Enriched uranium
Enriched uranium is a type of
Enriched uranium is a critical component for both civil nuclear power generation and military nuclear weapons. The International Atomic Energy Agency attempts to monitor and control enriched uranium supplies and processes in its efforts to ensure nuclear power generation safety and curb nuclear weapons proliferation.
There are about 2,000 tonnes of highly enriched uranium in the world,[2] produced mostly for nuclear power, nuclear weapons, naval propulsion, and smaller quantities for research reactors.
The 238U remaining after enrichment is known as
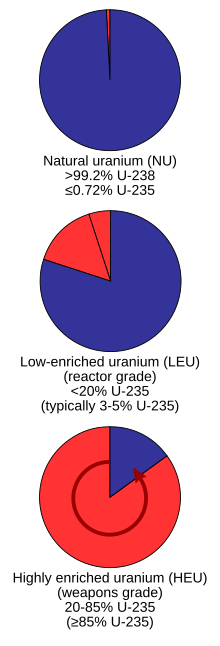
Grades
Uranium as it is taken directly from the Earth is not suitable as fuel for most nuclear reactors and requires additional processes to make it usable (
This is accomplished by a combination of chemical processes with the end product being concentrated uranium oxide, which is known as "yellowcake", contains roughly 80% uranium whereas the original ore typically contains as little as 0.1% uranium.[3]
After the milling process is complete, the uranium must next undergo a process of conversion, "to either

Reprocessed uranium (RepU)
Reprocessed uranium (RepU) is a product of
Low-enriched uranium (LEU)
Low-enriched uranium (LEU) has a lower than 20% concentration of 235U; for instance, in commercial LWR, the most prevalent power reactors in the world, uranium is enriched to 3 to 5% 235U. Slightly enriched uranium (SEU) has a concentration of under 2% 235U.[5]
High-assay LEU (HALEU)
High-assay LEU (HALEU) is enriched between 5% and 20%[6] and is called for in many small modular reactor (SMR) designs. Fresh LEU used in research reactors is usually enriched between 12% and 19.75% 235U; the latter concentration is used to replace HEU fuels when converting to LEU.[7]
Highly enriched uranium (HEU)
Highly enriched uranium (HEU) has a 20% or higher concentration of 235U. The fissile uranium in
The very first uranium bomb,
Later U.S. nuclear weapons usually use
HEU is also used in
Enrichment methods
Isotope separation is difficult because two isotopes of the same element have nearly identical chemical properties, and can only be separated gradually using small mass differences. (235U is only 1.26% lighter than 238U.) This problem is compounded because uranium is rarely separated in its atomic form, but instead as a compound (235UF6 is only 0.852% lighter than 238UF6). A cascade of identical stages produces successively higher concentrations of 235U. Each stage passes a slightly more concentrated product to the next stage and returns a slightly less concentrated residue to the previous stage.
There are currently two generic commercial methods employed internationally for enrichment: gaseous diffusion (referred to as first generation) and gas centrifuge (second generation), which consumes only 2% to 2.5%[13] as much energy as gaseous diffusion. Some work is being done that would use nuclear resonance; however, there is no reliable evidence that any nuclear resonance processes have been scaled up to production.
Diffusion techniques
Gaseous diffusion

Gaseous diffusion is a technology used to produce enriched uranium by forcing gaseous
Thermal diffusion
Thermal diffusion uses the transfer of heat across a thin liquid or gas to accomplish isotope separation. The process exploits the fact that the lighter 235U gas molecules will diffuse toward a hot surface, and the heavier 238U gas molecules will diffuse toward a cold surface. The S-50 plant at Oak Ridge, Tennessee, was used during World War II to prepare feed material for the Electromagnetic isotope separation (EMIS) process, explained later in this article. It was abandoned in favor of gaseous diffusion.
Centrifuge techniques
Gas centrifuge

The gas centrifuge process uses a large number of rotating cylinders in series and parallel formations. Each cylinder's rotation creates a strong
Zippe centrifuge

