Industrial wastewater treatment

| Part of a series on |
| Pollution |
|---|
 |
Industrial wastewater treatment describes the processes used for
Most industries produce some
Types
Industrial facilities may generate the following industrial wastewater flows:[citation needed]
- Manufacturing process wastestreams, which can include conventional pollutants (i.e. controllable with secondary treatment systems), toxic pollutants (e.g. solvents, heavy metals), and other harmful compounds such as nutrients
- Non-process wastestreams: cooling water, which produce thermal pollutionand other pollutants
- Industrial site drainage, generated both by manufacturing facilities, service industries and energy and mining sites
- Wastestreams from the energy and mining sectors: acid mine drainage, produced water from oil and gas extraction, radionuclides
- Wastestreams that are by-products of treatment or cooling processes: backwashing (water treatment), brine.
Contaminants
Industrial wastewater could add the following pollutants to receiving water bodies if the wastewater is not treated and managed properly:
- Heavy metals, including mercury, lead, and chromium
- food waste: Certain industries (e.g. food processing, slaughterhouse waste, paper fibers, plant material, etc.) discharge high concentrations of BOD, ammonia nitrogen and oil and grease.[5]: 180 [6]
- , etc.;
- , etc.
- Microplastics such as polyethylene and polypropylene beads, polyester and polyamide[10]
- Thermal pollution from power stations and industrial manufacturers
- Radionuclides from uranium mining, processing nuclear fuel, operating nuclear reactors, or disposal of radioactive waste.
- Some industrial discharges include persistent organic pollutants such as per- and polyfluoroalkyl substances (PFAS).[11][12]
Industrial sectors
The specific pollutants generated and the resultant effluent concentrations can vary widely among the industrial sectors.[citation needed]
Battery manufacturing
Battery manufacturers specialize in fabricating small devices for electronics and portable equipment (e.g., power tools), or larger, high-powered units for cars, trucks and other motorized vehicles. Pollutants generated at manufacturing plants includes cadmium, chromium, cobalt, copper, cyanide, iron, lead, manganese, mercury, nickel, silver, zinc, oil and grease.[13]
Centralized waste treatment
A centralized waste treatment (CWT) facility processes liquid or solid industrial wastes generated by off-site manufacturing facilities. A manufacturer may send its wastes to a CWT plant, rather than perform treatment on site, due to constraints such as limited land availability, difficulty in designing and operating an on-site system, or limitations imposed by environmental regulations and permits. A manufacturer may determine that using a CWT is more cost-effective than treating the waste itself; this is often the case where the manufacturer is a small business.[14]
CWT plants often receive wastes from a wide variety of manufacturers, including chemical plants, metal fabrication and finishing; and used oil and petroleum products from various manufacturing sectors. The wastes may be classified as
Chemical manufacturing
Organic chemicals manufacturing
The specific pollutants discharged by organic chemical manufacturers vary widely from plant to plant, depending on the types of products manufactured, such as bulk organic chemicals, resins, pesticides, plastics, or synthetic fibers. Some of the organic compounds that may be discharged are benzene, chloroform, naphthalene, phenols, toluene and vinyl chloride. Biochemical oxygen demand (BOD), which is a gross measurement of a range of organic pollutants, may be used to gauge the effectiveness of a biological wastewater treatment system, and is used as a regulatory parameter in some discharge permits. Metal pollutant discharges may include chromium, copper, lead, nickel and zinc.[16]
Inorganic chemicals manufacturing
The inorganic chemicals sector covers a wide variety of products and processes, although an individual plant may produce a narrow range of products and pollutants. Products include aluminum compounds; calcium carbide and calcium chloride; hydrofluoric acid; potassium compounds; borax; chrome and fluorine-based compounds; cadmium and zinc-based compounds. The pollutants discharged vary by product sector and individual plant, and may include arsenic, chlorine, cyanide, fluoride; and heavy metals such as chromium, copper, iron, lead, mercury, nickel and zinc.[17]
Electric power plants

Ash ponds, a type of surface impoundment, are a widely used treatment technology at coal-fired plants. These ponds use gravity to settle out large particulates (measured as total suspended solids) from power plant wastewater. This technology does not treat dissolved pollutants. Power stations use additional technologies to control pollutants, depending on the particular wastestream in the plant. These include dry ash handling, closed-loop ash recycling, chemical precipitation, biological treatment (such as an activated sludge process), membrane systems, and evaporation-crystallization systems.[18] Technological advancements in ion-exchange membranes and electrodialysis systems has enabled high efficiency treatment of flue-gas desulfurization wastewater to meet recent EPA discharge limits.[19] The treatment approach is similar for other highly scaling industrial wastewaters.
