Clomipramine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Anafranil, Clomicalm, others |
| Other names | Clomipramine; 3-Chloroimipramine; G-34586[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697002 |
| License data |
|
| Pregnancy category |
|
intravenous[2] | |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Clomipramine, sold under the brand name Anafranil among others, is a
It may also address certain fundamental features surrounding narcolepsy besides cataplexy (especially hypnagogic and hypnopompic hallucinations).[12] The evidence behind this, however, is less robust.
As with other antidepressants (notably including
It is typically taken by mouth, although intravenous preparations are sometimes used.[13][14]
Common side effects include dry mouth, constipation, loss of appetite, sleepiness, weight gain, sexual dysfunction, and trouble urinating.
Clomipramine was discovered in 1964 by the Swiss drug manufacturer
Medical uses
Clomipramine has a number of uses in medicine, including in the treatment of:
- Obsessive–compulsive disorder (OCD), which happens to be its only U.S. FDA-labeled indication.[17][18] Other regulatory agencies (such as the TGA of Australia and the MHRA of the UK) have also approved clomipramine for this indication.[19][20][21][22]
- Major depressive disorder (MDD), a popular off-label use in the US. It is approved by the Australian TGA and the United Kingdom MHRA for this indication. In Japan it is also approved for depression.[19][20][21][22][23][24] Some have suggested the possible superior efficacy of clomipramine compared to other antidepressants in the treatment of MDD,[25][26][27][28][29] especially the more severe,[30][31][32] although at the current time the evidence may be insufficient to more fully substantiate this claim.[33]
- Panic disorder with or without agoraphobia.[34][35]
- Body dysmorphic disorder[36]
- Repetitive self-injurious/self-harming behaviours in those with intellectual disability specifically.[37][38]
- The subtype of systemised paranoia characterised by somatic phenomena.[39][40]
- Compulsive nail-biting (onychophagia).[41]
- Cataplexy associated with narcolepsy. This is a TGA and MHRA-labeled indication for clomipramine.[21][22]
- Self-bloodletting[42]
- Premature ejaculation,[43] where it may be more effective than paroxetine[44]
- Depersonalization disorder[45]
- Chronic pain with or without organic disease, particularly headache of the tension type.[46]
- Developmental stuttering[47][48]
- Sleep paralysis, with or without narcolepsy
- Enuresis (involuntary urinating in sleep) in children. The effect may not be sustained following treatment, and alarm therapy may be more effective in both the short-term and the long-term.[49] Combining a tricyclic (such as clomipramine) with anticholinergic medication may be more effective for treating enuresis than the tricyclic alone.[49]
- Trichotillomania[50][51][52]
- In combination with The overall risk of seizures may have to be weighted against the refractory severity of the current illness and necessity of the amalgamation of treatment(s).
Although lithium is most-associated with the treatment of
In a
Contraindications
Contraindications include:[20]
- Known hypersensitivity to clomipramine, or any of the excipients or cross-sensitivity to tricyclic antidepressants of the dibenzazepine group
- Recent myocardial infarction
- Any degree of heart block or other cardiac arrhythmias
- Mania
- Severe liver disease
- Narrow angle glaucoma
- Untreated urinary retention
- It must not be given in combination or within 3 weeks before or after treatment with a monoamine oxidase inhibitor. (Moclobemide included; however, clomipramine may be initiated sooner at 48 hours following discontinuation of moclobemide.)
