Factor XIII
Chr. 6 p24.2-p23 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Chr. 1 q31-q32.1 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Factor XIII or fibrin stabilizing factor is a
Human XIII is a
Function
Within blood,
Both of the non-
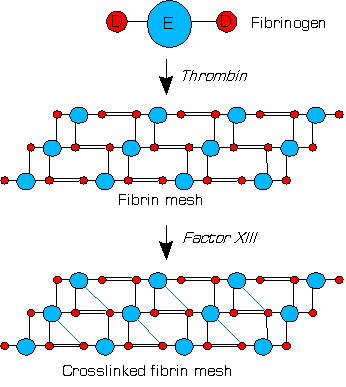
Factor XIIIa (dimer of two active A units) crosslinks fibrins within the clot by forming isopeptide bonds between various glutamines and lysines of the fibrins. These bonds make the clot physically more durable and protect it from premature enzymatic degradation (fibrinolysis).[1]
In humans,
-
Activation peptides (pink) of A units are removed by thrombin (IIa) in the presence of fibrin. B units (gray) are released with the help of calcium and A unit dimer is activated (XIIIa forms).
-
XIIIa crosslinks fibrin (simplified picture)
Genetics
Human factor XIII consist of A and B subunits. A subunit
- 1 code 5' UTR
- 2 code activation peptide
- 2–4 code β-sandwich
- 4–12 code catalytic domain
- 12–13 code β-barrel1
- 13–15 code β-barrel 2
B subunit gene is
Structure
Factor XIII of human blood is a
A units have a mass of about 83
- activation peptide (1–37)
- β-sandwich(38–184)
- 279 partake in catalysis
- β-barrel1 (516–628)
- β-barrel 2 (629–731)
B units are
Physiology
A subunits of human factor XIII are made primarily by platelets and other cells of bone marrow origin. B subunits are secreted into blood by hepatocytes. A and B units combine within blood to form heterotetramers of two A units and two B units. Blood plasma concentration of the heterotetramers is 14–48 mg/L and half-life is 9–14 days.[1]
A clot that has not been stabilized by FXIIIa is soluble in 5 mol/L urea, while a stabilized clot is resistant to this phenomenon.[2]
Factor XIII deficiency
Diagnostic use
Factor XIII levels are not measured routinely, but may be considered in patients with an unexplained bleeding tendency. As the enzyme is quite specific for monocytes and macrophages, determination of the presence of factor XIII may be used to identify and classify malignant diseases involving these cells.[4]
Discovery
Factor XIII Deficiency is also known as Laki–Lorand factor, after Kalman Laki and Laszlo Lorand, the scientists who first proposed its existence in 1948.[2] A 2005 conference recommended standardization of nomenclature.[4]
See also
References
- ^ S2CID 24703788.
- ^ PMID 17842715.
- ^ Dorgalaleh A, Naderi M, Hosseini MS, Alizadeh S, Hosseini S, Tabibian S, et al. (2015). "Factor XIII Deficiency in Iran: A Comprehensive Review of the Literature. Seminars in thrombosis and hemostasis". 41 (3): 323–29.
{{cite journal}}: Cite journal requires|journal=(help) - ^ S2CID 20424049.