Triazane
| |
| Names | |
|---|---|
| Systematic IUPAC name
Triazane[1] | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
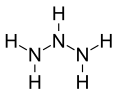
| N3H5 | |
| Molar mass | 47.061 g·mol−1 |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Triazane is an
diazene and ammonia.[3] Triazane was first synthesized as a ligand of the silver complex ion: tris(μ2-triazane-κ2N1,N3)disilver(2+).[clarification needed] Triazane has also been synthesized in electron-irradiated ammonia ices and detected as a stable gas-phase product after sublimation.[4]
Compounds containing the triazane skeleton
Several compounds containing the triazane skeleton are known, including 1-methyl-1-nitrosohydrazine (NH2−N(CH3)−N=O), produced from the solventless reaction of methylhydrazine (CH3NHNH2) and an alkyl nitrite (R–O–N=O):
- CH3NHNH2 + RONO → NH2N(CH3)NO + ROH
1-Methyl-1-nitrosohydrazine is a colorless solid, sensitive to impact, but not to friction. It melts at 45°C and decomposes at 121°C.
References
- ^ "triazane - PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information.
- ^ IUPAC Goldbook
- ISBN 9780123526519
- ^ Förstel, Maksyutenko, Jones, Sun, Chen, Chang, & Kaiser. "Detection of the Elusive Triazane Molecule ([N
3H
5]) in the Gas Phase", ChemPhysChem, 2015, 16, 3139.
External links
- 1-methyl-1-nitrosohydrazine, shows structure of 1-methyl-1-nitrosohydrazine
