Muscimol

| |
| |

| |
| Names | |
|---|---|
| IUPAC name
5-(Aminomethyl)-1,2-oxazol-3(2H)-one
| |
| Other names
Agarin, Pantherine, Agarine, Pantherin
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 774694 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.018.574 |
| EC Number |
|
IUPHAR/BPS |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
| UN number | 2811 3077 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties[1] | |
| C4H6N2O2 | |
| Molar mass | 114.104 g·mol−1 |
| Melting point | 184 to 185 °C (363 to 365 °F; 457 to 458 K) |
| very soluble | |
| Solubility in ethanol | slightly soluble |
| Solubility in methanol | very soluble |
| Pharmacology | |
| Legal status |
|
| Hazards | |
| GHS labelling:[2] | |

| |
| Danger | |
| H300 | |
| P264, P270, P301+P316, P321, P330, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Muscimol (also known as agarin or pantherine) is one of the principal
Muscimol went under clinical trial phase I for epilepsy, but the trial was discontinued.[4]
Muscimol, an
Biochemistry
Muscimol is one of the psychoactive compounds responsible for the effects of Amanita muscaria intoxication. Ibotenic acid, a neurotoxic secondary metabolite of Amanita muscaria, serves as a prodrug to muscimol when the mushroom is ingested or dried, converting to muscimol via decarboxylation.[6]
Muscimol is produced in the mushrooms

Pharmacology
Muscimol is a potent
Scientific studies have shown that dosing of the active ingredient muscimol is usually not precise as it has to be extracted from dried amanita mushroom. However, a psychoactive dose of muscimol is reported to be between 8 and 15 mg. As little as a gram of dried Amanita muscaria button may contain this amount of muscimol; however, the potency varies greatly among mushrooms. [14]
When consumed, a substantial percentage of muscimol goes un-metabolized and thus excreted in urine, a phenomenon exploited by Siberian practitioners of the traditional entheogenic use of Amanita muscaria.[15]
In patients with Huntington's disease and chronic schizophrenia, oral doses of muscimol have been found to cause a rise of both prolactin and growth hormone.[16]
During a test involving rabbits connected to an
Effects
These include euphoria, dream-like (lucid) state of mind, out-of-body experiences and synesthesia.[citation needed] Negative effects include mild to moderate nausea, stomach discomfort, increased salivation and muscle twitching or tremors. In large doses strong dissociation or delirium may be felt.[citation needed] Many of muscimol's effects are consistent with its pharmacology as a
Chemistry
Structure
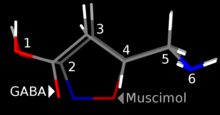
Muscimol was first isolated from Amanita pantherina by Onda in 1964,[20] and thought to be an amino acid or peptide. Structure was then elucidated by Takemoto,[21] Eugster,[22] and Bowden.[23] Muscimol is a semi-rigid isoxazole containing both alcohol and aminomethyl substituents.[24] Muscimol is commonly portrayed as a tautomer, where it adopts an amide-like configuration.[25] It is also commonly shown as a zwitterion.[26]
Isolation
Muscimol can be extracted from the flesh of the
In instances where pure muscimol is not required, such as recreational or spiritual use, a crude extract is often prepared by simmering dried Amanita muscaria in water for thirty minutes.[29]
Chemical synthesis
Muscimol was synthesized in 1965 by Gagneux,[30] who utilized a bromo-isoxazole starting material in a two step reaction. 3-bromo-5-aminomethyl-isoxazole (1) was refluxed in a mixture of methanol and potassium hydroxide for 30 hours, resulting in 3-methoxy-5-aminomethyl-isoxazole (2) with a yield of 60%.

