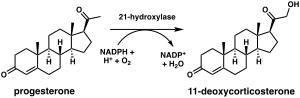
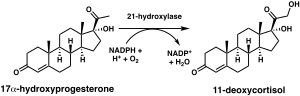
21-Hydroxylase
| Steroid 21-hydroxylase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
Steroid 21-hydroxylase is a
Steroid 21-hydroxylase is a member of the
In humans, the enzyme is localized in endoplasmic reticulum membranes of cells in adrenal cortex,[14][15] and is encoded by the CYP21A2 gene which is located near the CYP21A1P pseudogene that has high degree of sequence similarity. This similarity makes it difficult to analyze the gene at the molecular level, and sometimes leads to loss-of-function mutations of the gene due to intergenic exchange of DNA.
Gene
| CYP21A2 | |||
|---|---|---|---|
Gene ontology | |||
| Molecular function | |||
| Cellular component | |||
| Biological process | |||
| Sources:Amigo / QuickGO | |||
Ensembl |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) |
| ||||||||
| Location (UCSC) | Chr 6: 32.04 – 32.04 Mb | Chr 17: 35.02 – 35.02 Mb | |||||||
| PubMed search | [18] | [19] | |||||||
| View/Edit Human | View/Edit Mouse |

Steroid 21-hydroxylase in humans is encoded by the CYP21A2 gene that may be accompanied by one or several copies of the nonfunctional pseudogene CYP21A1P,[20][21] this pseudogene shares 98% of the exonic informational identity with the actual functional gene.[22][23]
Pseudogenes are common in genomes, and they originate as artifacts during the duplication process. Though often thought of as "junk DNA", research has shown that retaining these faulty copies can have a beneficial role, often providing regulation of their parent genes.[24]
In the mouse genome, the Cyp21a2 is a pseudogene and the Cyp21a1 is a functional gene.[25] In the chicken and quail, there is only a single Cyp21 gene, which locus is located between complement component C4 and TNX gene, along with Cenpa.[26]
CYP21A2 in humans is located in
Inside the
Due to the high degree of homology between the CYP21A2 gene and the CYP21A1P pseudogene and the complexity of the RCCX locus, it is difficult to perform molecular diagnostics for CYP21A2. The pseudogene can have single-nucleotide polymorphisms (SNP) that are identical or similar to those in the functional gene, making it difficult to distinguish between them. The pseudogene can also recombine with the functional gene, creating hybrid genes that have features of both. This can result in false-positive or false-negative results when testing for SNPs in the CYP21A2.[37]
The whole genome sequencing technology relies on breaking the DNA into small fragments, sequencing them, and then assembling them back together based on their overlaps. However, because of the high homology and variability of the CYP21A2 and its pseudogene, the fragments cannot be mapped unambiguously to either copy of the gene. This can lead to errors or gaps in the assembly, or missing some variants that are present in the gene.[38][37]
Therefore, to analyze the CYP21A2 gene accurately, a more specialized and sensitive method is needed, such as targeted long-read sequencing, which can sequence longer DNA fragments and capture more information about the gene structure and variation. However, this method is not widely available or affordable for clinical use.[42][43][44]
Protein
Steroid 21-hydroxylase, is a member of the cytochrome P450 family of monooxygenase enzymes, the protein has 494 amino acid residues with a molecular weight of 55,000. This enzyme is at most 28% homologous to other P-450 enzymes that have been studied.[45]
Structurally, the protein contains an evolutionarily conserved core of four
The iron(III) heme group that defines the active site resides in the center of each subunit. The human enzyme binds one substrate at a time.[12] In contrast, the well-characterized bovine enzyme can bind two substrates.[47] The human and bovine enzyme share 80% amino acid sequence identity, but are structurally different, particularly in loop regions, and also evident in secondary structure elements.[12]
Species
Variations of the steroid 21-hydroxylase can be found in all vertebrates.[48]
Cyp21 first emerged in
In vertebrates, such as fish, amphibians, reptiles, birds, and mammals, Cyp21 participates in the biosynthesis of glucocorticoids and mineralocorticoids, therefore, Cyp21 is essential for the regulation of stress response, electrolyte balance and blood pressure, immune system, and metabolism in vertebrates.[51]
Cyp21 is relatively conserved among mammals, and shows some variations in its structure, expression, and regulation.[51] Rhesus macaques and orangutans possess two copies of Cyp21, while chimpanzees have three, still, a pseudogene (CYP21A1P) is only present in humans among primates.[52]
Tissue and subcellular distribution
Steroid 21-hydroxylase is localized in
Unlike other enzymes of the cytochrome P450 superfamily of enzymes that are expressed in multiple tissues, with most abundant expression in the liver, in adult humans steroid 21-hydroxylase, along with steroid 11β-hydroxylase and aldosterone synthase, is almost exclusively expressed in the adrenal gland.[54][55]
As of 2023,[update] the main subcellular location for the encoded protein in human cells is not known, and is pending cell analysis.[56]
Function

The enzyme, steroid 21-hydroxylase hydroxylates steroids at the C21 position.[13]Steroids are a group of naturally occurring and synthetically produced organic compounds, steroids all share a four ring primary structure. The enzyme catalyzes the chemical reaction in which the hydroxyl group (-OH) is added at the C21 position of the steroid biomolecule. This location is on a side chain of the D ring.
