Macular degeneration
| Macular degeneration | |
|---|---|
| Other names | Age-related macular degeneration |
| Frequency | 8.7% global prevalence in 2020[2] |
Macular degeneration, also known as age-related macular degeneration (AMD or ARMD), is a medical condition which may result in
Macular degeneration typically occurs in older people,
The difference between the two forms is categorized by the change in the macula. Those with dry form AMD have drusen, cellular debris in their macula that gradually damages light-sensitive cells and leads to vision loss. In wet form AMD, blood vessels grow under the macula, causing blood and fluid to leak into the retina.[4]
Exercising, eating well, and not smoking may reduce the risk of macular degeneration.
Age-related macular degeneration is a main cause of central blindness among the working-aged population worldwide.
Signs and symptoms


Early or intermediate AMD may be asymptomatic, or it may present with blurred or decreased vision in one or both eyes. This may manifest initially as difficulty with reading or driving (especially in poorly lit areas).[2] Other symptoms of AMD include distortion of vision and blind spots (especially in and around the central visual field).[2]
Other signs and symptoms of macular degeneration include:
- Distorted vision in the form of metamorphopsia, in which a grid of straight lines appears wavy and parts of the grid may appear blank: Patients often first notice this when looking at things like miniblinds in their home or telephone poles while driving. There may also be central scotomas, shadows or missing areas of vision
- Slow recovery of visual function after exposure to bright light (photostress test)
- Visual acuity drastically decreasing (two levels or more), e.g.: 20/20 to 20/80[11]
- Blurred vision: Those with nonexudative (dry) macular degeneration may be asymptomatic or notice a gradual loss of central vision, whereas those with exudative (wet) macular degeneration often notice a rapid onset of vision loss (often caused by leakage and bleeding of abnormal blood vessels).
- Trouble discerning colors, specifically dark ones from dark ones and light ones from light ones
- A loss in contrast sensitivity
- Formed visual hallucinations and flashing lights have also been associated with severe visual loss secondary to wet AMD [12]
Macular degeneration by itself will not lead to total blindness. For that matter, only a small number of people with visual impairment are totally blind. In almost all cases, some vision remains, mainly peripheral. Other complicating conditions may lead to such an acute condition (severe stroke or trauma, untreated glaucoma, etc.), but few macular degeneration patients experience total visual loss.[13]
The area of the macula constitutes only about 2.1% of the retina, and the remaining 97.9% (the peripheral field) remains unaffected by the disease. Even though the macula provides such a small fraction of the visual field, almost half of the visual cortex is devoted to processing macular information.[14]
In addition, people with dry macular degeneration often do not experience any symptoms but can experience gradual onset of blurry vision in one or both eyes.[15][16] People with wet macular degeneration may experience acute onset of visual symptoms.[15][16]
Risk factors
Key risk factors are age, race/ethnicity, smoking, and family history.[17] Advanced age is the strongest predictor of AMD, particularly over 50.[18]
Race and ethnicity
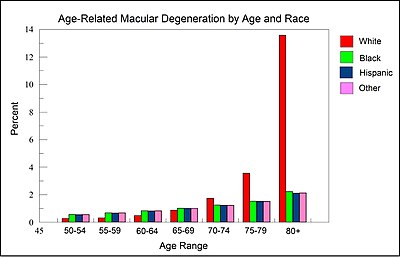
As illustrated by the Figure in this section, derived from data presented by the National Eye Institute of the United States,[19] among those over 80 years of age, White individuals are more than 6-fold more likely to develop AMD than Black or Hispanic individuals. Thus, white background is a major risk factor for AMD.
In Caucasian (White) skin, there is a specific group of polymorphic genes (with single nucleotide alterations) that encode for enzymes and transcription factors responsible for the early steps (including the first step, formation of L-DOPA from the amino acid tyrosine) of the melanin synthesis pathway. Many of these enzymes and transcription factors are reviewed by Markiewicz and Idowu.[20] Also, as reviewed by Sturm et al.[21] “increasing intracellular concentrations of either tyrosine or L-DOPA both result in an increase in melanogenesis” or formation of the black pigment melanin. Thus there appears to be an association between reduced L-DOPA production and white skin. As suggested by the Figure and information in this section, reduced L-DOPA, resulting in white skin, appears to be associated with an increased risk of macular degeneration for white individuals over the age of 80.
