Grepafloxacin
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Multum Consumer Information |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | 50% |
| Identifiers | |
| |
JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Grepafloxacin (trade name Raxar,
Clinical uses
Grepafloxacin was used for treating exacerbations of chronic bronchitis caused by susceptible bacteria (e.g. Haemophilus influenzae, Streptococcus pneumoniae, Moraxella catarrhalis),[4][5][6] community-acquired pneumonia (including those, in addition to the above germs, caused by Mycoplasma pneumoniae)[7][8] gonorrhea and non-gonococcal urethritis and cervicitis (for example caused by Chlamydia trachomatis or Ureaplasma urealyticum).[9][10]
Synthesis
The preparation of quinolones bearing a substituent at position 5 is complicated by the greater electrophilic character of the 8 position. One scheme for resolving the problem consists in blocking access to position 8 by first adding a readily removable group to that center.

The scheme starts with the conversion of the carboxylic acid in (1) to its dimethyloxazoline derivative (3) by reaction with the
Stereochemistry
Grepafloxacin contains a stereocenter and consists of two enantiomers. This is a
| Enantiomers of grepafloxacin | |
|---|---|
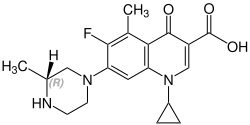 (R)-grepafloxacin CAS number: 146761-68-4 |
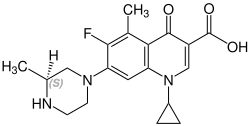 (S)-grepafloxacin CAS number: 146761-69-5 |
See also
- Quinolones
References
- ^ "Glaxo Wellcome voluntary withdrawn Raxar (Grepafloxacin)" (PDF). Food and Drug Administration. Retrieved 2014-10-12.
- ^ "Withdrawal of Product: RAXAR (grepafloxacin HCl) 600 mg Tablets, 400 mg Tablets, and 200 mg Tablets". U.S. Food and Drug Administration. Retrieved 2014-10-12.
- PMID 14992418.
- PMID 9449270.
- PMID 9484875.
- PMID 10588313.
- PMID 9484876.
- PMID 10719006.
- PMID 9484871.
- PMID 9785106.
- PMID 2002456.
- ^ WO 8906649; eidem, U.S. patent 4,920,120 (1989, 1990 both to Warner-Lambert).
- .