Pregnancy and lactation
Clomipramine use during pregnancy is associated with congenital heart defects in the newborn.[22][67] It is also associated with reversible withdrawal effects in the newborn.[68] Clomipramine is also distributed in breast milk and hence nursing while taking clomipramine is advised against.[18]
Side effects
Clomipramine has been associated with the side effects listed below:[17][18][19][20]
Very common (>10% frequency):
- Accommodationdefect
- Blurred vision
- Nausea
- Dry mouth (Xerostomia)
- Constipation
- Fatigue
- Dizziness
- Tremor
- Headache
- Myoclonus
- Drowsiness
- Somnolence
- Restlessness
- Micturition disorder
- Sexual dysfunction (erectile dysfunction and loss of libido)
- Hyperhidrosis (profuse sweating)
Common (1–10% frequency):
- Weight loss
- Dystonia
- Cognitive impairment
- Orthostatic hypotension
- Sinus tachycardia
- Clinically irrelevant ECG changes (e.g. T- and ST-wave changes) in patients of normal cardiac status
- Palpitations
- Tinnitus (hearing ringing in one's ears)
- Mydriasis (dilated pupils)
- Vomiting
- Abdominal disorders
- Diarrhoea
- Decreased appetite
- Increased transaminases
- Increased Alkaline phosphatase
- Speech disorders
- Paraesthesia
- Muscle hypertonia
- Dysgeusia
- Memory impairment
- Muscular weakness
- Disturbance in attention
- Confusional state
- Disorientation
- Hallucinations (particularly in elderly patients and patients with Parkinson's disease)
- Anxiety
- Agitation
- Sleep disorders
- Mania
- Hypomania
- Aggression
- Depersonalisation
- Insomnia
- Nightmares
- Aggravation of depression
- Delirium
- Galactorrhoea(lactation that is not associated with pregnancy or breastfeeding)
- Yawning
- Hot flush
- Dermatitis allergic (skin rash, urticaria)
- Photosensitivity reaction
- Pruritus (itching)
Uncommon (0.1–1% frequency):
- Convulsions
- Ataxia
- Arrhythmias
- Elevated blood pressure
- Activation of psychotic symptoms
Very rare (<0.01% frequency):
- Pancytopaenia— an abnormally low amount of all the different types of blood cells in the blood (including platelets, white blood cells and red blood cells).
- Leukopenia — a low white blood cell count.
- Agranulocytosis — a more severe form of leukopenia; a dangerously low neutrophil count which leaves one open to life-threatening infections due to the role of the white blood cells in defending the body from invaders.
- plateletsin the blood which are essential to clotting and hence this leads to an increased tendency to bruise and bleed, including, potentially, internally.
- Eosinophilia — an abnormally high number of eosinophils — the cells that fight off parasitic infections — in the blood.
- hypocalcaemia[low blood calcium]).
- Glaucoma
- Oedema(local or generalised)
- Alopecia(hair loss)
- Hyperpyrexia(a high fever that is above 41.5 °C)
- Hepatitis (liver swelling) with or without jaundice — the yellowing of the eyes, the skin, and mucous membranes due to impaired liver function.
- Abnormal ECG
- Anaphylactic and anaphylactoid reactions including hypotension
- Neuroleptic malignant syndrome (NMS) — a potentially fatal side effect of antidopaminergic agents such as antipsychotics, tricyclic antidepressants and antiemetics (drugs that relieve nausea and vomiting). NMS develops over a period of days or weeks and is characterised by the following symptoms:
- Tremor
- Muscle rigidity
- Mental status change (such as confusion, delirium, mania, hypomania, agitation, coma, etc.)
- Hyperthermia (high body temperature)
- Tachycardia (high heart rate)
- Blood pressure changes
- Diaphoresis(sweating profusely)
- Diarrhoea
- Alveolitis allergic (pneumonitis) with or without eosinophilia
- Purpura
- Conduction disorder (e.g. widening of hypokalaemia)
Individual side-effects may or may not be amendable to treatment. As noted below, bethanechol may alleviate anti-muscarinic/anti-cholinergic side-effects. It may also treat sexual side-effects common to clomipramine and SSRIs.[69][70]
Topiramate has been used to off-set the weight-gain induced from various antidepressants and antipsychotics,[71] and more broadly for general weight-loss (likewise with bupropion).[71] This option may be especially attractive in patients either overweight prior to clomipramine treatment or who have gained an undesirable amount of weight on it, as the weight-loss associated with topiramate may be very impressive indeed.[72][73][74][75][76]
Another potential advantage of topiramate in the adjunctive treatment of people taking clomipramine is engendered in its status as an anti-convulsant medication, thereby theoretically increasing the seizure-threshold in patients (which clomipramine decreases to an extent which precludes its dosage ranging above 250 m.g./d. in normal circumstances, likewise with
Tremor may be relieved with a beta-blocker (e.g.,
Withdrawal
Withdrawal symptoms may occur during gradual or particularly abrupt withdrawal of tricyclic antidepressant drugs. Possible symptoms include: nausea, vomiting, abdominal pain, diarrhea, insomnia, headache, nervousness, anxiety, dizziness and worsening of psychiatric status.