Environment and lifestyle
- Smoking: Smoking tobacco increases the risk of AMD by two to three times that of someone who has never smoked, and may be the most important modifiable factor in its prevention. A review of previous studies found "a strong association between current smoking and AMD. ... Cigarette smoking is likely to have toxic effects on the retina."[22]
- Hypertension (high blood pressure): In the ALIENOR study 2013, early and late AMD were not significantly associated with systolic or diastolic blood pressure (BP), hypertension, or use of antihypertensive medications, but elevated pulse pressure [(PP) systolic BP minus diastolic BP] was significantly associated with an increased risk of late AMD.[23]
- Atherosclerosis[24]
- Obesity: Abdominal obesity is a risk factor, especially among men[27]
- Fat intake: Consuming high amounts of certain fats, including omega-3 fatty acids may decrease the risk of AMD.[29]
- Exposure to
- A digital screen does not radiate harmful energy against human eyes, but staring at the screen for a long time without pauses does increase eye strain. There is no evidence to support the claim that exposure to digital screens contributes to the risk of macular degeneration.[30][16][31]
Genetics
AMD is a highly heritable condition.[17] Recurrence ratios for siblings of an affected individual are three- to six-fold higher than in the general population.[32] Genetic linkage analysis has identified 5 sets of gene variants at three locations on different chromosomes (1, 6 and 10) as explaining at least 50% of the risk.[33] These genes have roles regulating the immune response, inflammatory processes and homeostasis of the retina. Variants of these genes give rise to different kinds of dysfunction in these processes. Over time, this results in accumulation of intracellular and extracellular metabolic debris. This can cause scarring of the retina or breakdown of its vascularization.[34]
The list of genetic variations association with AMD include
Although genetic testing can lead to the identification of genetic variation which can predispose to AMD, the complex pathogenesis of the condition prevents the use of these tests in routine practice.[17] Nevertheless, they can be useful in selecting patients for clinical trials and analysing their response to treatment.[17] The three loci where identified gene variants are found are designated:
- Complement Factor H (CFH) on chromosome 1 at location 1q31.3[36]
- HTRA serine peptidase 1/Age Related Maculopathy Susceptibility 2 (HTRA1/ARMS2) on chromosome 10 at location 10q26[33]
- Complement Factor B/Complement Component 2 (CFB/CC2) on chromosome 6 at 6p21.3
Specific genes
- Polymorphisms in genes for complement system proteins: Variation in the genes for the complement system proteins factor H (CFH), factor B (CFB) and factor 3 (C3), among others, are strongly associated with a person's risk for developing AMD.[17] CFH is involved in inhibiting the inflammatory response.[17] The mutation in CFH (Y402H) results in reduced ability of the protein to localise to and protect tissues such as the retina from complement overactivation.[17] Absence of the complement factor H-related genes R3 and R1 protects against AMD.[37][38] Two independent studies in 2007 showed a certain common mutation Arg80Gly in the C3 gene, which is a central protein of the complement system, is strongly associated with the occurrence of AMD.[39][40] The authors of both papers consider their study to underscore the influence of the complement pathway in the pathogenesis of this disease.
- In two 2006 studies, another gene that has implications for the disease, called HTRA1 (encoding a secreted serine protease), was identified.[41][42]
- Six mutations of the gene SERPING1 (Serpin Peptidase Inhibitor, Clade G (C1 Inhibitor), Member 1) are associated with AMD. Mutations in this gene can also cause hereditary angioedema.[43]
- Fibulin-5 mutation: Rare forms of the disease are caused by genetic defects in fibulin-5, in an autosomal dominant manner. In 2004, Stone et al. performed a screen on 402 AMD patients and revealed a statistically significant correlation between mutations in fibulin-5 and incidence of the disease.
- Mitochondrial-related gene polymorphisms such as that in the MT-ND2 molecule, predicts wet AMD.[44][45]
Pathophysiology
The pathogenesis of age-related macular degeneration is not well known, although some theories have been put forward, including oxidative stress, mitochondrial dysfunction, and inflammatory processes.