Food industry

Wastewater generated from agricultural and
Processing of food from raw materials requires large volumes of high grade water. Vegetable washing generates water with high loads of particulate matter and some dissolved organic matter. It may also contain surfactants and pesticides.
Aquaculture facilities (fish farms) often discharge large amounts of nitrogen and phosphorus, as well as suspended solids. Some facilities use drugs and pesticides, which may be present in the wastewater.[21]
Dairy processing plants generate conventional pollutants (BOD, SS).[22]
Processing food for sale produces wastes generated from cooking which are often rich in plant organic material and may also contain
Food processing activities such as plant cleaning, material conveying, bottling, and product washing create wastewater. Many food processing facilities require on-site treatment before operational wastewater can be land applied or discharged to a waterway or a sewer system. High suspended solids levels of organic particles increase BOD and can result in significant sewer surcharge fees. Sedimentation, wedge wire screening, or rotating belt filtration (microscreening) are commonly used methods to reduce suspended organic solids loading prior to discharge.[citation needed]
Iron and steel industry
The production of iron from its ores involves powerful reduction reactions in blast furnaces. Cooling waters are inevitably contaminated with products especially ammonia and cyanide. Production of coke from coal in coking plants also requires water cooling and the use of water in by-products separation. Contamination of waste streams includes gasification products such as benzene, naphthalene, anthracene, cyanide, ammonia, phenols, cresols together with a range of more complex organic compounds known collectively as polycyclic aromatic hydrocarbons (PAH).[25]
The conversion of iron or steel into sheet, wire or rods requires hot and cold mechanical transformation stages frequently employing water as a lubricant and coolant. Contaminants include
Metal working
Many industries perform work on metal feedstocks (e.g. sheet metal, ingots) as they fabricate their final products. The industries include automobile, truck and aircraft manufacturing; tools and hardware manufacturing; electronic equipment and office machines; ships and boats; appliances and other household products; and stationary industrial equipment (e.g. compressors, pumps, boilers). Typical processes conducted at these plants include grinding, machining, coating and painting, chemical etching and milling, solvent degreasing, electroplating and anodizing. Wastewater generated from these industries may contain heavy metals (common heavy metal pollutants from these industries include cadmium, chromium, copper, lead, nickel, silver and zinc), cyanide and various chemical solvents, oil, and grease.[26][27]
Mines and quarries

The principal waste-waters associated with
Wastewater from metal mines and ore recovery plants are inevitably contaminated by the minerals present in the native rock formations. Following crushing and extraction of the desirable materials, undesirable materials may enter the wastewater stream. For metal mines, this can include unwanted metals such as zinc and other materials such as arsenic. Extraction of high value metals such as gold and silver may generate slimes containing very fine particles in where physical removal of contaminants becomes particularly difficult.[30]
Additionally, the geologic formations that harbour economically valuable metals such as
Nuclear industry
The waste production from the nuclear and radio-chemicals industry is dealt with as Radioactive waste.[citation needed]
Researchers have looked at the bioaccumulation of strontium by Scenedesmus spinosus (algae) in simulated wastewater. The study claims a highly selective biosorption capacity for strontium of S. spinosus, suggesting that it may be appropriate for use of nuclear wastewater.[31]
Oil and gas extraction
Petroleum refining and petrochemicals
Pollutants discharged at petroleum refineries and petrochemical plants include conventional pollutants (BOD, oil and grease, suspended solids), ammonia, chromium, phenols and sulfides.[33]
Pharmaceutical manufacturing
Pharmaceutical plants typically generate a variety of process wastewaters, including solvents, spent acid and caustic solutions, water from chemical reactions, product wash water, condensed steam, blowdown from air pollution control scrubbers, and equipment washwater. Non-process wastewaters typically include cooling water and site runoff. Pollutants generated by the industry include
Pulp and paper industry

Effluent from the
Smelters
The pollutants discharged by nonferrous
Typical treatment processes used in the industry are chemical precipitation, sedimentation and filtration.[36]: 145
Textile mills
Textile dyeing plants generate wastewater that contain synthetic (e.g., reactive dyes, acid dyes, basic dyes, disperse dyes, vat dyes, sulphur dyes, mordant dyes, direct dyes, ingrain dyes, solvent dyes, pigment dyes)[39] and natural dyestuff, gum thickener (guar) and various wetting agents, pH buffers and dye retardants or accelerators. Following treatment with polymer-based flocculants and settling agents, typical monitoring parameters include BOD, COD, color (ADMI), sulfide, oil and grease, phenol, TSS and heavy metals (chromium, zinc, lead, copper).
