Dinitrogen trioxide
| |

| |
| Names | |
|---|---|
| IUPAC name
N-Oxonitramide[1]
| |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.031.013 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 2421 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| N2O3 | |
| Molar mass | 76.011 g·mol−1 |
| Appearance | Deep blue liquid |
| Density |
|
| Melting point | −100.7[2] °C (−149.3 °F; 172.5 K) |
| Boiling point | 3.5 °C (38.3 °F; 276.6 K) (dissociates[2]) |
| reacts to form nitrous acid | |
| Solubility | soluble in ether |
| −16.0·10−6 cm3/mol | |
| Structure | |
| planar, Cs | |
| 2.122 D | |
| Thermochemistry | |
Heat capacity (C)
|
65.3 J/(mol·K) |
Std molar
entropy (S⦵298) |
314.63 J/(mol·K) |
Std enthalpy of (ΔfH⦵298)formation |
91.20 kJ/mol |
| Hazards | |
| GHS labelling:[3] | |
   
| |
| Danger | |
| H270, H280, H310, H310+H330, H314, H330 | |
| P220, P244, P260, P262, P264, P270, P271, P280, P284, P301+P330+P331, P302+P350, P303+P361+P353, P304+P340, P305+P351+P338, P310, P320, P321, P322, P361, P363, P370+P376, P403, P403+P233, P405, P410+P403, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Dinitrogen trioxide (also known as nitrous anhydride) is the inorganic compound with the formula N2O3. It is a nitrogen oxide. It forms upon mixing equal parts of nitric oxide and nitrogen dioxide and cooling the mixture below −21 °C (−6 °F):[4]
- •
NO + •
NO
2 ⇌ N
2O
3
Dinitrogen trioxide is only isolable at low temperatures, i.e. in the liquid and solid phases. In liquid and solid states, it has a deep blue color.[2] At higher temperatures the equilibrium favors the constituent gases, with KD = 193 kPa (25 °C).[5][clarification needed]
This compound is sometimes called "nitrogen trioxide", but this name properly refers to another compound, the (uncharged)
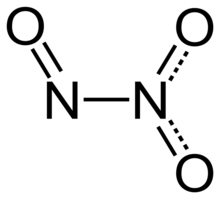
Structure and bonding
Dinitrogen trioxide molecule contains an
If the nitrous acid is not then used up quickly, it decomposes into nitric oxide and nitric acid. Nitrite salts are sometimes produced by adding N2O3 to water solutions of bases:
- N2O3 + 2 NaOH → 2 NaNO2 + H2O
Typically, N–N bonds are similar in length to that in
Similar to
References
- ^ "Dinitrogen trioxide".
- ^ ISBN 978-0-08-037941-8.
- ^ "Dinitrogen trioxide". pubchem.ncbi.nlm.nih.gov. Retrieved 23 December 2021.
- ^ ISBN 978-0-08-022057-4.
- ISBN 0-12-352651-5
- PMID 35653176.
- ISSN 1573-8779.