The imbalance between the production of damaged cellular components and degradation leads to the accumulation of harmful products, for example, intracellular lipofuscin and extracellular drusen. Incipient atrophy is demarcated by areas of retinal pigment epithelium (RPE) thinning or depigmentation that precede geographic atrophy in the early stages of AMD. In advanced stages of AMD, atrophy of the RPE (geographic atrophy) and/or development of new blood vessels (neovascularization) result in the death of photoreceptors and central vision loss.
In the dry (nonexudative) form, drusen accumulates between the retina and the choroid, causing atrophy and scarring to the retina. In the wet (exudative) form, which is more severe, blood vessels grow up from the choroid (neovascularization) behind the retina which can leak exudate and fluid and also cause hemorrhaging.
Early work demonstrated a family of immune mediators was plentiful in drusen.[46] Complement factor H (CFH) is an important inhibitor of this inflammatory cascade, and a disease-associated polymorphism in the CFH gene strongly associates with AMD.[17][47][48][49][50][51] Thus an AMD pathophysiological model of chronic low grade complement activation and inflammation in the macula has been advanced.[52][53] Lending credibility to this has been the discovery of disease-associated genetic polymorphisms in other elements of the complement cascade including complement component 3 (C3).[39]
A powerful predictor of AMD is found on chromosome 10q26 at LOC 387715. An insertion/deletion polymorphism at this site reduces expression of the ARMS2 gene though destabilization of its mRNA through deletion of the polyadenylation signal.[54] ARMS2 protein may localize to the mitochondria and participate in energy metabolism, though much remains to be discovered about its function.
Other gene markers of progression risk includes tissue inhibitor of metalloproteinase 3 (TIMP3), suggesting a role for extracellular matrix metabolism in AMD progression.[55] Variations in cholesterol metabolising genes such as the hepatic lipase, cholesterol ester transferase, lipoprotein lipase and the ATP-binding cassette A1 correlate with disease progression. The early stigmata of disease, drusen, are rich in cholesterol, offering face validity to the results of genome-wide association studies.[56]
Stages
In AMD there is a progressive accumulation of characteristic yellow deposits, called drusen (buildup of extracellular proteins and lipids), in the macula (a part of the retina), between the retinal pigment epithelium and the underlying choroid. This accumulation is believed to damage the retina over time. Amyloid beta, which builds up in Alzheimer's disease brains, is one of the proteins that accumulate in AMD, which is a reason why AMD is sometimes called "Alzheimer's of the eye" or "Alzheimer's of the retina".[57] AMD can be divided into 3 stages: early, intermediate, and late, based partially on the extent (size and number) of drusen.[1]
AMD-like pathology begins with small yellow deposits (drusen) in the macula, between the retinal pigment epithelium and the underlying choroid. Most people with these early changes (referred to as age-related maculopathy) still have good vision. People with drusen may or may not develop AMD. In fact, the majority of people over age 60 have drusen with no adverse effects. The risk of developing symptoms is higher when the drusen are large and numerous, and associated with the disturbance in the pigmented cell layer under the macula. Large and soft drusen are thought to be related to elevated cholesterol deposits.
Early AMD
Early AMD is diagnosed based on the presence of medium-sized drusen, about the width of an average human hair. Early AMD is usually asymptomatic.[1]
Intermediate AMD
Intermediate AMD is diagnosed by large drusen and/or any retinal pigment abnormalities. Intermediate AMD may cause some vision loss, but, like early AMD, it is usually asymptomatic.[1][58]
Recently, subgroups of intermediate AMD have been identified, which have a very high risk of progression toward late AMD. This subgroup (depending on the precise definitions) is termed nascent GA and/or iRORA (incomplete retinal pigment epithelium and outer retinal atrophy).[59][60] These 'high-risk' subgroups of intermediate AMD can be used to inform patients of theirs prognosis. In addition, these can be applied in clinical trials as endpoints.
