O-linked glycosylation
O-linked glycosylation is the attachment of a
Common types of O-glycosylation
O-N-acetylgalactosamine (O-GalNAc)
Addition of

Biosynthesis
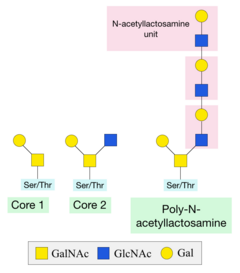
GalNAc is added onto a serine or threonine residue from a precursor molecule, through the activity of a GalNAc transferase enzyme.[1] This precursor is necessary so that the sugar can be transported to where it will be added to the protein. The specific residue onto which GalNAc will be attached is not defined, because there are numerous enzymes that can add the sugar and each one will favour different residues.[7] However, there are often proline (Pro) residues near the threonine or serine.[6]
Once this initial sugar has been added, other glycosyltransferases can catalyse the addition of additional sugars. Two of the most common structures formed are Core 1 and Core 2. Core 1 is formed by the addition of a galactose sugar onto the initial GalNAc. Core 2 consists of a Core 1 structure with an additional N-acetylglucosamine (GlcNAc) sugar.[6] A poly-N-acetyllactosamine structure can be formed by the alternating addition of GlcNAc and galactose sugars onto the GalNAc sugar.[6]
Terminal sugars on O-glycans are important in recognition by

Functions
O-GalNAc sugars are important in a variety of processes, including
O-GalNAc sugars are common on membrane
In order for leukocytes of the immune system to move into infected cells, they have to interact with these cells through
Mucins are a group of heavily O-glycosylated proteins that line the gastrointestinal and respiratory tracts to protect these regions from infection.[6] Mucins are negatively charged, which allows them to interact with water and prevent it from evaporating. This is important in their protective function as it lubricates the tracts so bacteria cannot bind and infect the body. Changes in mucins are important in numerous diseases, including cancer and inflammatory bowel disease. Absence of O-glycans on mucin proteins changes their 3D shape dramatically and often prevents correct function.[1][9]
O-N-acetylglucosamine (O-GlcNAc)
Addition of

O-GlcNAcylation differs from other O-glycosylation processes because there are usually no sugars added onto the core structure and because the sugar can be attached or removed from a protein several times.
Because O-GlcNAc can be added and removed, it is known as a dynamic modification and has a lot of similarities to
Additionally, O-GlcNAcylation can enhance the Warburg Effect, which is defined as the change that occurs in the metabolism of cancer cells to favour their growth.[6][14] Because both O-GlcNAcylation and phosphorylation can affect specific residues and therefore both have important functions in regulating signalling pathways, both of these processes provide interesting targets for cancer therapy.
O-Mannose (O-Man)

O-mannosylation involves the transfer of a mannose from a dolichol-P-mannose donor molecule onto the serine or threonine residue of a protein.[15] Most other O-glycosylation processes use a sugar nucleotide as a donor molecule.[7] A further difference from other O-glycosylations is that the process is initiated in the endoplasmic reticulum of the cell, rather than the Golgi apparatus.[1] However, further addition of sugars occurs in the Golgi.[15]
Until recently, it was believed that the process is restricted to
O-Galactose (O-Gal)
O-galactose is commonly found on lysine residues in collagen, which often have a hydroxyl group added to form hydroxylysine. Because of this addition of an oxygen, hydroxylysine can then be modified by O-glycosylation. Addition of a galactose to the hydroxyl group is initiated in the endoplasmic reticulum, but occurs predominantly in the Golgi apparatus and only on hydroxylysine residues in a specific sequence.[1][18]
While this O-galactosylation is necessary for correct function in all collagens, it is especially common in collagen types IV and V.[19] In some cases, a glucose sugar can be added to the core galactose.[7]
O-Fucose (O-Fuc)
Addition of fucose sugars to serine and threonine residues is an unusual form of O-glycosylation that occurs in the endoplasmic reticulum and is catalysed by two fucosyltransferases.[20] These were discovered in Plasmodium falciparum[21] and Toxoplasma gondii.[22]
Several different enzymes catalyse the elongation of the core fucose, meaning that different sugars can be added to the initial fucose on the protein.[20] Along with O-glucosylation, O-fucosylation is mainly found on epidermal growth factor (EGF) domains found in proteins.[7] O-fucosylation on EGF domains occurs between the second and third conserved cysteine residues in the protein sequence.[1] Once the core O-fucose has been added, it is often elongated by addition of GlcNAc, galactose and sialic acid.
Notch is an important protein in development, with several EGF domains that are O-fucosylated.[23] Changes in the elaboration of the core fucose determine what interactions the protein can form, and therefore which genes will be transcribed during development. O-fucosylation might also play a role in protein breakdown in the liver.[1]
O-Glucose (O-Glc)
Similarly to O-fucosylation, O-glucosylation is an unusual O-linked modification as it occurs in the endoplasmic reticulum, catalysed by O-glucosyltransferases, and also requires a defined sequence in order to be added to the protein. O-glucose is often attached to serine residues between the first and second conserved cysteine residues of EGF domains, for example in
Proteoglycans

Different types of proteoglycans exist, depending on the sugar that is linked to the oxygen atom of the residue in the protein. For example, the GAG
Lipids

Galactose or glucose sugars can be attached to a hydroxyl group of
Because both galactose and glucose sugars can be added to the ceramide lipid, we have two groups of glycosphingolipids. Galactosphingolipids are generally very simple in structure and the core galactose is not usually modified. Glucosphingolipids, however, are often modified and can become a lot more complex.
Biosynthesis of galacto- and glucosphingolipids occurs differently.[6] Glucose is added onto ceramide from its precursor in the endoplasmic reticulum, before further modifications occur in the Golgi apparatus.[8] Galactose, on the other hand, is added to ceramide already in the Golgi apparatus, where the galactosphingolipid formed is often sulfated by addition of sulfate groups.[6]
Glycogenin
One of the first and only examples of O-glycosylation on tyrosine, rather than on serine or threonine residues, is the addition of glucose to a tyrosine residue in glycogenin.[7] Glycogenin is a glycosyltransferase that initiates the conversion of glucose to glycogen, present in muscle and liver cells.[26]
Clinical significance
All forms of O-glycosylation are abundant throughout the body and play important roles in many cellular functions.
Lewis epitopes are important in determining
Hinge regions of
Changes in O-glycosylation are extremely common in cancer. O-glycan structures, and especially the terminal Lewis epitopes, are important in allowing tumor cells to invade new tissues during metastasis.[6] Understanding these changes in O-glycosylation of cancer cells can lead to new diagnostic approaches and therapeutic opportunities.[1]
See also
References
- ^ PMID 9673446.
- ^ S2CID 31369853.
- S2CID 25666819.
- PMID 19251655.
- PMID 22685409.
- ^ ISBN 9781621821328.
- ^ PMID 12042244.
- ^ ISBN 978-0-19-956911-3.
- ^ Varki A (1999). Essentials of Glycobiology. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press.
- PMID 28488703.
- PMID 24311690.
- ^ PMID 21391816.
- PMID 23992419.
- PMID 24918087.
- ^ PMID 19429925.
- PMID 9878797.
- PMID 22223806.
- PMID 1220686.
- PMID 21768090.
- ^ PMID 9228088.
- PMID 28916755.
- PMID 30514763.
- PMID 21924891.
- PMID 22872643.
- ^ PMID 29495527.
- ISBN 978-0-12-383864-3.
External links
- GlycoEP: In silico Platform for Prediction of N-, O- and C-Glycosites in Eukaryotic Protein Sequences
