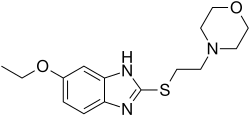
Fabomotizole
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Afobazole |
| Other names | Obenoxazine |
| Routes of administration | Oral |
| Legal status | |
| Legal status |
|
first-pass effect | |
| Metabolism | extensive hepatic |
| Onset of action | 0.85±0.13 hours |
| Elimination half-life | 0.82±0.54 hours |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |

Fabomotizole (
receptor antagonism, and sigma agonism suggested as potential mechanisms. Fabomotizole was shown to inhibit MAO-A reversibly and there might be also some involvement with serotonin receptors.[2][3][4][5][6] Clinical trials have shown fabomotizole to be well tolerated and reasonably effective for the treatment of anxiety.[7]
Experiments of mice have shown antimutagenic and antiteratogenic properties.[8]
Fabomotizole has found little clinical use outside Russia and has not been evaluated by the FDA.