Late AMD
In late AMD, enough retinal damage occurs that, in addition to drusen, people will also begin to experience symptomatic central vision loss. The damage can either be the development of atrophy or the onset of neovascular disease. Late AMD is further divided into two subtypes based on the types of damage: Geographic atrophy and Wet AMD (also called Neovascular AMD).[58][1]
Dry AMD
Dry AMD (also called nonexudative AMD) is a broad designation, encompassing all forms of AMD that are not neovascular (wet AMD). This includes early and intermediate forms of AMD, as well as the advanced form of dry AMD known as geographic atrophy. Dry AMD patients tend to have minimal symptoms in the earlier stages; visual function loss occurs more often if the condition advances to geographic atrophy. Dry AMD accounts for 80–90% of cases and tends to progress slowly. In 10–20% of people, dry AMD progresses to the wet type.
Geographic atrophy
Geographic atrophy (also called atrophic AMD) is an advanced form of AMD in which progressive and irreversible loss of retinal cells leads to a loss of visual function. There are multiple layers that make up the retina, and in geographic atrophy, there are three specific layers that undergo atrophy: the choriocapillaris, retinal pigment epithelium, and the overlying photoreceptors.
The three layers that undergo atrophy in geographic atrophy are all adjacent to each other. The photoreceptors are the most superficial and they are the cells that are responsible for converting energy from the light from the outside world, into an electrical signal to be sent to the brain. There are several functions of the retinal pigment epithelium. One of the main functions of the retinal pigment epithelium is to minimize oxidative stress. It does so by absorbing light, and thus preventing it from getting to the underlying layers. The layers underlying the retinal pigment epithelium are very vascularlized so they have very high oxygen tension. Thus, if light was to get to those layers, many free radicals would form and cause damage to nearby tissues. The deepest layer that undergoes atrophy in geographic atrophy is called the choriocappilaris. It is a capillary network that provides nutrients to the retinal pigment epithelium.
The pathophysiology of geographic atrophy is still uncertain. Some studies questioned whether it was due to a deficient retinal pigment epithelium, leading to increased oxidative stress.[61] Other studies have looked for inflammatory causes of damage.[62] Thus far, the medical community is still not certain. Recent studies have begun to look at each layer individually. They found that decreased blood flow in the choriocapillaris precedes atrophy of the retinal pigment epithelium and the overlying photoreceptors.[63] Since the choriocapillaris is a vascular layer, this may be used as an argument for why geographic atrophy could be a disease due to decreased blood flow.
Wet AMD
Diagnosis

Diagnosis of age-related macular degeneration depends on signs in the
Diagnosis of dry (or early stage) AMD may include the following clinical examinations as well as procedures and tests:
- The transition from dry to wet AMD can happen rapidly, and if it is left untreated can lead to legal blindness in as little as six months. To prevent this from occurring and to initiate preventive strategies earlier in the disease process, dark adaptation testing may be performed. A dark adaptometer can detect subclinical AMD at least three years earlier than it is clinically evident.[65]
- There is a loss of contrast sensitivity, so that contours, shadows, and color vision are less vivid. The loss in contrast sensitivity can be quickly and easily measured by a contrast sensitivitytest like Pelli Robson performed either at home or by an eye specialist.
- When viewing an Amsler grid, some straight lines appear wavy and some patches appear blank[66]
- When viewing a Snellen chart, at least 2 lines decline
- In dry macular degeneration, which occurs in 85–90 percent of AMD cases, drusen spots can be seen in Fundus photography
- Using an electroretinogram, points in the macula with a weak or absent response compared to a normal eye may be found
- Farnsworth-Munsell 100 hue testand Maximum Color Contrast Sensitivity test (MCCS) for assessing color acuity and color contrast sensitivity
- Optical coherence tomography is now used by most ophthalmologists in the diagnosis and the follow-up evaluation of the response to treatment with antiangiogenic drugs.
Diagnosis of wet (or late stage) AMD may include the following in addition to the above tests:
- Preferential hyperacuity perimetry changes (for wet AMD). Preferential hyperacuity perimetry is a test that detects drastic changes in vision and involves the macula being stimulated with distorted patterns of dots and the patient identification of where in the visual field this occurs.[67]
- In wet macular degeneration, angiography can visualize the leakage of bloodstream behind the macula. Fluorescein angiography allows for the identification and localization of abnormal vascular processes.