Overdose
Clomipramine overdose usually presents with the following symptoms:[17][19][20]
- Signs of central nervous system depression such as:
- stupor
- coma
- drowsiness
- restlessness
- ataxia
- Mydriasis
- Convulsions
- Enhanced reflexes
- Muscle rigidity
- Athetoid and choreoathetoid movements
- Serotonin syndrome - a condition with many of the same symptoms as neuroleptic malignant syndrome but has a significantly more rapid onset
- Cardiovascular effects including:
- arrhythmias (including Torsades de pointes)
- tachycardia
- QTc intervalprolongation
- conduction disorders
- hypotension
- shock
- heart failure
- cardiac arrest
- Apnoea
- Cyanosis
- Respiratory depression
- Vomiting
- Fever
- Sweating
- Oliguria
- Anuria
There is no specific antidote for overdose and all treatment is purely supportive and symptomatic.
Interactions
Clomipramine may interact with a number of different medications, including the
Fluvoxamine increases the serotoninergic effects of clomipramine and, likewise, clomipramine increases fluvoxamine levels.[90]
Pharmacology
Pharmacodynamics
| Site | CMI | DMC |
Species | Ref |
|---|---|---|---|---|
| SERT | 0.14–0.28 | 40 | Human/rat | [92][93][94] |
| NET | 38–53.7 | 0.32 | Human/rat | [92][93][94] |
| DAT | ≥2,190 | 2,100 | Human/rat | [92][93][94] |
| 5-HT1A | ≥7,000 | 19,000 | Human/und | [95][93][94] |
| 5-HT1B | >10,000 | ND | Human | [93] |
| 5-HT1D | >10,000 | ND | Human | [93] |
| 5-HT2A | 27–35.5 | 130 | Human/und | [95][93][94] |
| 5-HT2B | ND | ND | ND | ND |
| 5-HT2C | 64.6 | ND | Human | [93] |
| 5-HT3 | 460–985 | ND | Rodent | [93][96][97] |
| 5-HT6 | 53.8 | ND | Rat | [98] |
| 5-HT7 | 127 | ND | Rat | [99] |
| α1 | 3.2–38 | 190 | Human/und | [93][100][94] |
| α2 | 525–3,200 | 1,800 | Human/und | [93][100][94] |
β |
22,000 | 16,000 | Undefined | [94] |
D1 |
219 | 320 | Human/und | [96][94] |
D2 |
77.6–190 | 1,200 | Human/und | [93][100][94] |
D3 |
30–50.1 | ND | Human | [93][96] |
D4 |
ND | ND | ND | ND |
D5 |
ND | ND | ND | ND |
| H1 | 13–31 | 450 | Human/und | [101][100][94] |
| H2 | 209 | ND | Human | [101] |
| H3 | 9,770 | ND | Human | [101] |
| H4 | 5,750 | ND | Human | [101] |
mACh |
37 | 92 | Human/und | [100][94] |
| σ1 | 546 | ND | Rat | [102] |
hERG |
130 ( IC50 ) |
ND | Human | [103] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. | ||||
Clomipramine is a
Probably all “anticholinergic” side-effects may be successfully reversed in a majority of people with bethanechol chloride,[106][107][108] although knowledge of this amenability has unfortunately decreased in medical circles over the decades. It (bethanechol supplementation) arguably should, however, be seriously entertained when tricyclics which often carry significant anti-muscarinic effects (amitriptyline, protriptyline, imipramine, clomipramine) are prescribed, as it may alleviate potentially otherwise-limiting side-effects (blurry vision, dry mouth, urinary hesitancy/retention, etc.). This practice can make drugs of otherwise indispensably potent value more tolerable to certain patients and spare them needless suffering, hence-reducing the overall side-effect burden or concern thereof.
