Trazodone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Desyrel, Trittico, others[1] |
| Other names | AF-1161 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a681038 |
| License data | |
| Dependence liability | None[2] |
| Addiction liability | None[2] |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
Tmax)[10] | |
| Elimination half-life | • Trazodone (IR): 4–15 hours[11][12][13][14] • Trazodone (ER): 9–13 hours[15][12][13] • mCPP: 3–16 hours[11][12][14][16] |
| Excretion | Urine: 70–75%[7] Feces: 21%[7] |
| Identifiers | |
| |
JSmol) | |
| Melting point | 87 °C (189 °F) |
| |
| |
| (verify) | |
Trazodone, sold under many brand names,[1] is an antidepressant medication.[20] It is used to treat major depressive disorder, anxiety disorders, and insomnia.[20] The medication is taken orally.[20]
Common side effects include
Trazodone was approved for medical use in the United States in 1981.
Medical uses
Depression
The primary use of trazodone is the treatment of unipolar major depression with or without
Because trazodone has minimal anticholinergic activity, it was especially welcomed as a treatment for geriatric patients with depression when it first became available. Three double-blind studies reported trazodone had antidepressant efficacy similar to that of other antidepressants in geriatric patients. Unfortunately, a side effect of trazodone, orthostatic hypotension, which may cause dizziness and increase the risk of falling, can have devastating consequences for elderly patients.[28] Therefore, this side effect, along with sedation, often makes trazodone less acceptable for this population compared to newer compounds that share its lack of anticholinergic activity (but not the rest of its side effect profile). Still, trazodone is often helpful for geriatric patients with depression who have severe agitation and insomnia.[27]
Trazodone is usually used at a dosage of 150 to 300 mg/day for the treatment of depression.[17][13] Lower doses have also been used to augment other antidepressants or when initiating therapy.[17][13] Higher doses, up to 600 mg/day, have been used in more severe cases of depression (in hospitalized patients, for example).[29] Trazodone is usually administered multiple times per day, but once-daily administration may be similarly effective.[30]
Insomnia
Low-dose trazodone is used off-label in the treatment of insomnia and is considered to be effective and safe for this indication.[31][13][32] It may also be used to treat antidepressant-related insomnia.[33] Trazodone was the second-most prescribed agent for insomnia in the early 2000s even though most studies of trazodone for treatment of sleep disturbances have been in depressed individuals.[13][34][35]
The benefits of trazodone for insomnia must be weighed against potential adverse effects, such as
Trazodone is used at low doses in the range of 25 to 150 mg/day for insomnia.[31][40][36][38] Higher doses of 200 to 600 mg/day have also been studied.[31][35]
The
Other disorders
Trazodone is often used in the treatment of
Combination with other antidepressants
Trazodone is often used in combination with other antidepressants such as selective serotonin reuptake inhibitors in order to augment their antidepressant and anxiolytic effects and to reduce side effects such as sexual dysfunction, anxiety, and insomnia.[43][13][42][49]
Available forms
Trazodone is provided as the hydrochloride salt and is available in the form of 50 mg, 100 mg, 150 mg, and 300 mg oral tablets.[6] In Italy, it is also available as an oral solution (Trittico 60 mg/mL) with a dosing pipette marked at 25 mg and 50 mg.[50]
An extended-release oral tablet formulation at doses of 150 mg and 300 mg is also available.[51][52]
Side effects
Because of its lack of
Precautions for trazodone include known hypersensitivity to trazodone and under 18 years and combined with other antidepressant medications, it may increase the possibility of suicidal thoughts or actions.[54]