Histology
- Pigmentary changes in the retina – In addition to the pigmented cells in the iris (the colored part of the eye), there are pigmented cells beneath the retina. As these cells break down and release their pigment, dark clumps of released pigment and later, areas that are less pigmented may appear
- Exudative changes: hemorrhagesin the eye, hard exudates, subretinal/sub-RPE/intraretinal fluid
- Drusen, tiny accumulations of extracellular material that build up on the retina. While there is a tendency for drusen to be blamed for the progressive loss of vision, drusen deposits can be present in the retina without vision loss. Some patients with large deposits of drusen have normal visual acuity. If normal retinal reception and image transmission are sometimes possible in a retina when high concentrations of drusen are present, then, even if drusen can be implicated in the loss of visual function, there must be at least one other factor that accounts for the loss of vision.
Management
Treatment of AMD varies depending on the category of the disease at the time of diagnosis. In general, treatment is aimed at slowing down the progression of AMD.[68] As of 2018, there are no treatments to reverse the effects of AMD.[68] Early-stage and intermediate-stage AMD is managed by modifying known risk factors such as smoking cessation, management of hypertension and atherosclerosis and making dietary modifications.[2][68] For intermediate-stage AMD, management also includes antioxidant and mineral supplementation.[68][69] Advanced-stage AMD is managed based on the presence of choroidal neovascularization (CNV): dry AMD (no CNV present) or wet AMD (CNV present).[68] No effective treatments exist for dry AMD.[68] The CNV present in wet AMD is managed with vascular endothelial growth factor (VEGF) inhibitors.[68][70][69] Daily use of an Amsler grid or other home visual monitoring tools can be used to monitor for development of distorted vision, which may be a sign of disease progression.[2]
Dietary supplements
Dietary supplements may be suggested for people with AMD, with the goal of reducing damage to the cells in the retina with antioxidants. The formulations commonly suggested are known as
With regards to AREDS-1 compared with AREDS-2 formulations, there is only weak evidence comparing the effectiveness of each formulation and the effectiveness of lutein and zeaxanthin as a replacement in the AREDS-2 formulation.[5]
Dry AMD
Pegcetacoplan (Syfovre)[71][72] and avacincaptad pegol (Izervay)[73][74] are approved for medical use in the United States. In 2023 it was reported that the aging pigment lipofuscin can be broken down with the help of melanin and drugs through a newly discovered mechanism.[75] The pigment lipofuscin plays a central role in the development of dry AMD and Stargardt's disease. The clinical development of this mechanism, which has the potential to clear Bruch's membrane and to reduce formation of Drusen, is in preparation.
Wet AMD
Ranibizumab, aflibercept, brolucizumab, and faricimab are approved VEGF inhibitors for the treatment of CNV in wet AMD.[70][76] All three drugs are administered via intravitreal injection, meaning they are injected directly into the eye. Bevacizumab is another VEGF inhibitor that has been shown to have similar efficacy and safety as the previous two drugs, however, is not currently indicated for AMD.[69] AMD can also be treated with laser coagulation therapy.[77]
A randomized control trial found that
These anti-VEGF agents may be administered monthly or adaptively. For adaptive anti-VEGF treatment, two approaches are conventionally applied. In the case of pro re nata, the patient comes at fixed intervals, but treatment is only administered if an activity is detected (i.e., presence of fluid). In the case of treat-and-extend, the patients always receive treatment, but the interval to the next visit is extended if the lesion was inactive.[84] Recently, researchers have started to apply AI algorithms to predict the future need for treatment.[85][86][87]
The
Photodynamic therapy has also been used to treat wet AMD.[92] The drug verteporfin is administered intravenously; light of a certain wavelength is then applied to the abnormal blood vessels. This activates the verteporfin destroying the vessels.