Although clomipramine shows around 100- to 200-fold preference in affinity for the
The antidepressant effects of clomipramine are thought to be due to reuptake inhibition of serotonin and norepinephrine,
The exceptionally strong serotonin reuptake inhibition of clomipramine likely precludes the possibility of its antagonism of
Serotonergic activity
| Medication | SERT | NET | Dosage (mg/day) |
t1/2 (M)(hours) |
Cp (ng/mL) |
Cp / SERT ratio |
|---|---|---|---|---|---|---|
| Amitriptyline | 4.3 | 34.5 | 100–200 | 16 (30) | 100–250 | 23–58 |
| Amoxapine | 58.5 | 16.1 | 200–300 | 8 (30) | 200–500 | 3.4–8.5 |
| Butriptyline[92] | 1,360 | 5,100 | ? | ? | ? | ? |
| Clomipramine | 0.14–0.28 | 37 | 100–200 | 32 (70) | 150–500 | 536–3,570 |
| Desipramine | 17.5 | 0.8 | 100–200 | 30 | 125–300 | 7.1–17 |
| Dosulepin[119][120][121] | 8.3 | 45.5 | 150–225 | 25 (34) | 50–200 | 6.0–24 |
| Doxepin | 66.7 | 29.4 | 100–200 | 18 (30) | 150–250 | 2.2–3.7 |
| Imipramine | 1.4 | 37 | 100–200 | 12 (30) | 175–300 | 125–214 |
| Iprindole[92] | 1,620 | 1,262 | ? | ? | ? | ? |
| Lofepramine[92] | 70.0 | 5.4 | ? | ? | ? | ? |
| Nortriptyline | 18.5 | 4.4 | 75–150 | 31 | 60–150 | 3.2–8.1 |
| Protriptyline | 19.6 | 1.4 | 15–40 | 80 | 100–250 | 5.1–13 |
| Trimipramine[92] | 149 | 2,450 | 75–200 | 16 (30) | 100–300 | 0.67–2.0 |
| Citalopram | 1.4 | 5,100 | 20–40 | 36 | 75–150 | 54–107 |
| Escitalopram | 1.1 | 7,840 | 10–20 | 30 | 40–80 | 36–73 |
| Fluoxetine | 0.8 | 244 | 20–40 | 53 (240) | 100–500 | 125–625 |
| Fluvoxamine | 2.2 | 1,300 | 100–200 | 18 | 100–200 | 45–91 |
| Paroxetine | 0.34 | 40 | 20–40 | 17 | 30–100 | 300–1,000 |
| Sertraline | 0.4 | 417 | 100–150 | 23 (66) | 25–50 | 83–167 |
| Duloxetine | 1.6 | 11.2 | 80–100 | 11 | ? | ? |
| Milnacipran | 123 | 200 | ? | ? | ? | ? |
| Venlafaxine | 9.1 | 535 | 75–225 | 5 (11) | ? | ? |
| The values for the SERT and NET are Ki (nM). Note that in the Cp / SERT ratio, free versus protein-bound drug concentrations are not accounted for. | ||||||
| Medication | Dosage range (mg/day)[122] |
~80% SERT occupancy (mg/day)[123][124] |
Ratio (dosage / 80% occupancy) |
|---|---|---|---|
| Citalopram | 20–40 | 40 | 0.5–1 |
| Escitalopram | 10–20 | 10 | 1–2 |
| Fluoxetine | 20–80 | 20 | 1–4 |
| Fluvoxamine | 50–300 | 70 | 0.71–5 |
| Paroxetine | 10–60 | 20 | 0.5–3 |
| Sertraline | 25–200 | 50 | 0.5–4 |
| Duloxetine | 20–60 | 30 | 0.67–2 |
| Venlafaxine | 75–375 | 75 | 1–5 |
| Clomipramine | 50–250 | 10 | 5–25 |
Clomipramine is an extremely strong SRI by all accounts.
A positron emission tomography study found that a single low dose of 10 mg clomipramine to healthy volunteers resulted in 81.1% occupancy of the SERT, which was comparable to the 84.9% SERT occupancy by 50 mg fluvoxamine.[112] In the study, single doses of 5 to 50 mg clomipramine resulted in 67.2 to 94.0% SERT occupancy while single doses of 12.5 to 50 mg fluvoxamine resulted in 28.4 to 84.9% SERT occupancy.[112] Chronic treatment with higher doses was able to achieve up to 100.0% SERT occupancy with clomipramine and up to 93.6% SERT occupancy with fluvoxamine.[112] Other studies have found 83% SERT occupancy with 20 mg/day paroxetine and 77% SERT occupancy with 20 mg/day citalopram.[112][131] These results indicate that very low doses of clomipramine are able to substantially occupy the SERT and that clomipramine achieves higher occupancy of the SERT than SSRIs at comparable doses.[112][123] Moreover, clomipramine may be able to achieve more complete occupancy of the SERT at high doses, at least relative to fluvoxamine.[112]
If the ratios of the 80% SERT occupancy dosage and the approved clinical dosage range are calculated and compared for SSRIs, SNRIs, and clomipramine, it can be deduced that clomipramine is by far the strongest SRI used medically.
