Clinafloxacin
 | |
| Clinical data | |
|---|---|
| Other names | AM-1091, CI-960, PD127391[1] |
| Routes of administration | oral, IV[1] |
| ATC code |
|
| Pharmacokinetic data | |
| Bioavailability | 90% (oral)[2] |
| Protein binding | 0-10%[2] |
| Elimination half-life | 6.1 hours[3] |
| Identifiers | |
| |
JSmol) | |
| |
Clinafloxacin is an
Medical uses
Clinafloxacin has not been approved for any indication. The
Available forms
Clinafloxacin is available in both oral and intravenous formulations.[1]
Specific populations
There is a warning against using clinafloxacin in pregnant patients, due to possible damage to the developing fetus.[5]
Adverse effects
The use of clinafloxacin is associated with
The phototoxicity with clinafloxacin has been more associated with oral dosing as compared to intravenous dosing, though the studies that described this were subject to confounding by study site (that is, patients that received intravenous clinafloxacin were less mobile, and thereby received less sunlight exposure).[1]
The mechanism for clinafloxacin's phototoxicity involves the
The mechanism for clinafloxacin's effect on blood glucose is thought to involve stimulation of the pancreatic
The symptoms of clinafloxacin overdose are unknown.[7]
Interactions
Drug-drug interactions
Clinafloxacin inhibits multiple
There is also a known interaction of clinafloxacin with phenytoin, resulting in a decrease in the clearance of phenytoin from the body.[1] The increase in INR seen in patients taking both clinafloxacin and the anticoagulant warfarin has yet to be fully elucidated.[1]
Food-drug interactions
There are no known food-drug interactions with clinafloxacin.[2]
Pharmacology
Mechanism of action
Clinafloxacin's antibiotic mechanism of action, like other
Clinafloxacin has been described as a
Pharmacokinetics
The time that it takes for serum concentrations of clinafloxacin to reach the maximum concentration (Cmax) in healthy volunteers after taking a dose by mouth is 0.7 hours.[3] The elimination half-life in humans is 6.1 hours.[3] Steady state levels of clinafloxacin are achieved in 3 days of twice daily dosing by mouth in healthy volunteers.[1]
About half of an administered clinafloxacin dose is found unchanged in the urine, meaning that the drug is cleared from the blood stream roughly equally by
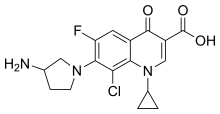
Chemistry
Clinafloxacin is a
History
In the 1990s, clinafloxacin showed promise as a novel, broad-spectrum fluoroquinolone antibiotic. However, further clinical trials raised serious concerns regarding its safety in humans, citing dangerously low blood sugar, drug-induced light sensitivity, and multiple drug-drug interactions.[1]
Research
Clinical trials have been done to assess its efficacy in sepsis and other serious and potentially life-threatening infections.[1]
See also
- Adverse effects of fluoroquinolones
References
- ^ ISBN 978-1444147520.
- ^ PMID 21834759.
- ^ S2CID 32251826.
- ^ "W-L withdraws clinafloxacin NDA". thepharmaletter.com. The Pharma Letter. Retrieved 14 September 2017.
- ^ a b c "Clinafloxacin". PubChem. U.S. National Library of Medicine. Retrieved 13 September 2017.
- S2CID 21890070.
- ^ "In case of emergency/overdose". clinafloxacin.com. Archived from the original on 12 April 2016. Retrieved 13 September 2017.
- S2CID 9396114.
- ^ "Clinafloxacin Hydrochloride (CAS 105956-99-8)". scbt.com. Santa Cruz Biotechnology, Inc. Retrieved 13 September 2017.