Cataract surgery could improve visual outcomes for people with AMD, though there have been concerns about surgery increasing the progression of AMD. A randomized controlled trial found that people who underwent immediate cataract surgery (within two weeks) had improved visual acuity and better quality of life outcomes than those who underwent delayed cataract surgery (6 months).[93]
Radiotherapy has been proposed as a treatment for wet AMD but the evidence to support the use of modern stereotactic radiotherapy combined with anti-VEGF is currently uncertain and is awaiting the results of ongoing studies.[94]
Nucleoside reverse transcription inhibitors like they are used in anti-HIV therapy was associated with a reduced risk of developing atrophic macular degeneration. This is because Alu elements undergo L1 (protein)-mediated reverse transcription in the cytoplasm resulting in DNA synthesis. First clinical trials are being prepared as of January 2021.[95]
Adaptive devices

Because peripheral vision is not affected, persons with macular degeneration can learn to use their remaining vision to partially compensate.[96] Assistance and resources are available in many countries and every state in the U.S.[97] Classes for "independent living" are given and some technology can be obtained from a state department of rehabilitation.
Adaptive devices can help people read. These include magnifying glasses, special eyeglass lenses, computer screen readers, electronic glasses, and TV systems that enlarge the reading material.
Computer screen readers such as
Video cameras can be fed into standard or special-purpose computer monitors, and the image can be zoomed in and magnified. These systems often include a movable table to move the written material.
Epidemiology

The prevalence of any age-related macular degeneration is higher in Europeans than in Asians and Africans.[99] There is no difference in prevalence between Asians and Africans.[99] The incidence of age-related macular degeneration and its associated features increases with age and is low in people <55 years of age.[100] Smoking is the strongest modifiable risk factor.[101] As of 2008, age-related macular degeneration accounts for more than 54% of all vision loss in the white population in the US.[102] An estimated 8 million Americans are affected with early age-related macular degeneration, of whom over 1 million will develop advanced age-related macular degeneration within the next 5 years. In the UK, age-related macular degeneration is the cause of blindness in almost 42% of those who go blind aged 65–74 years, almost two-thirds of those aged 75–84 years, and almost three-quarters of those aged 85 years or older.[102]
Research
Studies indicate drusen associated with AMD are similar in molecular composition to amyloid beta (Aβ) plaques and deposits in other age-related diseases such as Alzheimer's disease and atherosclerosis. This suggests that similar pathways may be involved in the etiologies of AMD and other age-related diseases.[103]
Genetic testing
Genetic testing can help identify whether a patient with AMD is at a greater risk of developing the condition and can inform disease progression.
Genome editing
CRISPR-Cas9 genome editing may be used to treat wet age-related macular degeneration caused by VEGFA. Scientists described an approach in which engineered lentiviruses are injected into the affected anatomical regions for transient editing that could reduce the area of choroidal neovascularization by 63% without inducing undesired off-target edits or anti-Cas9 immune responses.[105][106]
RPE, L-DOPA and Melanin in amelioration of wet AMD

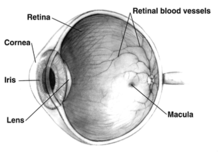
The retinal pigment epithelium (RPE) (see diagram) has an essential role in the eye. It secretes a large variety of factors including at least 22 proteins important in maintaining the structure, function and micro-environments on the two sides of the RPE.[107] (The two sides of the RPE include the choroid side, where blood vessels form and bring nourishment to the eye, and the photoreceptor side, with rods and cones that receive light signals.) In particular, the RPE secretes vascular endothelial growth factor (VEGF) at its basement membrane, with the VEGF reaching the choriocapillaris to maintain proper blood vessel formation in the choroid region.
Many factors, including genetic factors, hypoxia, oxidative stress and inflammatory stressors, may cause pathologic over-production of VEGF by the RPE. This over-production causes excess blood vessel formation in the choroid region (the choriocapillaris), which is a major cause of wet AMD.[108]
It was recently discovered that the aging pigment lipofuscin can be broken down with the help of melanin and drugs through a newly discovered mechanism (chemical excitation).[109] The pigment lipofuscin plays a central role in the development of dry AMD and geographic atrophy. This breakdown can be supported by medication. This discovery can be translated into the development of a therapy to treat dry AMD.
Artificial intelligence for prediction
Research is exploring if artificial intelligence can help in predicting wet AMD early enough to make prevention possible. A study tested an AI model for predicting whether people with wet AMD in one eye would develop it in the other within six months. Compared to doctors and optometrists the AI model predicted the development more accurately.[110][111]
Other types
There are a few other (rare) kinds of macular degeneration with similar symptoms but unrelated in etiology to Wet or Dry age-related macular degeneration. They are all genetic disorders that may occur in childhood or middle age.