Industrial oil contamination
Industrial applications where oil enters the wastewater stream may include vehicle wash bays, workshops, fuel storage depots, transport hubs and power generation. Often the wastewater is discharged into local sewer or trade waste systems and must meet local environmental specifications. Typical contaminants can include solvents, detergents, grit, lubricants and hydrocarbons.
Water treatment
Many industries have a need to treat water to obtain very high quality water for their processes. This might include pure chemical synthesis or boiler feed water. Also, some water treatment processes produce organic and mineral sludges from
Wood preserving
Wood preserving plants generate conventional and toxic pollutants, including arsenic, COD, copper, chromium, abnormally high or low pH, phenols, suspended solids, oil and grease.[40]
Treatment methods
The various types of contamination of wastewater require a variety of strategies to remove the contamination.
An industrial wastewater treatment plant may include one or more of the following rather than the conventional treatment sequence of sewage treatment plants:
- An API oil-water separator, for removing separate phase oil from wastewater.[42]: 180
- A clarifier, for removing solids from wastewater.[43]: 41–15
- A roughing filter, to reduce the biochemical oxygen demand of wastewater.[43]: 23–11
- A carbon filtration plant, to remove toxic dissolved organic compounds from wastewater.[42]: 210
- An advanced electrodialysis reversal (EDR) system with ion-exchange membranes.
Brine treatment
Brine treatment involves removing dissolved salt ions from the waste stream. Although similarities to seawater or brackish water desalination exist, industrial brine treatment may contain unique combinations of dissolved ions, such as hardness ions or other metals, necessitating specific processes and equipment.
Brine treatment systems are typically optimized to either reduce the volume of the final discharge for more economic disposal (as disposal costs are often based on volume) or maximize the recovery of fresh water or salts. Brine treatment systems may also be optimized to reduce electricity consumption, chemical usage, or physical footprint.
Brine treatment is commonly encountered when treating cooling tower blowdown, produced water from steam-assisted gravity drainage (SAGD), produced water from natural gas extraction such as coal seam gas, frac flowback water, acid mine or acid rock drainage, reverse osmosis reject, chlor-alkali wastewater, pulp and paper mill effluent, and waste streams from food and beverage processing.
Brine treatment technologies may include: membrane filtration processes, such as
Reverse osmosis may not be viable for brine treatment, due to the potential for fouling caused by hardness salts or organic contaminants, or damage to the reverse osmosis membranes from
Evaporation processes are the most widespread for brine treatment as they enable the highest degree of concentration, as high as solid salt. They also produce the highest purity effluent, even distillate-quality. Evaporation processes are also more tolerant of organics, hydrocarbons, or hardness salts. However, energy consumption is high and corrosion may be an issue as the prime mover is concentrated salt water. As a result, evaporation systems typically employ titanium or duplex stainless steel materials.
Brine management
Brine management examines the broader context of brine treatment and may include consideration of government policy and regulations,
Solids removal
Most solids can be removed using simple sedimentation techniques with the solids recovered as slurry or sludge. Very fine solids and solids with densities close to the density of water pose special problems. In such case filtration or ultrafiltration may be required. Although flocculation may be used, using alum salts or the addition of polyelectrolytes. Wastewater from industrial food processing often requires on-site treatment before it can be discharged to prevent or reduce sewer surcharge fees. The type of industry and specific operational practices determine what types of wastewater is generated and what type of treatment is required. Reducing solids such as waste product, organic materials, and sand is often a goal of industrial wastewater treatment. Some common ways to reduce solids include primary sedimentation (clarification), dissolved air flotation (DAF), belt filtration (microscreening), and drum screening.
