Plasminogen activator inhibitor-1
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 7: 101.13 – 101.14 Mb | Chr 5: 137.09 – 137.1 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
Plasminogen activator inhibitor-1 (PAI-1) also known as endothelial plasminogen activator inhibitor (serpin E1) is a protein that in humans is encoded by the SERPINE1 gene. Elevated PAI-1 is a risk factor for thrombosis and atherosclerosis.[5]
PAI-1 is a serine protease inhibitor (
The other PAI, plasminogen activator inhibitor-2 (PAI-2) is secreted by the placenta and only present in significant amounts during pregnancy. In addition, protease nexin acts as an inhibitor of tPA and urokinase. PAI-1, however, is the main inhibitor of the plasminogen activators.
Genetics
The PAI-1 gene is SERPINE1, located on chromosome 7 (7q21.3-q22). There is a common polymorphism known as 4G/5G in the promoter region. The 5G allele is slightly less transcriptionally active than the 4G.
Function
PAI-1's main function entails the inhibition of
PAI-1 is mainly produced by the endothelium (cells lining blood vessels), but is also secreted by other tissue types, such as adipose tissue.
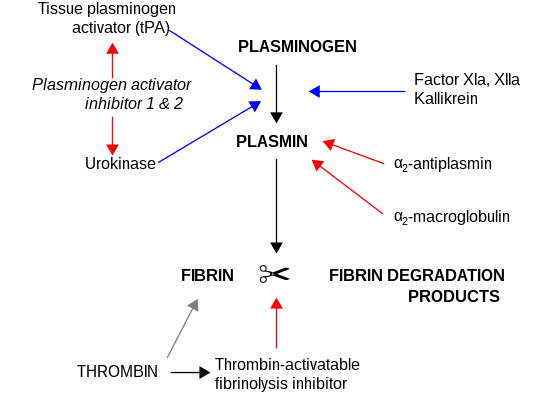
Role in disease
Congenital deficiency of PAI-1 has been reported; as fibrinolysis is not suppressed adequately, it leads to a
PAI-1 is present in increased levels in various disease states (such as a number of forms of cancer), as well as in obesity and the metabolic syndrome. It has been linked to the increased occurrence of thrombosis in patients with these conditions.
PAI-1 can induce cellular senescence.[7] PAI-1 can also be a component of the senescence-associated secretory phenotype (SASP).[8]
In inflammatory conditions in which fibrin is deposited in tissues, PAI-1 appears to play a significant role in the progression to fibrosis (pathological formation of connective tissue). Presumably, lower PAI levels would lead to less suppression of fibrinolysis and conversely a more rapid degradation of the fibrin.
Angiotensin II increases the synthesis of plasminogen activator inhibitor-1, so it accelerates the development of atherosclerosis.
Pharmacology
- arterial hypertension and activation of the renin–angiotensin system.[9]
- Annonacinone is a naturally occurring PAI-1 inhibitor found in plants of the Annonaceae family.[10]
- TM5441 is another small molecule PAI-1 inhibitor used in research.[11]
Interactions
Plasminogen activator inhibitor-1 has been shown to
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000106366 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000037411 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- S2CID 6651339.
- PMID 19672469.
- S2CID 237524296.
- PMID 29260442.
- PMID 15214776.
- PMID 27876785.
- PMID 24092817.
- PMID 11418606.
Further reading
- Mimuro J (May 1991). "[Type 1 plasminogen activator inhibitor: its role in biological reactions]". [Rinsho Ketsueki] the Japanese Journal of Clinical Hematology. 32 (5): 487–489. PMID 1870265.
- Binder BR, Christ G, Gruber F, Grubic N, Hufnagl P, Krebs M, et al. (April 2002). "Plasminogen activator inhibitor 1: physiological and pathophysiological roles". News in Physiological Sciences. 17 (2): 56–61. S2CID 21356023.
- Eddy AA (August 2002). "Plasminogen activator inhibitor-1 and the kidney". American Journal of Physiology. Renal Physiology. 283 (2): F209–F220. PMID 12110504.
- Wang J, Li J, Liu Q (August 2005). "Association between platelet activation and fibrinolysis in acute stroke patients". Neuroscience Letters. 384 (3): 305–309. S2CID 22979258.
- Schroeck F, Arroyo de Prada N, Sperl S, Schmitt M, Viktor M (2003). "Interaction of plasminogen activator inhibitor type-1 (PAI-1) with vitronectin (Vn): mapping the binding sites on PAI-1 and Vn". Biological Chemistry. 383 (7–8): 1143–1149. S2CID 37813055.
- Gils A, Declerck PJ (March 2004). "The structural basis for the pathophysiological relevance of PAI-I in cardiovascular diseases and the development of potential PAI-I inhibitors". Thrombosis and Haemostasis. 91 (3): 425–437. S2CID 3898268.
- Durand MK, Bødker JS, Christensen A, Dupont DM, Hansen M, Jensen JK, et al. (March 2004). "Plasminogen activator inhibitor-I and tumour growth, invasion, and metastasis". Thrombosis and Haemostasis. 91 (3): 438–449. S2CID 3898546.
- Harbeck N, Kates RE, Gauger K, Willems A, Kiechle M, Magdolen V, Schmitt M (March 2004). "Urokinase-type plasminogen activator (uPA) and its inhibitor PAI-I: novel tumor-derived factors with a high prognostic and predictive impact in breast cancer". Thrombosis and Haemostasis. 91 (3): 450–456. S2CID 19904733.
- Hertig A, Rondeau E (January 2004). "Plasminogen activator inhibitor type 1: the two faces of the same coin". Current Opinion in Nephrology and Hypertension. 13 (1): 39–44. S2CID 30785986.
- Hoekstra T, Geleijnse JM, Schouten EG, Kluft C (May 2004). "Plasminogen activator inhibitor-type 1: its plasma determinants and relation with cardiovascular risk". Thrombosis and Haemostasis. 91 (5): 861–872. S2CID 21576955.
- Lijnen HR (January 2005). "Pleiotropic functions of plasminogen activator inhibitor-1". Journal of Thrombosis and Haemostasis. 3 (1): 35–45. S2CID 37085650.
- De Taeye B, Smith LH, Vaughan DE (April 2005). "Plasminogen activator inhibitor-1: a common denominator in obesity, diabetes and cardiovascular disease". Current Opinion in Pharmacology. 5 (2): 149–154. PMID 15780823.
- Dellas C, Loskutoff DJ (April 2005). "Historical analysis of PAI-1 from its discovery to its potential role in cell motility and disease". Thrombosis and Haemostasis. 93 (4): 631–640. S2CID 8937106.
- Könsgen D, Mustea A, Lichtenegger W, Sehouli J (June 2005). "[Role of PAI-1 in gynaecological malignancies]". Zentralblatt für Gynakologie. 127 (3): 125–131. S2CID 260353538.
- Hermans PW, Hazelzet JA (November 2005). "Plasminogen activator inhibitor type 1 gene polymorphism and sepsis". Clinical Infectious Diseases. 41 (Suppl 7): S453–S458. PMID 16237647.
- Alessi MC, Poggi M, Juhan-Vague I (June 2007). "Plasminogen activator inhibitor-1, adipose tissue and insulin resistance". Current Opinion in Lipidology. 18 (3): 240–245. S2CID 27667588.
External links
- The MEROPS online database for peptidases and their inhibitors: I04.020 Archived 2020-05-31 at the Wayback Machine
- Plasminogen+Activator+Inhibitor+1 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Overview of all the structural information available in the PDB for UniProt: P05121 (Plasminogen activator inhibitor 1) at the PDBe-KB.