The enzyme is a member of the cytochrome P450 superfamily of monooxygenase enzymes. The cytochrome P450 enzymes catalyze many reactions involved in drug metabolism and synthesis of cholesterol, steroids and other lipids.
Steroid 21-hydroxylase is essential for the biosynthesis of cortisol and aldosterone.[57][58]
Mechanism
Steroid 21-hydroxylase is a cytochrome P450 enzyme that is notable for its substrate specificity and relatively high catalytic efficiency.[48]
Like other cytochrome P450 enzymes, steroid 21-hydroxylase participates in the
The chemical reaction in which steroid 21-hydroxylase catalyzes the addition of hydroxyl (-OH) to the C21 position of progesterone, 17α-hydroxyprogesterone and 21-desoxycortisone[60] was first described in 1952.[61]
Studies of the human enzyme expressed in yeast initially classified 17-hydroxyprogesterone as the preferred substrate for steroid 21-hydroxylase,[59][62][63] however, later analysis of the purified human enzyme found a lower KM and greater catalytic efficiency for progesterone over 17-hydroxyprogesterone.[12]
The
Clinical significance
Congenital adrenal hyperplasia
The classical forms occur in approximately 1 in 10000 to 1 in 20000 births globally,
The nonclassical form is the mildest condition, retaining about 20% to 50% of enzyme function.[58] This form is associated with mild and clinically silent cortisol impairment,[65] but an excess of androgens post-puberty.[66]
Non-classic congenital adrenal hyperplasia
Non-classical congenital adrenal hyperplasia caused by 21-hydroxylase deficiency (NCCAH) is a milder and late-onset congenital adrenal hyperplasia. Its prevalence rate in different ethnic groups varies from 1 in 1000 to 1 in 50.[58] Some people affected by the condition have no relevant signs and symptoms, while others experience symptoms of hyperandrogenism.[58][65][66]
Women with NCCAH usually have normal female genitalia at birth. In later life, the signs and symptoms of the condition may include acne, hirsutism, male-pattern baldness, irregular menstruation, and infertility.[58][65][25]
Fewer studies have been published about males with NCCAH comparing to those about females, because males are generally asymptomatic.[25][58] Males, however, may present with acne[67][68] and early balding.[69][70]
While symptoms are usually diagnosed after puberty, children may present with premature adrenarche.[71]
Research on other conditions
There is ongoing research on how
History
In the 1950s and 1960s, steroidogenic pathways that included cholesterol conversion to progesterone through a complex pathway involving multiple steps were identified, and, among them, a pathway for cortisol synthesis showing specific enzymatic steps that included hydroxylation reactions at position 21 (21-hydroxylation) mediated by cytochrome P450 enzymes.[73] Cytochrome P450 enzymes were then described, and steroid 21-hydroxylation was associated with cytochrome P450.[74][73]
In the 1980s and 1990s, partial-length bovine Cyp21 cDNA clones were identified as related to human CYP21A2.[75][73] Researchers discovered mutations in the CYP21A2 gene associated with congenital adrenal hyperplasia (CAH).[73]
From the 1990s onward, specific mutations were correlated with different forms/severity levels of CAH. Genotype/phenotype correlations were investigated for improved diagnostic accuracy.[73]
See also
References
- ^ "UniProt". www.uniprot.org. Archived from the original on 28 November 2023. Retrieved 26 November 2023.
- PMID 32442933.
- PMID 28677294.
- PMID 21446712.
- ^ "Information on EC 1.14.14.16 - steroid 21-monooxygenase - BRENDA Enzyme Database". www.brenda-enzymes.org. Archived from the original on 11 October 2020. Retrieved 21 September 2020.
- PMID 31839646.
- S2CID 7197547.
- PMID 16540460.
- PMID 14640134.
- ^ a b
 This article incorporates public domain material from "NCBI: CYP21A2 cytochrome P450 family 21 subfamily A member 2". Reference Sequence collection. National Center for Biotechnology Information. Retrieved 30 November 2020.
This article incorporates public domain material from "NCBI: CYP21A2 cytochrome P450 family 21 subfamily A member 2". Reference Sequence collection. National Center for Biotechnology Information. Retrieved 30 November 2020. This gene encodes a member of the cytochrome P450 superfamily of enzymes. The cytochrome P450 proteins are monooxygenases that catalyze many reactions involved in drug metabolism and synthesis of cholesterol, steroids, and other lipids. This protein localizes to the endoplasmic reticulum and hydroxylates steroids at the 21 position. Its activity is required for the synthesis of steroid hormones including cortisol and aldosterone. Mutations in this gene cause congenital adrenal hyperplasia. A related pseudogene is located near this gene; gene conversion events involving the functional gene and the pseudogene are thought to account for many cases of steroid 21-hydroxylase deficiency. Two transcript variants encoding different isoforms have been found for this gene.
