Germane
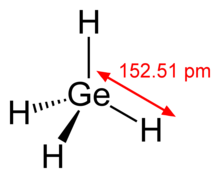
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Germane
| |||
| Other names
Germanium tetrahydride
Germanomethane Monogermane | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.029.055 | ||
| EC Number |
| ||
| 587 | |||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 2192 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| GeH4 | |||
| Molar mass | 76.62 g/mol | ||
| Appearance | Colorless gas | ||
| Odor | Pungent[1] | ||
| Density | 3.3 kg/m3 | ||
| Melting point | −165 °C (−265 °F; 108 K) | ||
| Boiling point | −88 °C (−126 °F; 185 K) | ||
| Low | |||
| Vapor pressure | >1 atm[1] | ||
| Viscosity | 17.21 μPa·s (theoretical estimate)[2] | ||
| Structure | |||
| Tetrahedral | |||
| 0 D | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Toxic, flammable, may ignite spontaneously in air | ||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H220, H280, H302, H330 | |||
| P210, P260, P264, P270, P271, P284, P301+P312, P304+P340, P310, P320, P330, P377, P381, P403, P403+P233, P405, P410+P403, P501 | |||
| NFPA 704 (fire diamond) | |||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
None[1] | ||
REL (Recommended)
|
TWA 0.2 ppm (0.6 mg/m3)[1] | ||
IDLH (Immediate danger) |
N.D.[1] | ||
| Safety data sheet (SDS) | ICSC 1244 | ||
| Related compounds | |||
Related compounds
|
Methane Silane Stannane Plumbane Germyl | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Germane is the
Occurrence
Germane has been detected in the atmosphere of Jupiter.[3]
Synthesis
Germane is typically prepared by reduction of germanium oxides, notably
- NaHGeO3 + KBH4 + H2O → KGeH3 + KB(OH)4
- KGeH3 + HO2CCH3 → GeH4 + KO2CCH3
Other methods for the synthesis of germane include
Reactions
Germane is weakly acidic. In liquid ammonia GeH4 is ionised forming NH4+ and GeH3−.[8] With alkali metals in liquid ammonia GeH4 reacts to give white crystalline MGeH3 compounds. The potassium (potassium germyl or potassium trihydrogen germanide KGeH3) and rubidium compounds (rubidium germyl or rubidium trihydrogen germanide RbGeH3) have the sodium chloride structure implying a free rotation of the trihydrogen germanide anion GeH3−, the caesium compound, caesium germyl or caesium trihydrogen germanide CsGeH3 in contrast has the distorted sodium chloride structure of TlI.[8]
Use in semiconductor industry
The gas decomposes near 600K (327°C; 620°F) to germanium and hydrogen. Because of its thermal
Safety
Germane is a highly
The
References
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0300". National Institute for Occupational Safety and Health (NIOSH).
- ISBN 978-0123958501
- doi:10.1086/160516.
- ^ W. L. Jolly "Preparation of the Volatile Hydrides of Groups IVA and VA by Means of Aqueous Hydroborate" Journal of the American Chemical Society 1961, volume 83, pp. 335-7.
- ^ US Patent 4,668,502
- ^ Girolami, G. S.; Rauchfuss, T. B.; Angelici, R. J. (1999). Synthesis and Technique in Inorganic Chemistry. Mill Valley, CA: University Science Books.
- ^ US Patent 7,087,102 (2006)
- ^ ISBN 978-0-08-037941-8.
- doi:10.1063/1.343633.
- .
- ^ Brauer, 1963, Vol.1, 715
- ^ a b Praxair MSDS Archived 2012-05-08 at the Wayback Machine accessed Sep. 2011
- ^ NIOSH Germane Registry of Toxic Effects of Chemical Substances (RTECS)accessed Sep. 2011
- PMID 4839911.
- ^ US EPA Germane
- .
External links
- Metaloids (manufacturer) datasheet
- Arkonic Specialty Gases China (manufacturer) datasheet
- Licensintorg Russia (process technology sale) Archived 2013-03-29 at the Wayback Machine
- Honjo Chemical Japan (manufacturer) Archived 2013-01-20 at the Wayback Machine
- Praxair datasheet
- Air liquide gas encyclopedia entry Archived 2013-07-30 at the Wayback Machine
- CDC - NIOSH Pocket Guide to Chemical Hazards
- Voltaix (manufacturer) datasheet Archived 2011-07-17 at the Wayback Machine
- Foshan Huate Gas Co., Ltd. (manufacturer)
- Horst Technologies, Russia (manufacturer)