Oils and grease removal
The effective removal of oils and grease is dependent on the characteristics of the oil in terms of its suspension state and droplet size, which will in turn affect the choice of separator technology. Oil in industrial waste water may be free light oil, heavy oil, which tends to sink, and emulsified oil, often referred to as soluble oil. Emulsified or soluble oils will typically required "cracking" to free the oil from its emulsion. In most cases this is achieved by lowering the pH of the water matrix.
Most separator technologies will have an optimum range of oil droplet sizes that can be effectively treated. Each separator technology will have its own performance curve outlining optimum performance based on oil droplet size. the most common separators are gravity tanks or pits, API oil-water separators or plate packs, chemical treatment via dissolved air flotations, centrifuges, media filters and hydrocyclones.
Analyzing the oily water to determine droplet size can be performed with a video particle analyser.
API oil-water separators
Hydrocyclone
Hydrocyclone separators operate on the process where wastewater enters the cyclone chamber and is spun under extreme centrifugal forces more than 1000 times the force of gravity. This force causes the water and oil droplets (or solid particles) to separate. The separated materials is discharged from one end of the cyclone where treated water is discharged through the opposite end for further treatment, filtration or discharge. Hydrocyclones can also be utilised in a variety of context from solid-liquid separation to oil-water separation.[55][56][57][58]
Removal of biodegradable organics
Biodegradable organic material of plant or animal origin is usually possible to treat using extended conventional sewage treatment processes such as activated sludge or trickling filter.[1][59] Problems can arise if the wastewater is excessively diluted with washing water or is highly concentrated such as undiluted blood or milk. The presence of cleaning agents, disinfectants, pesticides, or antibiotics can have detrimental impacts on treatment processes.[citation needed]
Activated sludge process
The activated sludge process is a type of biological wastewater treatment process for treating sewage or industrial wastewaters using aeration and a biological floc composed of bacteria and protozoa. It uses air (or oxygen) and microorganisms to biologically oxidize organic pollutants, producing a waste sludge (or floc) containing the oxidized material.
The activated sludge process for removing carbonaceous pollution begins with an aeration tank where air (or oxygen) is injected into the waste water. This is followed by a settling tank to allow the biological flocs (the sludge blanket) to settle, thus separating the biological sludge from the clear treated water. Part of the waste sludge is recycled to the aeration tank and the remaining waste sludge is removed for further treatment and ultimate disposal.Trickling filter process
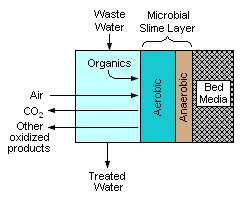

A trickling filter consists of a bed of
Removal of other organics
Synthetic organic materials including solvents, paints, pharmaceuticals, pesticides, products from coke production and so forth can be very difficult to treat. Treatment methods are often specific to the material being treated. Methods include advanced oxidation processing, distillation, adsorption, ozonation, vitrification, incineration, chemical immobilisation or landfill disposal. Some materials such as some detergents may be capable of biological degradation and in such cases, a modified form of wastewater treatment can be used.
Removal of acids and alkalis
Acids and alkalis can usually be neutralised under controlled conditions. Neutralisation frequently produces a precipitate that will require treatment as a solid residue that may also be toxic. In some cases, gases may be evolved requiring treatment for the gas stream. Some other forms of treatment are usually required following neutralisation.
Waste streams rich in hardness ions as from de-ionisation processes can readily lose the hardness ions in a buildup of precipitated calcium and magnesium salts. This precipitation process can cause severe furring of pipes and can, in extreme cases, cause the blockage of disposal pipes. A 1-metre diameter industrial marine discharge pipe serving a major chemicals complex was blocked by such salts in the 1970s. Treatment is by concentration of de-ionisation waste waters and disposal to landfill or by careful pH management of the released wastewater.