Obsessive–compulsive disorder
Clomipramine was the first drug that was investigated for and found to be effective in the treatment of OCD.[15][133] In addition, it was the first drug to be approved by the FDA in the United States for the treatment of OCD.[134] The effectiveness of clomipramine in the treatment of OCD is far greater than that of other TCAs, which are comparatively weak SRIs; a meta-analysis found pre- versus post-treatment effect sizes of 1.55 for clomipramine relative to a range of 0.67 for imipramine and 0.11 for desipramine.[135] In contrast to other TCAs, studies have found that clomipramine and SSRIs, which are more selective SRIs, have similar effectiveness in the treatment of OCD.[135] However, multiple meta-analyses have found that clomipramine nonetheless retains a significant effectiveness advantage relative to SSRIs;[136] in the same meta-analysis mentioned previously, the effect sizes of SSRIs in the treatment of OCD ranged from 0.81 for fluoxetine to 1.36 for sertraline (relative to 1.55 for clomipramine).[135] However, the effectiveness advantage for clomipramine has not been apparent in head-to-head comparisons of clomipramine versus SSRIs for OCD.[136] The differences in effectiveness findings could be due to differences in methodologies across non-head-to-head studies.[135][136]
Relatively high doses of SSRIs are needed for effectiveness in the treatment of OCD.
In addition to serotonin reuptake inhibition, clomipramine is also a mild but clinically significant antagonist of the
Although clomipramine is probably more effective in the treatment of OCD compared to SSRIs, it is greatly inferior to them in terms of
Pharmacokinetics
The
Although the normal maximum-recommended total daily dosage of clomipramine is 250 milligrams, treatment-resistant cases of depression
Dose-increases between 25 m.g. and 150 m.g., barring significant drug-drug interactions which may elevate clomipramine blood-levels, should be titrated in doses of 50 m.g. (25 m.g. in the case of
Chemistry
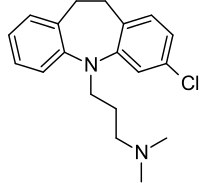
Clomipramine is a
History
Clomipramine was developed by
Clomipramine was eventually approved in the United States for the treatment of OCD in 1989 and became available in 1990.
Society and culture
Generic names
Clomipramine is the
Brand names
Clomipramine is marketed throughout the world mainly under the brand names Anafranil and Clomicalm for use in humans and animals, respectively.[159][169]
Veterinary uses
In the U.S., clomipramine is only licensed to treat separation anxiety in dogs for which it is sold under the brand name Clomicalm.[170] It has proven effective in the treatment of obsessive–compulsive disorders in cats and dogs.[171][172] In dogs, it has also demonstrated similar efficacy to fluoxetine in treating tail chasing.[173] In dogs some evidence suggests its efficacy in treating noise phobia.[174]
Clomipramine has also demonstrated efficacy in treating
References
- ^ ISBN 978-1-4757-2085-3. Archivedfrom the original on 15 February 2017.
- ISBN 978-0-470-97948-8.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ ISBN 978-1-60913-345-0. Archivedfrom the original on 8 September 2017.
- ^ a b c d e f g h i j "Clomipramine Hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 8 December 2015. Retrieved 2 December 2015.
- ^ Swedo, S E et al. “A double-blind comparison of clomipramine and desipramine in the treatment of trichotillomania (hair pulling).” The New England journal of medicine vol. 321,8 (1989): 497-501. doi:10.1056/NEJM198908243210803
- ^ Adebayo, Kazeem Olaide et al. “Body dysmorphic disorder in a Nigerian boy presenting as depression: a case report and literature review.” International journal of psychiatry in medicine vol. 44,4 (2012): 367-72. doi:10.2190/PM.44.4.f
- ^ Phillips, K.A., Albertini, R.S., Siniscalchi, J.M., Khan, A. and Robinson, M., 2001. Effectiveness of pharmacotherapy for body dysmorphic disorder: a chart-review study. Journal of Clinical Psychiatry, 62(9), pp.721-727.
- ^ Pallanti, S. and Koran, L.M., 1996. Intravenous, pulse-loaded clomipramine in body dysmorphic disorder: two case reports. CNS Spectrums, 1(2), pp.54-57.
- ^ Shapiro, W. R. (1975). Treatment of cataplexy with clomipramine. Archives of neurology, 32(10), 653-656.
- ^ Dauvilliers Y, Siegel JM, Lopez R, Torontali ZA, Peever JH. Cataplexy—clinical aspects, pathophysiology and management strategy. Nature Reviews Neurology. 2014 Jul;10(7):386-95..