- Vitelliform macular dystrophy
- Sorsby's fundus dystrophy is an autosomal dominant, retinal disease characterized by sudden acuity loss resulting from untreatable submacular neovascularisation
- Stargardt's disease(juvenile macular degeneration, STGD) is an autosomal recessive retinal disorder characterized by juvenile-onset macular dystrophy, alterations of the peripheral retina, and subretinal deposition of lipofuscin-like material.
Similar symptoms with a very different etiology and different treatment can be caused by
Notable cases
See also
- Ophthalmology
- Macula of retina
- Visual impairment
- Gene therapy for color blindness
- Gene therapy of the human retina
- Stem cell therapy for macular degeneration
- macular pucker
References
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab "Facts About Age-Related Macular Degeneration". National Eye Institute. June 2015. Archived from the original on 22 December 2015. Retrieved 21 December 2015.
- ^ S2CID 236926930.
- ^ PMID 26319344.
- ^ "Age-Related Macular Degeneration". WebMD. Retrieved 15 March 2022.
- ^ PMID 37702300.
- PMID 26060829.
- PMID 36312607.
- ^ AMDF (18 May 2016). "Risk Factors for Macular Degeneration - AMDF". Retrieved 23 May 2024.
- PMID 26063472.
- ^ "Age-related Macular Degeneration". 22 October 2013. Archived from the original on 22 October 2013. Retrieved 5 November 2018.
- ^ "Nandhana Eye Hospital". nandhanaeyehospital.com. Retrieved 19 May 2023.
- ^ "Age-Related Macular Degeneration PPP – Updated 2015". American Academy of Ophthalmology. 29 January 2015. Archived from the original on 13 November 2018. Retrieved 13 November 2018.
- ^ Roberts DL (September 2006). "The First Year – Age Related Macular Degeneration". Marlowe & Company: 100.
- ^ Roberts DL (September 2006). "The First Year – Age Related Macular Degeneration". Marlowe & Company: 20.
- ^ S2CID 205396269.
- ^ S2CID 25049931.
- ^ S2CID 226274874.
- PMID 21609220.
- ^ "Age-Related Macular Degeneration (AMD) Tables". National Eye Institute.
- PMID 32210602.
- PMID 11602344.
- PMID 16151432.
- PMID 23404120.
- PMID 29042759.
- PMID 21851605.
- PMID 28456421.
- PMID 21422060.
- PMID 19901214.
- PMID 17502507.
- ^ PMID 29576617.
- ^ "Are Computer Glasses Worth It?". American Academy of Ophthalmology. 27 April 2017. Retrieved 2 February 2020.
- S2CID 15830119.
- ^ PMID 35113155. Retrieved 3 February 2022.
- ^ a b "Understanding Wet AMD". EverydayHealth.com. 10 June 2022. Retrieved 19 May 2023.
- PMID 33330470.
- S2CID 31337096.
- S2CID 21178921.
- PMID 20843825.
- ^ PMID 17634448.
- S2CID 8595223.
- S2CID 19577378.
- S2CID 85725181.
- ^ Hirschler B (7 October 2008). "Gene discovery may help hunt for blindness cure". Reuters. Archived from the original on 11 October 2008. Retrieved 7 October 2008.
- PMID 19151382.
- PMID 18461138.

- S2CID 24601453.
- PMID 15870199.
- PMID 17167412.
- PMID 16849663.
- PMID 16936733.
- S2CID 32716116.
- PMID 20711712.
- PMID 21566137.
- S2CID 205344383.
- PMID 20385819.
- PMID 20385826.
- PMID 26088679.
- ^ PMID 23332590.
- S2CID 211263484.
- PMID 33766801.
- PMID 27239555.
- PMID 26852158.
- S2CID 86851083.
- ^ PMID 29400919.
- PMID 26522707.
- ^ "Amsler grid". myhealth.alberta.ca. Retrieved 19 May 2023.
- PMID 24788016.
- ^ a b c d e f g Bishop P. Age-related macular degeneration. BMJ Best Practice. 2018.
- ^ PMID 25856365.