While trazodone is not a true member of the
Suicide
Antidepressants may increase the risk of suicidal thoughts and behaviors in children and young adults. Close monitoring for emergence of suicidal thoughts and behaviors is thus recommended.[56]
Sedation
Since trazodone may impair the mental and/or physical abilities required for performance of potentially hazardous tasks, such as operating an automobile or machinery, the patient should be cautioned not to engage in such activities while impaired. Compared to the reversible MAOI antidepressant drug moclobemide, more impairment of vigilance occurs with trazodone.[57] Trazodone has been found to impair driving ability.[58]
Cardiac
Case reports have noted cardiac arrhythmias emerging in relation to trazodone treatment, both in patients with pre-existing mitral valve prolapse and in patients with negative personal and family histories of cardiac disease.[59]
Priapism
A relatively rare side effect associated with trazodone is priapism, likely due to its antagonism at α-adrenergic receptors.[62] More than 200 cases have been reported, and the manufacturer estimated that the incidence of any abnormal erectile function is about one in 6,000 male patients treated with trazodone. The risk for this side effect appears to be greatest during the first month of treatment at low dosages (i.e. <150 mg/day). Early recognition of any abnormal erectile function is important, including prolonged or inappropriate erections, and should prompt discontinuation of trazodone treatment. Spontaneous orgasms have also been reported with trazodone in men.[63]
Clinical reports have described trazodone-associated psychosexual side effects in women as well, including increased libido, priapism of the clitoris, and spontaneous orgasms.[59][64]
Others
Rare cases of liver toxicity have been observed, possibly due to the formation of reactive metabolites.[65]
Elevated prolactin concentrations have been observed in people taking trazodone.[29][66] They appear to be increased by around 1.5- to 2-fold.[29][66]
Studies on trazodone and
Trazodone does not seem to worsen periodic limb movements during sleep.[68]
Trazodone is associated with increased risk of falls in older adults.[28] It has also been associated with increased risk of hip fractures in older adults.[69]
Pregnancy and lactation
Sufficient data in humans are lacking. Use should be justified by the severity of the condition to be treated.[70][71]
Overdose
There are reported cases of high doses of trazodone precipitating serotonin syndrome.[72] There are also reports of patients taking multiple SSRIs with trazodone and precipitating serotonin syndrome.[72]
Trazodone appears to be relatively safer than
Interactions
Trazodone is metabolized by several liver enzymes, including
A study found that ritonavir, a strong CYP3A4 and CYP2D6 inhibitor and moderate CYP1A2 inducer, increased trazodone peak levels by 1.34-fold, increased
Combination of trazodone with selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants (TCAs), or monoamine oxidase inhibitors has a theoretical risk of serotonin syndrome.[17][13] However, trazodone has been studied in combination with SSRIs and seemed to be safe in this context.[17][13] On the other hand, cases of excessive sedation and serotonin syndrome have been reported with the combinations of trazodone and fluoxetine or paroxetine.[11] This may be due to combined potentiation of the serotonin system.[11] However, it may also be related to the fact that fluoxetine and paroxetine are strong inhibitors of CYP2D6 and fluoxetine is additionally a weak or moderate inhibitor of CYP3A4.[11][80] Accordingly, fluoxetine has been reported to result in increased levels of trazodone and mCPP by 1.31- to 1.65-fold and by 2.97- to 3.39-fold, respectively.[11][81]