- (PDF) from the original on 25 March 2020. Retrieved 17 October 2009.
- ^ PMID 25855791.
- ^ S2CID 36860068.
- ^ PMID 27267697.
- S2CID 18927484.
- ^ a b c ENSG00000231852, ENSG00000206338, ENSG00000233151, ENSG00000232414, ENSG00000235134 GRCh38: Ensembl release 89: ENSG00000198457, ENSG00000231852, ENSG00000206338, ENSG00000233151, ENSG00000232414, ENSG00000235134 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024365 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^
 This article incorporates public domain material from "CYP21A2 cytochrome P450 family 21 subfamily A member 2 [ Homo sapiens (human) ]". Reference Sequence collection. National Center for Biotechnology Information.
This article incorporates public domain material from "CYP21A2 cytochrome P450 family 21 subfamily A member 2 [ Homo sapiens (human) ]". Reference Sequence collection. National Center for Biotechnology Information.
- S2CID 220295067.
- PMID 3486422.
- PMID 28521877.
- NCBI NBK549832.
- ^ PMID 2999780.
- PMID 15153492.
- ^ S2CID 25061446.
- PMID 2983330.
- PMID 14656967.
- ^ S2CID 36582994.
- ^ PMID 34394006.
- PMID 18179706.
- PMID 11487476.
- PMID 37401054.
- ^ PMID 23241443.
- PMID 22771554.
- ^ PMID 35422767.
- ^ S2CID 232133292.
- PMID 24071710.
- ^ "Polymerase Chain Reaction (PCR) Fact Sheet". Genome.gov. Archived from the original on 3 December 2023. Retrieved 3 December 2023.
- ^ "Southern blotting — Knowledge Hub". GeNotes. Archived from the original on 3 December 2023. Retrieved 3 December 2023.
- PMID 37842302.
- S2CID 263742489.
- S2CID 263800944.
- ^ "Steroid 21-hydroxylase | DrugBank Online". go.drugbank.com. Archived from the original on 28 November 2023. Retrieved 27 November 2023.
- PMID 11178272.
- PMID 22262854.
- ^ PMID 12424893.
- S2CID 21649057.
- S2CID 232296805.
- ^ S2CID 22716959.
- PMID 24977709.
- ISBN 978-3-319-12107-9.
- PMID 24523112.
- ^ "Tissue expression of CYP21A2". The Human Protein Atlas. Archived from the original on 28 November 2023. Retrieved 27 November 2023.
- ^ "CYP21A2 Subcellular RNA expression". The Human Protein Atlas. Archived from the original on 28 November 2023. Retrieved 27 November 2023.
- PMID 17666484.
- ^ PMID 30272171.
- ^ PMID 12464252.
- PMID 13220521.
- PMID 14934270.
- PMID 2502949.
- PMID 1707279.
- ^ "NCBI: CYP21A2 cytochrome P450 family 21 subfamily A member 2". National Center for Biotechnology Information. Archived from the original on 28 October 2020. Retrieved 30 November 2020.
This gene encodes a member of the cytochrome P450 superfamily of enzymes. The cytochrome P450 proteins are monooxygenases that catalyze many reactions involved in drug metabolism and synthesis of cholesterol, steroids, and other lipids. This protein localizes to the endoplasmic reticulum and hydroxylates steroids at the 21 position. Its activity is required for the synthesis of steroid hormones including cortisol and aldosterone. Mutations in this gene cause congenital adrenal hyperplasia. A related pseudogene is located near this gene; gene conversion events involving the functional gene and the pseudogene are thought to account for many cases of steroid 21-hydroxylase deficiency. Two transcript variants encoding different isoforms have been found for this gene.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ S2CID 221884108.
- ^ PMID 21051590.
- PMID 19936418.
- S2CID 23469344.
- PMID 16912124.
Loss of scalp hair in females and males is embarrassing, requiring treatment with 5α-reductase inhibitors
- from the original on 14 November 2020. Retrieved 25 March 2021.
- PMID 20671993.
- from the original on 2 February 2024. Retrieved 2 February 2024.
- ^ PMID 35947694.
- S2CID 27237884.
- PMID 33505400.
External links
- GeneReviews/NCBI/NIH/UW entry on 21-Hydroxylase-Deficient Congenital Adrenal Hyperplasia Archived 31 May 2010 at the Wayback Machine
- OMIM entry on 21-Hydroxylase-Deficient Congenital Adrenal Hyperplasia Archived 29 June 2011 at the Wayback Machine
- Synthesis of Desoxycorticosterone from Progesterone through 21-Hydroxylase (Image) Archived 26 April 2021 at the Wayback Machine
- Steroid+21-Hydroxylase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Human CPS1 genome location and CPS1 gene details page in the UCSC Genome Browser.
- Human CYP21A2 genome location and CYP21A2 gene details page in the UCSC Genome Browser.
- Overview of all the structural information available in the PDB for UniProt: P08686 (Steroid 21-hydroxylase) at the PDBe-KB.
This article incorporates text from the United States National Library of Medicine, which is in the public domain.