- ^ PMID 26857947.
- ^ "Syfovre- pegcetacoplan injection, solution". DailyMed. 23 February 2023. Retrieved 6 June 2023.
- ^ "FDA Approves Syfovre (pegcetacoplan injection) as the First and Only Treatment for Geographic Atrophy (GA), a Leading Cause of Blindness" (Press release). Apellis Pharmaceuticals. 17 February 2023. Archived from the original on 17 February 2023. Retrieved 18 February 2023 – via GlobeNewswire.
- ^ "Novel Drug Approvals for 2023". U.S. Food and Drug Administration (FDA). 18 August 2023. Retrieved 25 August 2023.
- ^ "Iveric Bio Receives U.S. FDA Approval for Izervay (avacincaptad pegol intravitreal solution), a New Treatment for Geographic Atrophy" (Press release). Astellas Pharma Inc. 4 August 2023. Retrieved 25 August 2023 – via PR Newswire.
- ^ Lyu Y, Tschulakow AV, Wang K, Brash DE, Schraermeyer U. Chemiexcitation and melanin in photoreceptor disc turnover and prevention of macular degeneration. Proc Natl Acad Sci U S A. 2023;120(20):e2216935120.
- PMID 32107066.
- PMID 17636773.
- PMID 23870813.
- PMID 25220133.
- ^ Copley C, Hirschler B (24 April 2012). "Novartis challenges UK Avastin use in eye disease". Reuters. Archived from the original on 22 May 2013.
- PMID 21417187.
- ^ "FDA Approves New Drug Treatment for Age-Related Macular Degeneration". FDA.gov. U.S. Food and Drug Administration. Archived from the original on 20 November 2015.
- ^ "FDA Approves Eylea for Macular Degeneration". www.medpagetoday.com. 19 November 2011. Archived from the original on 28 May 2013.
- S2CID 47012435.
- PMID 34185055.
- PMID 28837729.
- PMID 33971352.
- ^ "Age-Related Macular Degeneration PPP – Updated 2015". American Academy of Ophthalmology Preferred Practice Pattern. 29 January 2015. Archived from the original on 21 October 2016. Retrieved 22 October 2016.
- PMID 26804762.
- PMID 26493180.
- PMID 17636773.
- PMID 12709292.
- PMID 28206671.
- S2CID 221326233.
- S2CID 231761522.
- ^ "Low Vision Rehabilitation Delivery Model". Mdsupport.org. Archived from the original on 7 November 2010. Retrieved 11 January 2011.
- ^ "Agencies, Centers, Organizations, & Societies". Mdsupport.org. 1 September 2005. Archived from the original on 11 June 2011. Retrieved 11 January 2011.
- ^ "WHO Disease and injury country estimates". World Health Organization. 2009. Archived from the original on 11 November 2009. Retrieved 11 November 2009.
- ^ PMID 25104651.
- ^ "Age-related macular degeneration - Symptoms, diagnosis and treatment". BMJ Best Practice. Retrieved 13 November 2018.
- PMID 24368940.
- ^ PMID 19027484.
- S2CID 24601453.
- ^ "EYE-RISK - EYE-RISK". www.eyerisk.eu. Retrieved 15 May 2021.
- PMID 33580230.
- S2CID 230508724. Retrieved 7 March 2021.
- PMID 23663427.
- PMID 18653375.
- ^ Lyu Y, Tschulakow AV, Wang K, Brash DE, Schraermeyer U. Chemiexcitation and melanin in photoreceptor disc turnover and prevention of macular degeneration. Proc Natl Acad Sci U S A. 2023;120(20):e2216935120.
- S2CID 243121774.
- S2CID 218682821.
- ^ "Judi Dench 'can't read any more due to failing eyesight Archived 2016-10-19 at the Wayback Machine", The Guardian, 23 February 2014
- ^ "Joan bows out to a standing ovation Archived 2016-04-20 at the Wayback Machine", The Guardian, 13 May 2014
- ^ "Patrons of the Macular Society Archived 2013-02-08 at the Wayback Machine", Macular Society. (notable for doing the voice of Wallace from Wallace and Gromit until 2012)
External links
- Macular degeneration at Curlie