The Zippe-type centrifuge is an improvement on the standard gas centrifuge, the primary difference being the use of heat. The bottom of the rotating cylinder is heated, producing convection currents that move the 235U up the cylinder, where it can be collected by scoops. This improved centrifuge design is used commercially by Urenco to produce nuclear fuel and was used by Pakistan in their nuclear weapons program.
Laser techniques
Laser processes promise lower energy inputs, lower capital costs and lower tails assays, hence significant economic advantages. Several laser processes have been investigated or are under development. Separation of isotopes by laser excitation (SILEX) is well developed and is licensed for commercial operation as of 2012. Separation of isotopes by laser excitation is a very effective and cheap method of uranium separation, able to be done in small facilities requiring much less energy and space than previous separation techniques. The cost of uranium enrichment using laser enrichment technologies is approximately $30 per SWU[17] which is less than a third of the price of gas centrifuges, the current standard of enrichment. Separation of isotopes by laser excitation could be done in facilities virtually undetectable by satellites.[18] More than 20 countries have worked with laser separation over the past two decades, the most notable of these countries being Iran and North Korea, though all countries have had very limited success up to this point.
Atomic vapor laser isotope separation (AVLIS)
Molecular laser isotope separation (MLIS)
Molecular laser isotope separation uses an infrared laser directed at UF6, exciting molecules that contain a 235U atom. A second laser frees a fluorine atom, leaving uranium pentafluoride, which then precipitates out of the gas.
Separation of isotopes by laser excitation (SILEX)
Other techniques
Aerodynamic processes


Aerodynamic enrichment processes include the Becker jet nozzle techniques developed by E. W. Becker and associates using the
Electromagnetic isotope separation

