Aetokthonotoxin

| |
| Names | |
|---|---|
| IUPAC name
5,7-Dibromo-2-(2,3,5-tribromoindol-1-yl)-1H-indole-3-carbonitrile
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
PubChem CID
|
|
| |
| |
| Properties | |
| C17H6Br5N3 | |
| Molar mass | 651.776 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
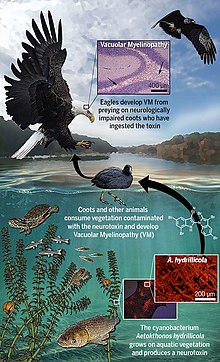
Aetokthonotoxin (AETX), colloquially known as eagle toxin, is a chemical compound that was identified in 2021 as the cyanobacterial neurotoxin causing
Biosynthesis

The biosynthesis of AETX and the functions of the enzymes AetA, AetB, AetD, AetE, and AetF were described in 2022.[5] AetF, a FAD-dependent halogenase, brominates L-tryptophan at the C5 position. The 5-bromo-L-tryptophan can then undergo two separate reactions. One route involves a second bromination by AetF at C7 to yield 5,7-dibromo-L-tryptophan. This molecule then goes on to react with AetD, an iron-dependent nitrile synthase, to form dibromo-indole-3-carbonitrile. The second route for 5-bromo-L-tryptophan involves the tryptophanase AetE, which converts the 5-bromo-L-tryptophan into 5-bromoindole. 5-bromoindole can then go on to react with a different FAD-dependent halogenase AetA to form 2,3,5-tribromoindole. 2,3,5-tribromoindole and dibromo-indole-3-carbonitrile then undergo biaryl coupling facilitated by the cytochrome P-450 AetB to form AETX.
See also
References
- PMID 33766860.
- ^ "Avian vacuolar myelinopathy". USGS National Wildlife Health Center. Archived from the original on 6 October 2014. Retrieved 24 October 2013.
- PMID 15465716.
- )
- ^ S2CID 246702060.
