Sterol 14-demethylase
| sterol 14-demethylase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
In
enzymology, a sterol 14-demethylase (EC 1.14.13.70) is an enzyme of the cytochrome P450 (CYP) superfamily. It is any member of the CYP51 family. It catalyzes a chemical reaction
such as:
- obtusifoliol + 3 O2 + 3 NADPH + 3 H+ 4alpha-methyl-5alpha-ergosta-8,14,24(28)-trien-3beta-ol + formate + 3 NADP+ + 4 H2O
The 4
substrates here are obtusifoliol, O2, NADPH, and H+, whereas its 4 products are 4alpha-methyl-5alpha-ergosta-8,14,24(28)-trien-3beta-ol, formate, NADP+, and H2O
.
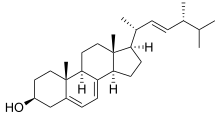
Although the
antifungal medications have been developed to inhibit 14α-demethylase activity and prevent the production of this key compound.[3]
Nomenclature
This enzyme belongs to the family of
biosynthesis of steroids.[2]
These are not the typical CYP subfamilies, but only one subfamily is created for each major taxonomic group. CYP51A for Animals, CYP51B for Bacteria. CYP51C for
monocots
only so far) have individual sequence numbers.
| CYP subfamily | etymology | kingdom |
|---|---|---|
| CYP51A | Animals | Metazoa
|
| CYP51B | Bacteria | Bacteria |
| CYP51C | Chromista | Chromista |
| CYP51D | Dictyostelium | Amoebozoa |
| CYP51E | Euglenozoa | Excavata |
| CYP51F | Fungi | Fungus |
| CYP51G | Green plants | Archaeplastida |
| CYP51H | monocots in Archaeplastida
|
Function
The biological role of this protein is also well understood. The
fungal infections
(Richardson et al.). Seeking new means to treat such infections, drug researchers have begun targeting the 14α-demethylase enzyme in fungi; destroying the fungal cell's ability to produce ergosterol causes a disruption of the plasma membrane, thereby resulting in cellular leakage and ultimately the death of the pathogen (DrugBank).
azoles to the prosthetic heme group in the enzyme's active site causes a characteristic shift in CYP51 absorbance, creating what is commonly referred to as a type II difference spectrum.[9][10]
Prolonged use of
antifungals has resulted in the emergence of drug resistance among certain fungal strains.[3] Mutations in the coding region of CYP51 genes, overexpression of CYP51, and overexpression of membrane efflux transporters can all lead to resistance to these antifungals.[11][12][13][14][15] Consequently, the focus of azole research is beginning to shift towards identifying new ways to circumvent this major obstacle.[3]
Structure
This section needs to be updated. (January 2022) |
As of late 2007, 6
structures have been solved for this class of enzymes, with PDB accession codes 1H5Z, 1U13, 1X8V, 2BZ9, 2CI0, and 2CIB
.
References
Further reading
- Bak S, Kahn RA, Olsen CE, Halkier BA (1997). "Cloning and expression in Escherichia coli of the obtusifoliol 14 alpha-demethylase of Sorghum bicolor (L.) Moench, a cytochrome P450 orthologous to the sterol 14 alpha-demethylases (CYP51) from fungi and mammals". Plant J. 11 (2): 191–201. PMID 9076987.
- Aoyama Y, Yoshida Y (1991). "Different substrate specificities of lanosterol 14a-demethylase (P-45014DM) of Saccharomyces cerevisiae and rat liver for 24-methylene-24,25-dihydrolanosterol and 24,25-dihydrolanosterol". Biochem. Biophys. Res. Commun. 178 (3): 1064–71. PMID 1872829.
- Aoyama Y, Yoshida Y (1992). "The 4 beta-methyl group of substrate does not affect the activity of lanosterol 14 alpha-demethylase (P-450(14)DM) of yeast: difference between the substrate recognition by yeast and plant sterol 14 alpha-demethylases". Biochem. Biophys. Res. Commun. 183 (3): 1266–72. PMID 1567403.
- Alexander K, Akhtar M, Boar RB, McGhie JF, Barton DH (1972). "The removal of the 32-carbon atom as formic acid in cholesterol biosynthesis". Journal of the Chemical Society, Chemical Communications (7): 383. .

