Kearns–Sayre syndrome
| Kearns–Sayre syndrome | |
|---|---|
| Other names | Oculocraniosomatic disorder or Oculocranionsomatic neuromuscular disorder with ragged red fibers |
| Specialty | Ophthalmology |
Kearns–Sayre syndrome (KSS), oculocraniosomatic disorder or oculocranionsomatic neuromuscular disorder with ragged red fibers is a
Signs and symptoms
Individuals with KSS present initially in a similar way to those with typical CPEO. Onset is in the first and second decades of life.[citation needed]
The first symptom of this disease is a unilateral ptosis, or difficulty opening the eyelids, that gradually progresses to a bilateral ptosis. As the ptosis worsens, the individual commonly extends their neck, elevating their chin in an attempt to prevent the eyelids from occluding the visual axis. Along with the insidious development of ptosis, eye movements eventually become limited causing a person to rely more on turning the head side to side or up and down to view objects in the peripheral visual field.[citation needed]
Mitochondrial retinopathy

Kearns and Sayre described patients with "pigmentary degeneration" on funduscopy, night vision abnormalities, and some histologic similarities, but also clinical differences, to retinitis pigmentosa[2] Subsequently, the retinal phenotype of KSS was described as retinitis pigmentosa, atypical retinitis pigmentosa, tapetoretinal degeneration, salt-and-pepper retinopathy, and pigmentary retinopathy. As the clinical characterization, however, was not always comprehensive the term "mitochondrial retinopathy" appears most accurate and the diagnosis of RP may have been imprecise.[3] Patients with KSS show widespread granular pigmented alterations in the posterior fundus which correspond to granular patterns on fundus autofluorescence imaging. Associated changes on optical coherence tomography (OCT) include reflectivity changes predominantly at the level of the ellipsoid and interdigitation zone and an increased distance between the ellipsoid band and the retinal pigment epithelium.[3] Night blindness may be seen in patients with KSS. Visual acuity loss is usually mild and only occurs in 40–50% of patients.[4]
Cardiac conduction abnormalities
These most often occur years after the development of ptosis and ophthalmoplegia.
Cerebral folate deficiency
Kearns-Sayre patients are consistently found to have
Cause and prevalence
As characterized in Kearns's original publication in 1965 and in later publications, inconsistent features of KSS that may occur are weakness of facial, pharyngeal, trunk, and extremity muscles,
Kearns–Sayre syndrome occurs spontaneously in the majority of cases. In some cases it has been shown to be inherited through mitochondrial, autosomal dominant, or autosomal recessive inheritance. There is no predilection for race or sex, and there are no known risk factors. As of 1992 there were only 226 cases reported in published literature.[9] Although NIH and other studies estimate occurrence in the population to be 1–3 and some as high as 9 in 100,000 individuals but a failure to be referred to specialist centres and recognise the disease symptoms is common [9]
Genetics
KSS is the result of deletions in
There are other factors involved in the manifestation of a mitochondrial disease besides the size and location of a mutation. Mitochondria replicate during each cell division during gestation and throughout life. Because the mutation in mitochondrial disease most often occurs early in gestation in these diseases, only those mitochondria in the mutated lineage are defective. This results in an uneven distribution of dysfunctional mitochondria within each cell, and among different tissues of the body. This describes the term
More recent studies have concluded that mtDNA duplications may also play a significant role in determining what phenotype is present. Duplications of mtDNA seem to be characteristic of all cases of KSS and Pearson's syndrome, while they are absent in CPEO.[13][14]
Deletions of mtDNA in KSS vary in size (1.3–8kb), as well as position in the
Diagnosis
A
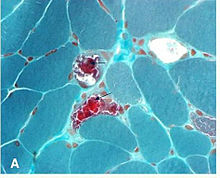
Biopsy findings
It is not necessary to biopsy an ocular muscle to demonstrate histopathologic abnormalities. Cross-section of muscle fibers stained with
Laboratory studies
Blood
Management
Currently there is no curative treatment for KSS. Because it is a rare condition, there are only case reports of treatments with very little data to support their effectiveness. Several promising discoveries have been reported which may support the discovery of new treatments with further research. Satellite cells are responsible for muscle fiber regeneration. It has been noted that mutant mtDNA is rare or undetectable in satellite cells cultured from patients with KSS. Shoubridge et al. (1997) asked the question whether wildtype mtDNA could be restored to muscle tissue by encouraging muscle regeneration. In the forementioned study, regenerating muscle fibers were sampled at the original biopsy site, and it was found that they were essentially homoplasmic for wildtype mtDNA.[11] Perhaps with future techniques of promoting muscle cell regeneration and satellite cell proliferation, functional status in KSS patients could be greatly improved.[citation needed]
One study described a patient with KSS who had reduced serum levels of
A screening ECG is recommended in all patients presenting with CPEO. In KSS, implantation of
Screening for endocrinologic disorders should be performed, including measuring
In December 2022 researchers reported a study with modest results in five patients affected by either Pearson syndrome or Kearns–Sayre syndrome.[20][21]
History
The triad of CPEO, bilateral pigmentary retinopathy, and cardiac conduction abnormalities was first described in a case report of two patients in 1958 by Thomas P. Kearns (1922–2011), MD., and George Pomeroy Sayre (1911–1992), MD.[22] A second case was published in 1960 by Jager and co-authors reporting these symptoms in a 13-year-old boy.[23] Previous cases of patients with CPEO dying suddenly had been published, occasionally documented as from a cardiac dysrhythmia. Other cases had noted a peculiar pigmentation of the retina, but none of these publications had documented these three pathologies occurring together as a genetic syndrome.[24] Kearns published a defining case in 1965 describing nine unrelated cases with this triad.[24] In 1988, the first connection was made between KSS and large-scale deletions of muscle mitochondrial DNA (abbreviated mtDNA)[25][26] Since this discovery, numerous deletions in mitochondrial DNA have been linked to the development of KSS.[27][28][29]
References
- S2CID 24560049.
- PMID 13558799.
- ^ S2CID 235822866.
- ^ ISBN 9780781763790.
- S2CID 44622892.
- PMID 25539952.
- PMID 20731822.
- PMID 791168.
- ^ a b c Kearns-Sayre Syndrome at eMedicine
- S2CID 40192499.
- ^ PMID 9361028.
- PMID 1635816.
- ^ PMID 7951243.
- ISBN 9780781763790.
- ^ PMID 11715003
- ^ Rubin, Richard M.; Sadun, Alfredo A. (2008). "Ch. 9.17 Ocular Myopathies". In Yanoff, Myron; Duker, Jason (eds.). Ophthalmology (Online Textbook) (3rd ed.). Mosby.
- S2CID 27569662.
- PMID 12379588.
- ^ Kearns-Sayre Syndrome~diagnosis at eMedicine
- ^ "Moms' mitochondria may refresh cells in sick kids". www.science.org. Retrieved 2023-01-09.
- S2CID 254998216.
- .
- PMID 13789175.
- ^ PMID 16693635.
- S2CID 30046555.
- S2CID 6811844.
- S2CID 30046555.
- PMID 10480366. Archived from the originalon 2000-09-29.
- PMID 8411717.
External links
- kearns_sayre at NINDS
- Kearns Sayre syndrome at NIH's Office of Rare Diseases
