expansion coefficients of AlN at 300 K are 4.2×10−6 K−1along a-axis and 5.3×10−6 K−1 along c-axis.[17]
Stability and chemical properties
Aluminium nitride is stable at high temperatures in inert atmospheres and melts at about 2,200 °C (2,470 K; 3,990 °F). In a vacuum, AlN decomposes at ~1,800 °C (2,070 K; 3,270 °F). In the air, surface oxidation occurs above 700 °C (973 K; 1,292 °F), and even at room temperature, surface oxide layers of 5–10 nm thickness have been detected. This oxide layer protects the material up to 1,370 °C (1,640 K; 2,500 °F). Above this temperature bulk oxidation occurs. Aluminium nitride is stable in hydrogen and carbon-dioxide atmospheres up to 980 °C (1,250 K; 1,800 °F).[18]
The material dissolves slowly in mineral acids through grain-boundary attack and in strong alkalies through attack on the aluminium-nitride grains. The material hydrolyzes slowly in water. Aluminium nitride is resistant to attack from most molten salts, including chlorides and cryolite.[19]
Aluminium nitride can be patterned with a Cl2-based
Manufacture
AlN is synthesized by the
Applications
AlN is also used to build piezoelectric micromachined ultrasound transducers, which emit and receive ultrasound and which can be used for in-air rangefinding over distances of up to a meter.[26][27]
Metallization methods are available to allow AlN to be used in electronics applications similar to those of alumina and beryllium oxide. AlN nanotubes as inorganic quasi-one-dimensional nanotubes, which are isoelectronic with carbon nanotubes, have been suggested as chemical sensors for toxic gases.[28][29]
Currently there is much research into developing light-emitting diodes to operate in the ultraviolet using gallium nitride based semiconductors and, using the alloy aluminium gallium nitride, wavelengths as short as 250 nm have been achieved. In 2006, an inefficient AlN LED emission at 210 nm was reported.[30]
AlN-based high electron mobility transistors (HEMTs) have attracted a high level of attention due to AlN’s superior properties, such as better thermal management, reduced buffer leakage, and excellent integration for all nitride electronics. AlN buffer layer is a critical building block for AlN-based HEMTs, and it has been grown by using MOCVD or MBE on different substrates. Building on top of AlN buffer, n-channel devices with 2D electron gas (2DEG) and p-channel devices with 2D hole gas (2DHG) have been demonstrated. The combination of high-density 2DEG and 2DHG on the same semiconductor platform makes it a potential candidate for CMOS devices.
Among the applications of AlN are
- opto-electronics,
- dielectric layers in optical storage media,
- electronic substrates, chip carriers where high thermal conductivity is essential,
- military applications,
- as a crucible to grow crystals of gallium arsenide,
- steel and semiconductor manufacturing.
See also
- Boron nitride
- Aluminium phosphide
- Aluminium arsenide
- Aluminium antimonide
- Gallium nitride
- Indium nitride
- Aluminium oxynitride
- Titanium aluminium nitride, TiAlN or AlTiN
References
- ^ a b Haynes, p. 4.45.
- S2CID 91552821.
- ^ Haynes, p. 12.85.
- doi:10.1103/physrevb.82.075208.)
{{cite journal}}: CS1 maint: untitled periodical (link - ^ S2CID 207780348. Retrieved 2020-04-03.
- ^ Haynes, p. 12.80.
- .
- ^ Haynes, p. 5.4.
- ISBN 978-966-8449-53-6.
- .
- ^ a b c "AlN – Aluminium Nitride". Ioffe Database. Sankt-Peterburg: FTI im. A. F. Ioffe, RAN. Retrieved 2014-01-01.
- ISSN 1984-6428.
- ^ S2CID 250782290.
- ISSN 0021-8979.
- S2CID 233936255.
- S2CID 90262793.
- ISSN 0021-8979.
- ISBN 978-0-8493-8912-2.
- S2CID 137160554.
- S2CID 20028149.
- S2CID 118571039.
- ISSN 0002-7820. Retrieved 2023-06-26.
- ISSN 2365-709X.
- ^ Tsuruoka, Doug (2014-03-17). "Apple, Samsung Cellphone Filter Orders Lift Avago". Investor's Business Daily.
- ^ "ACPF-7001: Agilent Technologies Announces FBAR Filter for U.S. PCS Band Mobile Phones and Data Cards". wirelessZONE. EN-Genius Network Ltd. 2002-05-27. Retrieved 2008-10-18.
- ^ "A Gestural Interface for Smart Watches".
- ^ Przybyla, R.; al, et (2014). "3D Ultrasonic Gesture Recognition". International Solid State Circuits Conference. San Francisco. pp. 210–211.
- .
- .
- S2CID 4373542.
Cited sources
- Haynes, William M., ed. (2016). ISBN 9781498754293.