Smokers have lower levels of trazodone and higher ratios of mCPP to trazodone.[11][82] Trazodone levels were 30% lower in smokers and mCPP to trazodone ratio was 1.29-fold higher in smokers, whereas mCPP concentrations were not different between smokers and non-smokers.[82] Smoking is known to induce CYP1A2, and this may be involved in these findings.[11]
Pharmacology
Pharmacodynamics
| Site | Trazodone | mCPP | Species | Ref |
|---|---|---|---|---|
| SERT | 160–>10,000[84] | 202–432 | Human | [83][85][86] |
| NET | ≥8,500 | ≥1,940 | Human | [86][85] |
| DAT | ≥7,400 | ND | Human | [86][83] |
| 5-HT1A | 96–118 | 44–400 | Human | [83][87][88] |
| 5-HT1B | >10,000 | 89–501 | Human | [83][89] |
| 5-HT1D | 106 | 210–1,300 | Human | [83][88][90] |
| 5-HT1E | >10,000 | ND | Human | [83] |
| 5-HT1F | ND | ND | ND | ND |
| 5-HT2A | 20–45 | 32–398 | Human | [83][91][92][93] |
| 5-HT2B | 74–189 | 3.2–63 | Human | [83][91][94][95] |
| 5-HT2C | 224–402 | 3.4–251 | Human | [91][96][97][93] |
| 5-HT3 | >10,000 | 427 | Human | [83] |
| 5-HT4 | ND | ND | ND | ND |
| 5-HT5A | >10,000 | 1,354 | Human | [83] |
| 5-HT6 | >10,000 | 1,748 | Human | [83] |
| 5-HT7 | 1,782 | 163 | Human | [83] |
| α1 | 12–42 | 97–2,900 | Human | [85][87][88][98] |
| α1A | 153 | 1,386 | Human | [83] |
| α1B | ND | 915 | Human | [83] |
| α1D | ND | ND | ND | ND |
| α2 | 106–490 | 112–570 | Human | [87][85][88][98] |
| α2A | 728 | 145 | Human | [83] |
| α2B | ND | 106 | Human | [83] |
| α2C | 155 | 124 | Human | [83] |
β |
>10,000 | 2,500 | Human | [83][88] |
| β1 | >10,000 | 2,359 | Human | [83] |
| β2 | >10,000 | 3,474 | Human | [83] |
D1 |
3,730 | 7,000 | Human | [83][88] |
D2 |
≥3,500 | >10,000 | Human | [83][87][99][88] |
D3 |
353 | >10,000 | Rat | [83][88] |
D4 |
703 | ND | Human | [83] |
D5 |
>10,000 | >10,000 | Human | [83][88] |
| H1 | 220–1,100 | 326 | Human | [83][98][87] |
| H2 | 3,290 | ND | Human | [83] |
| H3 | >10,000 | ND | Guinea pig | [83] |
| H4 | >10,000 | ND | Human | [83] |
mAChRs |
>10,000 | >10,000 | Human | [83][99][87][88] |
nAChRs |
>10,000 | >10,000 | Human | [83] |
| σ1 | >10,000 | ND | Rat | [83] |
| σ2 | 536 | 8,350 | Rat | [83] |
| I1 | ND | 759 | Rat | [83] |
| NMDAR (MK-801) |
>10,000 | ND | Rat | [83] |
VDCCs |
>10,000 | 6,043 | Rat | [83] |
| Values are Ki (nM) . The smaller the value, the more strongly the drug binds to the site.
| ||||
Trazodone is a mixed
Trazodone has a minor
Trazodone's 5-HT2A receptor antagonism and weak serotonin reuptake inhibition form the basis of its common label as an antidepressant of the serotonin antagonist and reuptake inhibitor (SARI) type.[42]
Target occupancy studies
Studies have estimated occupancy of target sites by trazodone based on trazodone concentrations in blood and brain and on the affinities of trazodone for the human targets in question.[107][49][12] Roughly half of brain 5-HT2A receptors are blocked by 1 mg of trazodone and essentially all 5-HT2A receptors are saturated at 10 mg of trazodone, but the clinically effective hypnotic doses of trazodone are in the 25–100 mg range.[31][40] The occupancy of the serotonin transporter (SERT) by trazodone is estimated to be 86% at 100 mg/day and 90% at 150 mg/day.[17][107] Trazodone may almost completely occupy the 5-HT2A and 5-HT2C receptors at doses of 100 to 150 mg/day.[17][107] Significant occupancy of a number of other sites may also occur.[17][107] However, another study estimated much lower occupancy of the SERT and 5-HT2A receptors by trazodone.[12]
| Target | Estimated target occupancy | ||
|---|---|---|---|
| 50 mg/day | 100 mg/day | 150 mg/day | |
| SERT | 75% | 86% | 90% |
| 5-HT1A | 91% | 95% | 97% |
| 5-HT1D | 91% | 95% | 97% |
| 5-HT2A | 97% | 98% | 99% |
| 5-HT2B | 94% | 97% | 98% |
| 5-HT2C | 83% | 91% | 94% |
| 5-HT7 | 39% | 56% | 66% |
| α1A | 88% | 94% | 96% |
| α2A | 61% | 75% | 82% |
| α2C | 88% | 94% | 96% |
| D4 | 62% | 76% | 83% |
| H1 | 84% | 91% | 94% |
| Very low (<25–33%): NET, DAT, 5-HT1B, 5-HT1E, 5-HT3, 5-HT5A, 5-HT6, β1, β2, D5, H4, mAChRs, nAChRs. Low (<50%): D1, D2. Not determined: α1B, α2B, D3. Note: Another study estimated much lower occupancies.[12] | |||
