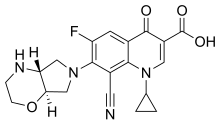
Finafloxacin
 | |
| Clinical data | |
|---|---|
| Trade names | Xtoro |
| Routes of administration | otic, oral, intavenous |
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 10 hours |
| Identifiers | |
| |
JSmol) | |
| |
| |
Finafloxacin (Xtoro) is a
Medical uses
Finafloxacin is used to treat a type of ear infection called acute otitis externa caused by Pseudomonas aeruginosa and Staphylococcus aureus bacteria.[3] In the clinical trial that led to the drug's approval, finafloxacin shortened the time to cessation of ear pain from an average of 6.8 days in patients taking a placebo to 3.5 days.[3]
Finafloxacin cannot be purchased
Available forms
Finafloxacin is commercially available as a 0.3%
Specific populations
Pregnancy
Finafloxacin is classified as pregnancy category C, meaning that the risk for harming a developing fetus has not been ruled out.[5]
Pediatrics
The efficacy and safety profile of finafloxacin ear drops are unknown in children younger than the age of 1 years old.[4]
Geriatrics
There are no limitations against using finafloxacin ear drops in the elderly.[4]
Adverse effects
The spectrum of adverse effects caused by finafloxacin vary by the method of administration. People that have administered finafloxacin into their ears in the form of drops have experienced ear
People that are allergic to other quinolones may be allergic to finafloxacin as well, and use may result in an
Overdose
It is not thought that an overdose of the otic suspension is likely to cause severe or life-threatening symptoms.[8]
Interactions
Owing to the local effect of administering finafloxacin into the ears, it is unlikely that it will affect or be affected by other medications that are administered into the
Pharmacology
Mechanism of action
Finafloxacin is a
Finafloxacin has demonstrated
Pharmacokinetics
Finafloxacin has good oral bioavailability, meaning that a substantial portion of a dose taken by mouth reaches a person's systemic circulation.[6] Some people have experienced unintentional, quantifiable absorption of finafloxacin into systemic circulation after administering the drug via the ear.[6]
The
Chemistry
The chemical structure of finafloxacin has been described as a "fluorinated
There are some notable differences between the chemistry of finafloxacin and related fluoroquinolones. For example, the 8-cyano-substituent is not found in ciprofloxacin, and
The chemical structure of finafloxacin is nearly identical to that of pradofloxacin.
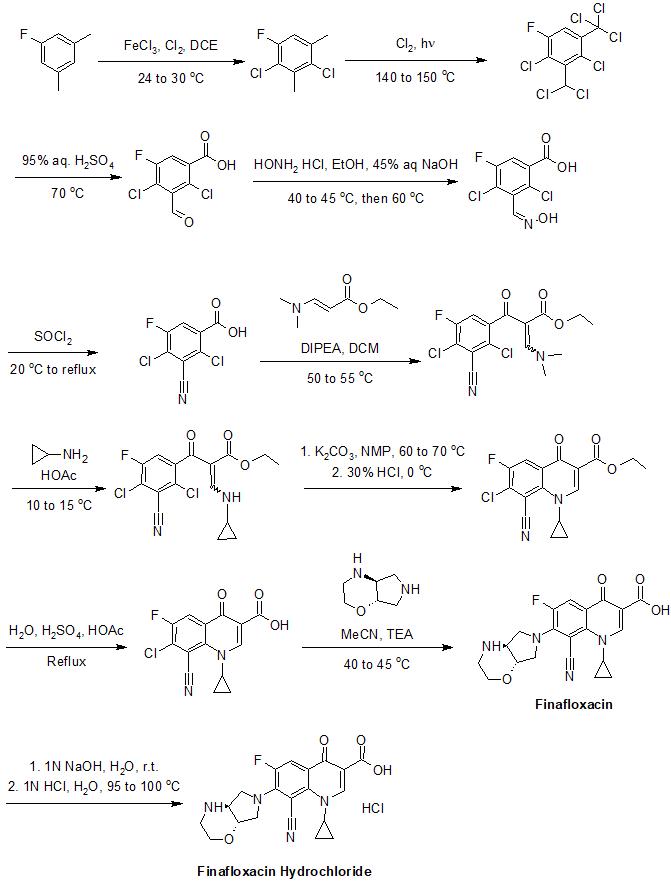
Synthesis
The synthesis of finafloxacin has been described in detail in its patents.[11] An example of its synthesis is provided below:[11]
History
Finafloxacin is the first
Research
Owing to its high bactericidal activity in acidic environments, Bartoletti et al have speculated that finafloxacin may be useful in the treatment of
References
- ^ "Health Canada New Drug Authorizations: 2016 Highlights". Health Canada. 14 March 2017. Retrieved 7 April 2024.
- ^ "FDA approves Xtoro to treat swimmer's ear". Food and Drug Administration. December 17, 2014.
- ^ a b c d e f "Finafloxacin: New fluoroquinolone for acute otitis externa". pharmacist.com. American Pharmacists Association. February 1, 2015. Retrieved 14 August 2017.
- ^ a b c d "finafloxacin (Otic route)". drugs.com. Retrieved 15 August 2017.
- ^ "Rx Update: Xtoro (Finafloxacin Otic Suspension)". contemporaryclinic.pharmacytimes.com. Contemporary Clinic 2017 Pharmacy & Healthcare Communications, LLC. Retrieved 14 August 2017.
- ^ S2CID 207488603.
- ^ PMID 27215369.
- ^ a b "Xtoro". drugs.com. Retrieved 15 August 2017.
- PMID 17016449.
- PMID 21596526.
- ^ a b "Finafloxacin". pharmacodia.com. Retrieved 14 August 2017.
- ^ Poh LC (July 29, 2017). "Biotech sector poised to deliver more health and wealth". SPH Digital News. The Straits Times. Retrieved 15 August 2017.
- ^ "Xtoro Approval History". drugs.com. Retrieved 15 August 2017.
- ^ a b Lane EJ (January 27, 2015). "Singapore's MerLion eyes partner for Phase III of Finafloxacin urinary infection trials". Questex LLC. FierceBiotech. Retrieved 15 August 2017.
- S2CID 27148906.