- ^ Chen C. N. (1980). The use of clomipramine as an REM sleep suppressant in narcolepsy. Postgraduate medical journal, 56 Suppl 1, 86–89.
- ^ Karameh, Wael Karameh, and Munir Khani. “Intravenous Clomipramine for Treatment-Resistant Obsessive-Compulsive Disorder.” The international journal of neuropsychopharmacology vol. 19,2 pyv084. 28 July 2015, doi:10.1093/ijnp/pyv084
- ^ Sallee, F R et al. “Pulse intravenous clomipramine for depressed adolescents: double-blind, controlled trial.” The American journal of psychiatry vol. 154,5 (1997): 668-73. doi:10.1176/ajp.154.5.668
- ^ ISBN 978-1-118-30801-1.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ a b c d e f g h i "ANAFRANIL (CLOMIPRAMINE HYDROCHLORIDE) CAPSULE [MALLINCKRODT, INC.]". DailyMed. Mallinckrodt, Inc. October 2012. Archived from the original on 3 December 2013. Retrieved 30 November 2013.
- ^ a b c d e f g h "Anafranil (clomipramine) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Archived from the original on 3 December 2013. Retrieved 30 November 2013.
- ^ a b c d e f g h i j "ANAFRANIL (clomipramine)" (PDF). TGA eBusiness Services. NOVARTIS Pharmaceuticals Australia Pty Limited. 7 December 2012. Archived from the original on 12 August 2017. Retrieved 30 November 2013.
- ^ a b c d e "Anafranil 75mg SR Tablets - Summary of Product Characteristics (SPC) - (eMC)". electronic Medicines Compendium. Novartis Pharmaceuticals UK Ltd. 8 October 2012. Archived from the original on 14 July 2014. Retrieved 2 July 2014.
- ^ ISBN 978-0-85711-084-8.
- ^ ISBN 978-0-9805790-9-3.
- ^ "Clomipramine 50 mg Capsules, Hard - Summary of Product Characteristics (SmPC) - (eMC)". www.medicines.org.uk. Retrieved 27 January 2019.
- ^ "Kusuri-no-Shiori(Drug Information Sheet)". www.rad-ar.or.jp. Retrieved 28 September 2023.
- ^ Danish University Antidepressant Group. (1990). Paroxetine: a selective serotonin reuptake inhibitor showing better tolerance, but weaker antidepressant effect than clomipramine in a controlled multicenter study. Journal of Affective Disorders, 18(4), 289-299.
- S2CID 837142.
- ^ Gillman, Ken, and Last updated Mar. "Clomipramine: Potent SNRI Anti-Depressant."
- ^ Vestergaard, P., Gram, L. F., Kragh-Sørensen, P., Bech, P., Reisby, N., & Bolwig, T. G. (1993). Therapeutic potentials of recently introduced antidepressants. Danish University Antidepressant Group. Psychopharmacology series, 10, 190–198. https://doi.org/10.1007/978-3-642-78010-3_18
- ^ Montgomery SA, Baldwin DS, Blier P, Fineberg NA, Kasper S, Lader M, Lam RW, Lépine JP, Möller HJ, Nutt DJ, Rouillon F. Which antidepressants have demonstrated superior efficacy? A review of the evidence. International clinical psychopharmacology. 2007 Nov 1;22(6):323-9.
- ^ Wijkstra J, Lijmer J, Balk FJ, Geddes JR, Nolen WA. Pharmacological treatment for unipolar psychotic depression: systematic review and meta-analysis. The British Journal of Psychiatry. 2006 May;188(5):410-5.
- ^ Bayes, A. and Parker, G., 2019. How to choose an antidepressant medication. Acta Psychiatrica Scandinavica, 139(3), pp.280-291.
- ^ Anderson, I.M., 2000. Selective serotonin reuptake inhibitors versus tricyclic antidepressants: a meta-analysis of efficacy and tolerability. Journal of affective disorders, 58(1), pp.19-36.
- S2CID 16921407.
- from the original on 3 December 2013.
- PMID 9375591.
- (PDF) from the original on 3 December 2013.
- PMID 1429420.
- PMID 8735578.
- PMID 10435772.
- S2CID 22832400.
- PMID 1929772.