(2) was then refluxed in concentrated

Chemists report having struggled to reproduce these results.[31][32] More dependable and scalable procedures have been developed, two examples being the syntheses of McCarry[33] and Varasi.[26]
McCarry's synthesis is a three step synthesis involving a

Varasi's synthesis is notable for its inexpensive starting materials and mild conditions. It begins with the combination of 2,3-Dichloro-1-propene (6),
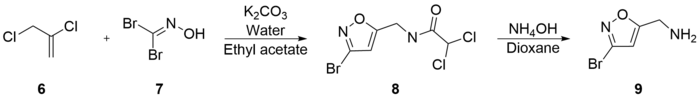
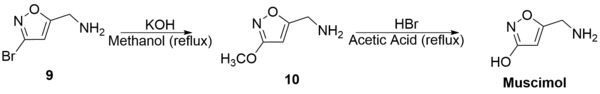
(9) was then refluxed with potassium hydroxide in methanol to generate 5-Aminomethyl-3-methoxyisoxazole (10) with a 66% yield. Subsequent reflux of (10) with hydrobromic acid and acetic acid generated muscimol with a yield of 62%. The overall synthetic yield was 30%.[26]
Toxicity
The median lethal dose in mice is 3.8 mg/kg s.c, 2.5 mg/kg i.p. The LD50 in rats is 4.5 mg/kg i.v, 45 mg/kg orally.[34]
Human deaths are rare, mainly occurring in young children, the elderly, or those with serious chronic illnesses.[35]
Legal status
Australia
Muscimol is considered a Schedule 9 prohibited substance in Australia under the Poisons Standard (October 2015). A Schedule 9 substance is a substance "which may be abused or misused, the manufacture, possession, sale or use of which should be prohibited by law except when required for medical or scientific research, or for analytical, teaching or training purposes with approval of Commonwealth and/or State or Territory Health Authorities."[36]
United States
Neither
Muscimol may be regulated on a state level. Louisiana State Act 159 banned the possession and cultivation of the Amanita muscaria except for ornamental or aesthetic purposes. Except as a constituent of lawfully manufactured food or dietary supplements, the act outlaws preparations of the Amanita muscaria intended for human consumption, including muscimol.[40]
See also
- Ibotenic acid
- Gaboxadol
- Thiomuscimol
- CI-966
- Z-drugs
References
- The Merck Index, 12th Edition
- ^ "Muscimol". pubchem.ncbi.nlm.nih.gov.
- S2CID 13364321.
- ISSN 0148-396X.
- PMID 37732408.
- ^ PMID 30565258.
- PMID 985999.
- ^ PMID 12747324.
- ISBN 9780849351853.
- PMID 12171573.
- PMID 7589211.
- PMID 17005728.
- PMID 8386310.
- ^ "Amanita muscaria - an overview | ScienceDirect Topics".
- ISBN 978-0-19-514664-6.
- PMID 162520.
- S2CID 26824149.
- PMID 24049236.
- ^ Ty M. "Exploring Muscimol: Amanita Muscaria's Most Prevalent Psychoactive Compound". www.acslab.com. ACS Laboratory. Retrieved 26 October 2023.
- PMID 14199180.
- PMID 14266560.
- PMID 5891631.
- S2CID 4178793.
- S2CID 93397543.
- ^ "Muscimol". pubchem.ncbi.nlm.nih.gov.
- ^ .
- ^ "Cellulose Phosphate: Product Information" (PDF). Sigma Aldrich. Retrieved 23 April 2020.
- PMID 14291871.
- ^ Heinrich C. "Erowid Psychoactive Amanitas (A. muscaria & A. pantherina) Vault: Amanita muscaria Preparation for Beginners". erowid.org. Retrieved 6 May 2020.
- ^ .
- .
- .
- ^ .
- ^ "Erowid Psychoactive Amanitas Vault: Chemistry". erowid.org. Retrieved 2018-04-05.
- OCLC 29913834., Preview Google Books.
- ^ "Poisons Standard". The Government of Australia. October 2015.
- ^ "Controlled Substance Schedules". deadiversion.usdoj.gov. US Department of Justice. Retrieved 6 May 2020.
- ^ Erowid. "Erowid Psychoactive Amanitas Vault: Legal Status". erowid.org. Retrieved 6 May 2020.
- ^ Sam O (May 31, 2023). "Mood-altering mushroom sales bloom despite safety concerns". Health News Florida. Retrieved June 6, 2023.
- ^ "Louisiana Act No 159". legis.la.gov. Louisiana State Legislature. Retrieved 6 May 2020.