Removal of toxic materials
Toxic materials including many organic materials, metals (such as zinc, silver, cadmium, thallium, etc.) acids, alkalis, non-metallic elements (such as arsenic or selenium) are generally resistant to biological processes unless very dilute. Metals can often be precipitated out by changing the pH or by treatment with other chemicals. Many, however, are resistant to treatment or mitigation and may require concentration followed by landfilling or recycling. Dissolved organics can be incinerated within the wastewater by the advanced oxidation process.
Smart capsules
Removal of thermal pollution
To remove heat from wastewater generated by power plants or manufacturing plants, and thus to reduce thermal pollution, the following technologies are used:
- cooling ponds, engineered bodies of water designed for cooling by evaporation, convection, and radiation
- cooling towers, which transfer waste heat to the atmosphere through evaporation or heat transfer
- cogeneration, a process where waste heat is recycled for domestic or industrial heating purposes.[61]
Other disposal methods
Some facilities such as oil and gas wells may be permitted to pump their wastewater underground through injection wells. However, wastewater injection has been linked to induced seismicity.[62]
Costs and trade waste charges
Economies of scale may favor a situation where industrial wastewater (with pre-treatment or without treatment) is discharged to the sewer and then treated at a large municipal sewage treatment plant. Typically, trade waste charges are applied in that case. Or it might be more economical to have full treatment of industrial wastewater on the same site where it is generated and then discharging this treated industrial wastewater to a suitable surface water body. This effectively reduces wastewater treatment charges collected by municipal sewage treatment plants by pre-treating wastewaters to reduce concentrations of pollutants measured to determine user fees.[63]: 300–302
Industrial wastewater plants may also reduce raw water costs by converting selected wastewaters to reclaimed water used for different purposes.
Society and culture
Global goals
The international community has defined the treatment of industrial wastewater as an important part of sustainable development by including it in Sustainable Development Goal 6. Target 6.3 of this goal is to "By 2030, improve water quality by reducing pollution, eliminating dumping and minimizing release of hazardous chemicals and materials, halving the proportion of untreated wastewater and substantially increasing recycling and safe reuse globally".[64] One of the indicators for this target is the "proportion of domestic and industrial wastewater flows safely treated".[65]
See also
- Best management practice for water pollution (BMP)
- List of waste water treatment technologies
- Purified water (for industrial use)
- Water purification (for drinking water)
References
- ^ ISBN 0-07-041878-0.
- OCLC 48053912.
- ISSN 1476-1777.
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ "Pollution Prevention Case Studies". Washington, D.C.: U.S. Environmental Protection Agency (EPA). 11 August 2021.
- OCLC 48053912.
- ISBN 978-1-119-30450-0 – via Google Books.
- PMID 25889547.
- S2CID 39997737.
- S2CID 4073701.
- S2CID 58567561.
- PMID 32452041.
- S2CID 219974801.
- ^ "Battery Manufacturing Effluent Guidelines". EPA. 12 June 2017.
- ^ "Chapter 4. Description of the Industry". Development Document for Effluent Limitations Guidelines for the Centralized Waste Treatment Industry (Report). EPA. August 2000. EPA 821-R-00-020.
- ^ "Centralized Waste Treatment Effluent Guidelines". EPA. 24 January 2022.
- ^ Development Document for Effluent Limitations Guidelines, New Source Performance Standards and Pretreatment Standards for the Organic Chemicals, Plastics And Synthetic Fibers Point Source Category; Volume I (Report). EPA. October 1987. EPA 440/1-87/009.
- ^ EPA (1982). "Inorganic Chemicals Manufacturing Point Source Category." Code of Federal Regulations, 40 CFR 415
- ^ a b "Effluent Limitations Guidelines and Standards for the Steam Electric Power Generating Point Source Category". EPA. 30 September 2015.
- ^ "Lowering Cost and Waste in Flue Gas Desulfurization Wastewater Treatment". Power Mag. Electric Power. March 2017. Retrieved 6 April 2017.
- ^ European Environment Agency. Copenhagen, Denmark. "Indicator: Biochemical oxygen demand in rivers (2001)." Archived 2006-09-18 at the Wayback Machine
- ^ EPA (2002-09-12). "Effluent Limitations Guidelines and New Source Performance Standards for the Concentrated Aquatic Animal Production Point Source Category." Proposed rule. Federal Register, 67 FR 57876
- ^ "Dairy Products Processing Effluent Guidelines". EPA. 30 November 2018.