- ^ Mestermann S, Rudtke L, Brazdis RM, Tsaktanis T, Kornhuber J, Thürauf N. Successful Pharmacologic Treatment of Self-Bloodletting with Factitious Chronic Anemia (Lasthénie de Ferjol Syndrome) with High-Dose Serotonergic Medication: A Case Report. Behav Sci (Basel). 2024 Mar 14;14(3):237. doi: 10.3390/bs14030237. PMID: 38540540.
- (PDF) from the original on 3 December 2013.
- ^ Waldinger, M. D., Zwinderman, A. H., & Olivier, B. (2004). On-demand treatment of premature ejaculation with clomipramine and paroxetine: a randomized, double-blind fixed-dose study with stopwatch assessment. European urology, 46(4), 510–516. https://doi.org/10.1016/j.eururo.2004.05.005
- PMID 8456168.
- .
- PMID 7775365.
- PMID 10770483.
- ^ PMID 26789925.
- ^ "Trichotillomania Medication". Medscape Reference. WebMD. 10 October 2013. Archived from the original on 3 December 2013. Retrieved 1 December 2013.
- PMID 21797657.
- PMID 34582562.
- S2CID 34432532.
- PMID 6435460.
- PMID 8331098.
- S2CID 44813426.
- S2CID 25452860.
- S2CID 146533651.
- PMID 18648320.
- PMID 35342656.
- PMID 30834169.
- PMID 17388706.
- S2CID 18029965.
- PMID 7811162.
- ^ Wilson, M., & Tripp, J. (2022). Clomipramine. In StatPearls. StatPearls Publishing.
- PMID 38350669.
- PMID 16406480.
- ^ PMID 10744432.
- PMID 2405155.
- PMID 15654489.
- ^ S2CID 34960274.
- ^ Lundt L, Brownsmith C (May 2005). "Why me? One youth's quest for sanity" (PDF). Current Psychiatry. 4 (5): 85–99.
- PMID 23630412.
- PMID 35767803.
- S2CID 260239945.
- S2CID 1925913.
- PMID 11130931.
- S2CID 54136953.
- S2CID 46037419.
- S2CID 28452756.
- S2CID 12336027.
- PMID 25709352.
- S2CID 23946424.
- PMID 8784659.
- PMID 1459499.
- PMID 895917.
- PMID 9262526.
- PMID 19031375.
- PMID 20435959.
- PMID 34381507.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Archived from the original on 8 September 2017. Retrieved 14 August 2017.
- ^ PMID 9537821.
- ^ PMID 11454918.
- ^ S2CID 30166780.
- ^ PMID 3816971.
- ^ PMID 9686407.
- PMID 2533080.
- PMID 7680751.
- PMID 8397408.
- ^ PMID 6086881.
- ^ S2CID 14274150.
- PMID 8359206.
- PMID 18634780.
- ^ ISBN 978-1-107-72992-6.
- ISBN 978-0-19-939514-9. Archivedfrom the original on 8 September 2017.
- ^ Boyson S. J. (1988). Bethanechol for anticholinergic side effects. Annals of neurology, 23(4), 422–423. https://doi.org/10.1002/ana.410230429
- S2CID 42755526.
- PMID 8101048.
- ISBN 978-1-4614-3710-9. Archivedfrom the original on 8 September 2017.
- ISBN 978-3-0348-8391-7. Archivedfrom the original on 8 September 2017.
- ISBN 978-0-205-35556-3.
[...] desmethylclomipramine levels may be twice as high as those of clomipramine (Rudorfer & Potter, 1999).
- ^ S2CID 12259485.
- S2CID 14928733.
- ^ PMID 17471183.
- ISBN 978-0-340-94008-2. Archivedfrom the original on 8 September 2017.
- ^ PMID 27824534.
- PMID 25038602.
- ^ ISBN 978-0-07-176939-6.
- ISBN 978-1-136-28009-2.
- ISBN 978-0-7817-2845-4.
- ISBN 978-0-7295-8605-4.
- ^ ISBN 978-0-19-984550-7.
- ^ S2CID 30691618.
- S2CID 17470375.
- ^ ISBN 978-0-87630-589-8.
- ^ ISBN 978-0-8147-8661-1.
- ISBN 978-0-19-936258-5.
- ^ "PrANAFRANIL® Clomipramine Hydrochloride Tablets" (PDF). Health Canada drug database.
- ^ "Anafranil SR 75 mg Prolonged-release Tablets; Summary of Product Characteristics" (PDF). Health Products Regulatory Authority of Ireland. 20 December 2021. CRN00CP8H.
