Chlorine azide
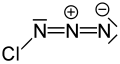
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Chlorine azide
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| Properties | |||
| ClN3 | |||
| Molar mass | 77.4731 g/mol | ||
| Appearance | Yellow-orange liquid; colorless gas | ||
| Melting point | −100 °C (−148 °F; 173 K) | ||
| Boiling point | −15 °C (5 °F; 258 K) | ||
| Solubility | Soluble[vague] in butane, pentane, benzene, methanol, ethanol, diethyl ether, acetone, chloroform, carbon tetrachloride, and carbon disulfide; slightly soluble in water | ||
| Structure | |||
| orthorhombic | |||
| Cmc 21, No. 36[1] | |||
| Explosive data | |||
| Shock sensitivity | Extreme | ||
| Friction sensitivity | Extreme | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Extremely sensitive explosive | ||
| NFPA 704 (fire diamond) | |||
| Related compounds | |||
Related compounds
|
Hydrazoic acid Fluorine azide Bromine azide Iodine azide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Chlorine azide (ClN3) is an inorganic compound that was discovered in 1908 by Friedrich Raschig.[2] Concentrated ClN3 is notoriously unstable and may spontaneously detonate at any temperature.[3]
Preparation and handling
Chlorine azide is prepared by passing
chlorine gas over silver azide, or by an addition of acetic acid to a solution of sodium hypochlorite and sodium azide.[4]
Explosive characteristics
Chlorine azide is extremely sensitive. It may explode, sometimes even without apparent provocation; it is thus too sensitive to be used commercially unless first diluted in solution. Chlorine azide reacts explosively with
ethene, methane, propane, phosphorus, silver azide, and sodium. On contact with acid, chlorine azide decomposes, evolving toxic and corrosive hydrogen chloride gas.[5]
Regulatory information
Its shipment is subject to strict reporting requirements and regulations by the
US Department of Transportation
.
References
External links
 Media related to Chlorine azide at Wikimedia Commons
Media related to Chlorine azide at Wikimedia Commons



