EA-3990
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
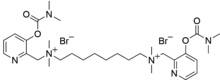
N1,N8-Bis({3-[(dimethylcarbamoyl)oxy]pyridin-2-yl}methyl)-N1,N1,N8,N8-tetramethyloctane-1,8-bis(aminium) dibromide | |
| Identifiers | |
3D model (
JSmol ) |
|
| |
| |
| Properties | |
| C30H50N6O4 · Br2 | |
| Molar mass | 718.7 g/mol |
| Appearance | white, odorless crystalline solid. |
| Density | 1.33 g/cm3 |
| Melting point | 190–191 °C |
| Solubility | soluble in alcohols, acetic acid and chloroform |
| Vapor pressure | negligible |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
6.3 µg/kg for mice and 2.6 µg/kg for rabbits via IV |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
EA-3990 is a deadly
Patent assigned to US army for EA-3990 among other similar nerve agents was filed in December 7, 1967.[3]
Lethality
EA-3990 lethality in humans is unknown but estimates have been made.
Carbamates like EA-3990 are well absorbed by the lungs, gastrointestinal tracts, and the skin.
Intravenous LD50 for EA-3990 is 0.0063 mg/kg for mice and 0.0026 mg/kg for rabbits.[3]
Properties
EA-3990's CAS is 110913-95-6, mass 718.7 g/mol,[1] melting point 190–191 °C,[3] density 1.33 g/cm3, vapor pressure is negligible, and it is soluble in alcohols, acetic acid and chloroform. It is a white, odorless crystalline solid. EA-3990 evaporates slowly in to the air; thus it can be classified as being extremely persistent in the environment if any possible effects of external factors like sun light and water (air humidity) upon it are neglected. Various salts other than bromide have been reported.[1]
Synthesis
Two methods have been described for synthesizing EA-3990 along with similar nerve agents.
The 2-dimethylaminomethyl-3-dimethylcarbamoxypyridine precursor is prepared via a
In the first method 2
In the second method 2 mol and 1 mol of the previous reagents used in the first method are added together, but also a catalytic amount of sodium iodide in acetonitrile is added to the solution, which is then allowed to stand for 6 days. Crystalline material is usually formed during this period and it is then collected by filtration.[3]
In both methods, after filtration, the crystalline product is triturated with acetone. If no solid separates, ethyl acetate is added to precipitate the crude product. The product is then dissolved in hot ethanol and treated with decolorizing charcoal. Ethyl acetate is added to the filtered solution to precipitate the crystalline product. E-3990 is then collected and dried. Yield is 63%.[3][6]
See also
References
- ^ OCLC 82473582.
- PMID 24179466.
- ^ a b c d e f US patent 04512246, Harold Z. Sommer, Havre De Grace, John Krenzer, Oak Park, Omer O. Owens, Jacob I. Miller, "Chemical agents", issued 1987-06-30, assigned to US Secretary of Army
- OCLC 433545336.
- ^ FAS Staff (2013). "Types of Chemical Weapons: Nerve Agents [Table. Toxicological Data]". Washington, DC: Federation of American Scientists [FAS]. Archived from the original on November 26, 2016. Retrieved March 20, 2018.
- ^ a b c US patent 4677204A, Harold Z. Sommer, Havre de Grace, Omer O. Owens, "Chemical agents", issued 1987-06-30, assigned to US Secretary of Army