- ^ "Clomipramine Capsules, Hard; Package leaflet: Information for the patient" (PDF). Electronic Medicines Compendium (EMC). July 2017.
- PMID 11691690.
- ^ Ananth, J., 1986. Clomipramine: an antiobsessive drug. The Canadian Journal of Psychiatry, 31(3), pp.253-258.
- ^ ISBN 978-0-88048-707-8.
- ^ ISBN 978-1-139-49626-1.
- ^ S2CID 6582600.
- ^ PMID 10986733.
- ^ PMID 20623923.
- PMID 8780435.
- PMID 8083135.
- S2CID 7172333.
- PMID 22932229.
- S2CID 40042242.
- S2CID 73291517.
- ^ ISBN 978-1-118-89025-7.
- PMID 2180623.
- S2CID 6818721.
- ^ Amsterdam, J.D., García‐España, F. and Rosenzweig, M., 1997. Clomipramine augmentation in treatment‐resistant depression. Depression and Anxiety, 5(2), pp.84-90.
- ^ Gomez, J.M. and Perramon, C.T., 2000. Combined treatment with venlafaxine and tricyclic antidepressants in depressed patients who had partial response to clomipramine or imipramine: initial findings. J Clin Psychiatry, 61(4), pp.285-289.
- ^ DeVeaugh-Geiss, J., Landau, P. and Katz, R., 1989. Treatment of obsessive compulsive disorder with clomipramine. Psychiatric Annals, 19(2), pp.97-99.
- ^ Górska, N., Słupski, J., Cubała, W.J., Wiglusz, M.S. and Gałuszko-Węgielnik, M., 2018. Antidepressants in epilepsy. neurologia i neurochirurgia polska, 52(6), pp.657-661.
- ^ Rowland, D.L., De Gouveia Brazao, C.A. and Koos Slob, A., 2001. During the initial titration, it may be necessary to divide the total daily dose up to thrice (to reduce certain side-effects) but once the full optimal dose has been reached, most people can be sustained on a once-at-bedtime dose, or on a prn basis 3 to 5 hours before sexual intercourse in the case of premature ejaculation. Effective daily treatment with clomipramine in men with premature ejaculation when 25 mg (as required) is ineffective. BJU international, 87(4), pp.357-360.
- ^ Mohammed, M.A., CLOMIPRAMINE AS DAILY DOSE IN THE TREATMENT OF PATIENTS WITH PREMATURE.
- ^ ISBN 978-94-007-5805-6. Archivedfrom the original on 8 September 2017.
- ^ Psychopharmacology Bulletin. National Institute of Mental Health. 1984.
Clomipramine or 3-chloro-imipramine (Figure 4) is imipramine with a chlorine atom added to one of its rings.
- ISBN 978-0-471-95052-3.
- ISBN 978-3-527-64632-6. Archivedfrom the original on 8 September 2017.
- ISBN 978-1-56053-470-9.
- ISBN 978-0-19-162675-3. Archivedfrom the original on 8 September 2017.
- ^ ISBN 978-3-88763-075-1.
- ^ PMID 19557250.
- ISBN 978-1-55642-922-4.
- ISBN 978-0-440-50847-2.
- ^ "The history of obsessive-compulsive disorder". ocdhistory.net. Retrieved 4 November 2023.
- ISBN 978-0-8151-8624-3.
Until very recently, the "gold standard" of pharmacologic therapy for obsessive-compulsive disorder has been clomipramine. At this time, however, the SSRIs (selective serotonin reuptake inhibitors) have become the drugs of choice.
- ISBN 978-0-444-52002-9.
- ISBN 978-0-19-530703-0.
- ISBN 978-1-4616-3230-6.
- ISBN 978-94-011-4439-1. Archivedfrom the original on 15 February 2017.
- ^ a b c "Clomipramine". Archived from the original on 12 August 2017. Retrieved 12 August 2017.
- ^ "CLOMICALM clomipramine tablet CLOMIPRAMINE- clomipramine powder Novartis Animal Health US, Inc". Drugs@FDA. Novartis Animal Health US, Inc. December 1998. Archived from the original on 3 December 2013. Retrieved 1 December 2013.
- PMID 9631696.
- S2CID 42730627. Archived from the original(PDF) on 3 December 2013. Retrieved 30 November 2013.
- S2CID 2817066. Archived from the original(PDF) on 3 December 2013. Retrieved 30 November 2013.
- PMID 11349411.
- PMID 15485047.
- PMID 15634861.