- ^ Technical Development Document for the Final Effluent Limitations Guidelines and Standards for the Meat and Poultry Products Point Source Category (Report). EPA. 2004. EPA 821-R-04-011.
- ^ "Fats, Oils, & Grease". Special Wastewater Discharge Requirements. Laurel, MD: Washington Suburban Sanitary Commission. 29 September 2021.
- ^ "7. Wastewater Characterization". Development Document for Final Effluent Limitations Guidelines and Standards for the Iron and Steel Manufacturing Point Source Category (Report). EPA. 2002. pp. 7–1ff. EPA 821-R-02-004.
- ^ "Metal Finishing Effluent Guidelines". EPA. 5 July 2019.
- ^ "Metal Products and Machinery Effluent Guidelines". EPA. 13 July 2021.
- ^ Development Document for Effluent Limitations Guidelines and Standards for the Mineral Mining and Processing Category (Report). EPA. July 1979. EPA 440/1-76/059b.
- ^ Development Document for the Coal Mining Category (Report). EPA. September 1982. EPA 440/1-82/057.
- ^ a b Development Document for Final Effluent Limitations Guidelines and New Source Performance Standards for the Ore Mining and Dressing Point Source Category (Report). EPA. November 1982. EPA 440/1-82/061.
- PMID 24919131.
- ^ Development Document for Interim Final Effuent Limitations Guidelines and Proposed New Source Performance Standards for the Oil and Gas Extraction Point Source Category (Report). EPA. September 1976. pp. 41–45. EPA 440/1-76/055a.
- ^ Guide for the Application of Effluent Limitations Guidelines for the Petroleum Refining Industry (Report). EPA. June 1985. p. 5.
- ^ "Chapters 5–7" (PDF). Development Document for Final Effluent Limitations Guidelines and Standards for the Pharmaceutical Manufacturing Point Source Category (Report). EPA. July 1998. EPA 821-R-98-005.
- ^ Permit Guidance Document: Pulp, Paper and Paperboard Manufacturing Point Source Category (Report). EPA. 2000. pp. 4–1ff. EPA-821-B-00-003.
- ^ a b c Development Document for Effluent Limitations Guidelines and Standards for the Nonferrous Metals Manufacturing Point Source Category; Volume 1 (Report). EPA. May 1989. EPA 440/1-89/019.1.
- ^ EPA (1984). "Nonferrous Metals Manufacturing Point Source Category." Code of Federal Regulations, 40 CFR 421
- ^ "Textile Mills Effluent Guidelines". EPA. 30 June 2017.
- ISBN 978-1-84569-695-5.
- ^ "Timber Products Processing Effluent Guidelines". EPA. 13 March 2018.
- JSTOR 25042250..
- ^ OCLC 1988397.
- ^ OCLC 4493039.
- S2CID 227523802.
- S2CID 234110033.
- PMID 34340085.
- S2CID 164218318.
- S2CID 237700530.
- S2CID 250296056.
- S2CID 199070455.
- S2CID 233658167.
- ISSN 0892-6875.
- S2CID 259493806.
- LCCN 67019834.
- S2CID 258368865.
- S2CID 237698803.
- S2CID 240531707.
- ISSN 0892-6875.
- LCCN 67019834.
- PMID 28731297.
- ^ Profile of the Fossil Fuel Electric Power Generation Industry (Report). EPA. September 1997. p. 24. EPA/310-R-97-007.
- ISSN 1525-2027.
- OCLC 1176821.
- ^ United Nations (2017) Resolution adopted by the General Assembly on 6 July 2017, Work of the Statistical Commission pertaining to the 2030 Agenda for Sustainable Development (A/RES/71/313)
- ^ Ritchie, Roser, Mispy, Ortiz-Ospina. "Measuring progress towards the Sustainable Development Goals, Goal 6" SDG-Tracker.org, website (2018).
Further reading
- Water Environment Federation (2020). Industrial Wastewater Management, Treatment & Disposal; Manual of Practice FD-3 (3rd ed.). Alexandria, VA: Water Environment Federation. ISBN 978-1-57278-369-0.
External links
- Water Environment Federation - Professional society
- Industrial Wastewater Treatment Technology Database - EPA