Correspondence to clinical effects
This section needs to be updated. The reason given is: Needs to be updated in light of new occupancy studies.. (October 2020) |
Trazodone may act predominantly as a 5-HT2A receptor antagonist to mediate its therapeutic benefits against
The combined actions of 5-HT2A and 5HT2C receptor antagonism with serotonin reuptake inhibition only occur at moderate to high doses of trazodone.[112] Doses of trazodone lower than those effective for antidepressant action are frequently used for the effective treatment of insomnia.[112] Low doses exploit trazodone's potent actions as a 5-HT2A receptor antagonist, and its properties as an antagonist of H1 and α1-adrenergic receptors, but do not adequately exploit its SERT or 5-HT2C inhibition properties, which are weaker.[112] Since insomnia is one of the most frequent residual symptoms of depression after treatment with an SSRI, a hypnotic is often necessary for patients with a major depressive episode.[112] Not only can a hypnotic potentially relieve the insomnia itself, but treating insomnia in patients with major depression may also increase remission rates due to improvement of other symptoms such as loss of energy and depressed mood.[112] Thus, the ability of low doses of trazodone to improve sleep in depressed patients may be an important mechanism whereby trazodone can augment the efficacy of other antidepressants.[112]
Trazodone's potent α1-adrenergic blockade may cause some side effects like orthostatic hypotension and sedation.[113] Conversely, along with 5-HT2A and H1 receptor antagonism, it may contribute to its efficacy as a hypnotic. Trazodone lacks any affinity for the muscarinic acetylcholine receptors, so does not produce anticholinergic side effects.
mCPP, a non-selective
Pharmacokinetics
Absorption
Trazodone is well-
Distribution
Trazodone is not sequestered into any
Metabolism
The
mCPP is formed from trazodone by CYP3A4 and is metabolized via hydroxylation by CYP2D6 (to a para-hydroxylated metabolite).
Elimination
The elimination of trazodone is biphasic: the first phase's half-life (distribution) is 3 to 6 hours, and the following phase's half-life (elimination) is 4.1 to 14.6 hours.[11][12][13][14] The elimination half-life of extended-release trazodone is 9.1 to 13.2 hours.[15][12][13] The elimination half-life of mCPP is 2.6 to 16.0 hours and is longer than that of trazodone.[11][12][14] Metabolites are conjugated to gluconic acid or glutathione and around 70 to 75% of 14C-labelled trazodone was found to be excreted in the urine within 72 hours.[124] The remaining drug and its metabolites are excreted in the faeces via biliary elimination. Less than 1% of the drug is excreted in its unchanged form.[119] After an oral dose of trazodone, it was found to be excreted 20% in the urine as TPA and conjugates, 9% as the dihydrodiol metabolite, and less than 1% as unconjugated mCPP.[11] mCPP is glucuronidated and sulfated similarly to other trazodone metabolites.[11]
Chemistry
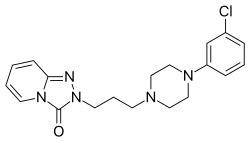
Trazodone is a
History
Trazodone was developed in
Society and culture
Generic names
Trazodone is the
Brand names
Trazodone has been marketed under a large number of brand names throughout the world.[133][135] Major brand names include Desyrel (worldwide), Donaren (Brazil), Molipaxin (Ireland, United Kingdom), Oleptro (United States), Trazorel (Canada), and Trittico (worldwide).[133][135]
Research
Trazodone may be effective in the treatment of
Trazodone may be useful in the treatment of certain symptoms like
Trazodone has been studied as an
Trazodone may be useful in the treatment of certain
Trazodone may be useful in promoting motor recovery after stroke.[42][168]
Veterinary use
Trazodone has been used to reduce anxiety and stress, to improve sleep, and to produce sedation in dogs and cats in veterinary medicine.[169][170][171]
See also
References
- ^ a b "Trazodone". Drugs.com. Retrieved 9 February 2019.
