Ramoplanin
 Structure of Ramoplanin A2, with the variable acyl sidechain in blue. | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
JSmol) | |
| |
| |
| | |
Ramoplanin (
Mechanism
It exerts its bacteriocidal effect by inhibiting cell wall biosynthesis, acting by inhibiting the transglycosylation step of peptidoglycan synthesis.[3] Ramoplanin specifically binds to and sequesters lipid intermediates I and II, preventing intracellular glycosyltransferase (MurG) and other steps of the peptidoglycan assembly system.[4]
Uses
Its development has been fast-tracked by the
Ramoplanin is "particularly useful" in cases E. faecalis no matter its sensitivity to vancomycin.[2]
Chemistry
Ramoplanin is a mixture of three related compounds that vary in the acyl group on the Asn N-terminus, with the most abundant form (shown in the infobox) being A2.[4]: Fig. 1
Synthesis
Biosynthesis
The biosynthesis is performed by a 33-gene cluster containing
Total synthesis
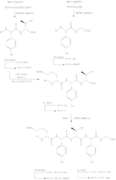
The general synthesis of Ramoplanin A1, A2 and A3 aglycons entails the preparation of residues 3-9 (heptapeptide 15), pentadepsipeptide 26 (residues 1, 2 and 15–17) along with pentapeptide 34 (residues 10–14), subsequent coupling, and cyclization to create the 49-membered aglycon core of the compound.[10] The synthesis of Ramoplanin A2 aglycon and A3 aglycon are very similar to scheme 6, where ramoplanin A1 aglycon requires the corresponding acyl group and DMF, while ramoplanin A3 aglycon synthesis requires both Bu
4NF, i-PrOH, and then treatment with the acyl group and DMF.[4]
- Ramoplanin A2 Aglycon Synthesis
-
1 - Synthesis of Hpg3-Phe9 Subunit
-
2 - Fmoc-L-HAsn(Trt)-Obn Preparation
-
3 - Synthesis of Depsipeptide subunit
-
4 - Synthesis of Orn10-Gly14 Subunit
-
5 - Preparation of acyl group
-
6 - Deprotection