- ^ ISBN 978-0-8247-4497-7.
- FDA. Retrieved 22 October 2023.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ a b "Desyrel (trazodone HCl)". DailyMed. 19 March 2013. Retrieved 4 April 2022.
- ^ a b "Trazodone hydrochloride tablet". DailyMed. 29 June 2021. Retrieved 3 April 2022.
- ^ a b c Truven Health Analytics, Inc. DrugPoint System (Internet) [cited 2013 Oct 1]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- ^ "Trazodone". DrugBank. Retrieved 7 June 2015.
- ISBN 978-3-642-18500-7.
- ^ "MicroMedex DrugPoints – Trazodone". Pharmacy Choice. Retrieved 20 April 2017.
- ^ S2CID 19585113.
- ^ S2CID 6068710.
- ^ PMID 23192413.
- ^ S2CID 36104356.
- ^ S2CID 26833385.
- ISBN 978-1-58562-523-9.
- ^ PMID 31379379.
- ^ S2CID 45097940.
- ISBN 978-1-60913-345-0.
- ^ a b c d e f g h "Trazodone". The American Society of Health-System Pharmacists. 17 March 2021. Retrieved 24 December 2021.
- ^ "Trazodone Use During Pregnancy". Drugs.com. Retrieved 7 January 2018.
- ISBN 978-0-521-85702-4.
- ISBN 978-0-7817-6879-5.
- ISBN 978-0-85711-156-2.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Trazodone – Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ ISBN 978-1-58562-309-9.
- ^ PMID 25735839.
- ^ S2CID 265772823.
- PMID 2211561.
- ^ PMID 29552421.
- ^ PMID 26088119.
- PMID 8010365.
- PMID 15816789.
- ^ PMID 16517453.
- ^ PMID 29680424.
- PMID 32714220.
- ^ PMID 29761479.
- ^ S2CID 243329243.
- ^ S2CID 2977125.
- PMID 27998379.
- ^ S2CID 205452630.
- ^ S2CID 10638259.
- S2CID 30562398.
- ISBN 978-0-85711-084-8.
- PMID 12804478.
- S2CID 38329753.
- S2CID 45471039.
- ^ S2CID 53042045.
- ^ https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_000219_022323_RCP.pdf&sys=m0b1l3
- PMID 21431085.
- ^ "Trazodone (Oral Route) Proper Use Clinic". www.mayoclinic.org – Mayo Clinic. Retrieved 11 February 2020.
- PMID 26459471.
- ^ "Webmd.com". Webmd.com. Retrieved 14 March 2014.
- PMID 16913164.
- ^ "FDA – Trazodone Prescribing Information" (PDF).
- PMID 2677245.
- S2CID 3425898.
- ^ ISBN 978-1-58562-309-9.
- ^ "Trazodone Product Monograph" (PDF). 2015.
- ^ "Highlights of Prescribing Information" (PDF). U.S. Food and Drug Administration. 2017.
- PMID 3573170.
- S2CID 39522632.
- PMID 19674253.
- PMID 15978881.
- ^ S2CID 41490922.
- PMID 34097124.
- PMID 28822709.
- S2CID 22681213.
- PMID 12655908.
- PMID 3768252.
- ^ a b Cushing TA (24 April 2018). "Selective Serotonin Reuptake Inhibitor Toxicity". Medscape. WebMD LLC. Retrieved 22 December 2018.
- PMID 15975258.
- S2CID 21487479.
- PMID 3321131.
- ^ PMID 9616194.
- S2CID 1979283.
- ^ S2CID 11521715.
- ^ PMID 29061081.
- PMID 24569517.
- ^ a b c
Maes M, Westenberg H, Vandoolaeghe E, Demedts P, Wauters A, Neels H, Meltzer HY (October 1997). "Effects of trazodone and fluoxetine in the treatment of major depression: therapeutic pharmacokinetic and pharmacodynamic interactions through formation of meta-chlorophenylpiperazine". Journal of Clinical Psychopharmacology. 17 (5): 358–64. PMID 9315986.
- ^ PMID 8675966.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak al am Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ Roth, BL; Driscol, J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 25 May 2018.
- ^ PMID 9400006.
- ^ PMID 9537821.
- ^ S2CID 21236268.
- ^ S2CID 46730665.
- S2CID 35553281.
- PMID 1652050.
- ^ S2CID 8938111.
- PMID 7582481.
- ^ PMID 19505264.
- PMID 11104741.
- PMID 10498829.
- PMID 15081042.
- S2CID 24930608.
- ^ PMID 6086881.
- ^ PMID 8100134.
- ^ S2CID 24965789.
- ^ S2CID 27389008.
- PMID 23357028.
- ISBN 978-0-7817-6879-5.
- S2CID 13007057.
- ^ PMID 7255270.
- ^ S2CID 44968564.
- ^ S2CID 28804036.
- ^ S2CID 25140746.
- PMID 12096167.
- S2CID 7459081.
- PMID 10065919.
- ^ ISBN 978-1-107-68646-5.
- S2CID 43202340.
- S2CID 25709000.
- PMID 2311675.
- S2CID 38913696.
- S2CID 901077.
- PMID 1575270.
- ^ a b "Trazodone". www.drugbank.ca. Retrieved 31 January 2019.
- PMID 11393588.)
{{cite journal}}: CS1 maint: DOI inactive as of April 2024 (link - S2CID 21305772.
- ISBN 978-3-527-62147-7.
- PMID 21073812.
- PMID 1037253.
- ISBN 978-3-527-66445-0.
- ISBN 978-3-527-64564-0.
- ISBN 978-0-08-086111-1.
- ^ Wegener G (30 March 2016). "Ban & Silvestrini's Trazodone". International Network for the History of Neuropsychopharmacology. Archived from the original on 20 March 2017. Retrieved 4 June 2017.
- S2CID 34798378.
- ^ "Trazodone: Common sleep drug is little-known antidepressant". Consumer Reports. August 2015.
- ISBN 978-1-4612-4828-6.
- ISBN 978-1-4757-2085-3.
- ^ ISBN 978-3-88763-075-1.
- ISBN 978-94-011-4439-1.
- ^ a b c "Trazodone". Drugs.com.
- PMID 33967614.
- S2CID 7499389.
- ^ a b "Bupropion/Trazodone - AdisInsight".
- S2CID 52813769.
- ^ S2CID 207477620.
- PMID 21715413.
- S2CID 207939482.
- PMID 25719754.
- PMID 24899767.
- PMID 15495135.
- PMID 21328305.
- PMID 33189083.
- S2CID 13491951.
- S2CID 228158773.
- S2CID 212417082.
- S2CID 8766330.
- ^ PMID 20807960.
- PMID 25991654.
- PMID 14583971.
- S2CID 219586994.
- S2CID 209411316.
- PMID 14583952.
- S2CID 37252070.
- PMID 23358935.
- S2CID 9178257.
- S2CID 247006915.
- S2CID 232217908.
- PMID 28247977.
- PMID 15948431.
- PMID 30564156.
- S2CID 21793648.
- S2CID 293910.
- PMID 20032921.
- PMID 34475580.
- S2CID 76667125.
- PMID 29415813.