In the
Chemical methods
One chemical process has been demonstrated to pilot plant stage but not used for production. The French CHEMEX process exploited a very slight difference in the two isotopes' propensity to change
Plasma separation
Plasma separation process (PSP) describes a technique that makes use of
Separative work unit
"Separative work"—the amount of separation done by an enrichment process—is a function of the concentrations of the feedstock, the enriched output, and the depleted tailings; and is expressed in units that are so calculated as to be proportional to the total input (energy / machine operation time) and to the mass processed. Separative work is not energy. The same amount of separative work will require different amounts of energy depending on the efficiency of the separation technology. Separative work is measured in Separative work units SWU, kg SW, or kg UTA (from the German Urantrennarbeit – literally uranium separation work).
- 1 SWU = 1 kg SW = 1 kg UTA
- 1 kSWU = 1 tSW = 1 t UTA
- 1 MSWU = 1 ktSW = 1 kt UTA
Cost issues
In addition to the separative work units provided by an enrichment facility, the other important parameter to be considered is the mass of natural uranium (NU) that is needed to yield a desired mass of enriched uranium. As with the number of SWUs, the amount of feed material required will also depend on the level of enrichment desired and upon the amount of 235U that ends up in the depleted uranium. However, unlike the number of SWUs required during enrichment, which increases with decreasing levels of 235U in the depleted stream, the amount of NU needed will decrease with decreasing levels of 235U that end up in the DU.
For example, in the enrichment of LEU for use in a light water reactor it is typical for the enriched stream to contain 3.6% 235U (as compared to 0.7% in NU) while the depleted stream contains 0.2% to 0.3% 235U. In order to produce one kilogram of this LEU it would require approximately 8 kilograms of NU and 4.5 SWU if the DU stream was allowed to have 0.3% 235U. On the other hand, if the depleted stream had only 0.2% 235U, then it would require just 6.7 kilograms of NU, but nearly 5.7 SWU of enrichment. Because the amount of NU required and the number of SWUs required during enrichment change in opposite directions, if NU is cheap and enrichment services are more expensive, then the operators will typically choose to allow more 235U to be left in the DU stream whereas if NU is more expensive and enrichment is less so, then they would choose the opposite.
When converting uranium (hexafluoride, hex for short) to metal, 0.3% is lost during manufacturing.[26][27]
Downblending
The opposite of enriching is downblending; surplus HEU can be downblended to LEU to make it suitable for use in commercial nuclear fuel.
The HEU feedstock can contain unwanted uranium isotopes:
The blendstock can be NU or DU; however, depending on feedstock quality, SEU at typically 1.5 wt% 235U may be used as a blendstock to dilute the unwanted byproducts that may be contained in the HEU feed. Concentrations of these isotopes in the LEU product in some cases could exceed
A major downblending undertaking called the Megatons to Megawatts Program converts ex-Soviet weapons-grade HEU to fuel for U.S. commercial power reactors. From 1995 through mid-2005, 250 tonnes of high-enriched uranium (enough for 10,000 warheads) was recycled into low-enriched uranium. The goal is to recycle 500 tonnes by 2013. The decommissioning programme of Russian nuclear warheads accounted for about 13% of total world requirement for enriched uranium leading up to 2008.[14]
The
Global enrichment facilities
The following countries are known to operate enrichment facilities: Argentina, Brazil, China, France, Germany, India, Iran, Japan, the Netherlands, North Korea, Pakistan, Russia, the United Kingdom, and the United States.
Codename
During the Manhattan Project, weapons-grade highly enriched uranium was given the codename oralloy, a shortened version of Oak Ridge alloy, after the location of the plants where the uranium was enriched.[36] The term oralloy is still occasionally used to refer to enriched uranium.
See also
References
- ISBN 9789264103283.
- ^ Cochran (Natural Resources Defense Council), Thomas B. (12 June 1997). "Safeguarding Nuclear Weapon-Usable Materials in Russia" (PDF). Proceedings of international forum on illegal nuclear traffic. Archived from the original (PDF) on 22 July 2012.
- ^ Nuclear Fuel Cycle Overview, Uranium milling. World Nuclear Association, update April 2021
- ^ "Radiological Sources of Potential Exposure and/or Contamination". U.S. Army Center for Health Promotion and Preventive Medicine. June 1999. p. 27. Retrieved 1 July 2019.
- S2CID 225435681.
- ^ Herczeg, John W. (28 March 2019). "High-assay low enriched uranium" (PDF). energy.gov. Archived (PDF) from the original on 9 October 2022.
- ^ Glaser, Alexander (6 November 2005). About the Enrichment Limit for Research Reactor Conversion : Why 20%? (PDF). The 27th International Meeting on Reduced Enrichment for Research and Test Reactors (RERTR. Princeton University. Archived (PDF) from the original on 9 October 2022. Retrieved 18 April 2014.
- ^ a b Forsberg, C. W.; Hopper, C. M.; Richter, J. L.; Vantine, H. C. (March 1998). "Definition of Weapons-Usable Uranium-233" (PDF). ORNL/TM-13517. Oak Ridge National Laboratories. Archived from the original (PDF) on 2 November 2013. Retrieved 30 October 2013.
- ^ Sublette, Carey (4 October 1996). "Nuclear Weapons FAQ, Section 4.1.7.1: Nuclear Design Principles – Highly Enriched Uranium". Nuclear Weapons FAQ. Retrieved 2 October 2010.
- doi:10.2172/10120434. Archived(PDF) from the original on 9 October 2022. Retrieved 19 December 2007.
The enrichment of the pin and of one of the hemispheres was 97.67 w/o, while the enrichment of the other hemisphere was 97.68 w/o.
- ^ "Nuclear Weapons FAQ". Retrieved 26 January 2013.
- S2CID 122507063.
- ^ "Uranium Enrichment". world-nuclear.org. Archived from the original on 1 July 2013. Retrieved 14 April 2013.
- ^ a b c d "Lodge Partners Mid-Cap Conference 11 April 2008" (PDF). Silex Ltd. 11 April 2008. Archived (PDF) from the original on 9 October 2022.
- ^ Adams, Rod (24 May 2011). "McConnell asks DOE to keep using 60-year-old enrichment plant to save jobs". Atomic Insights. Archived from the original on 28 January 2013. Retrieved 26 January 2013.
- ^ "Paducah enrichment plant to be closed - World Nuclear News". www.world-nuclear-news.org.
- ^ S2CID 100862135.
- ^ ProQuest 204555310.
- ^ F. J. Duarte and L. W. Hillman (Eds.), Dye Laser Principles (Academic, New York, 1990) Chapter 9.
- ^ "GE Signs Agreement With Silex Systems of Australia To Develop Uranium Enrichment Technology" (Press release). GE Energy. 22 May 2006. Archived from the original on 14 June 2006.
- ^ "GE Hitachi Nuclear Energy Selects Wilmington, N.C. as Site for Potential Commercial Uranium Enrichment Facility". Business Wire. 30 April 2008. Retrieved 30 September 2012.
- ^ Broad, William J. (20 August 2011). "Laser Advances in Nuclear Fuel Stir Terror Fear". The New York Times. Retrieved 21 August 2011.
- ^ Associated Press (27 September 2012). "Uranium Plant Using Laser Technology Wins U.S. Approval". The New York Times.
- S2CID 44245091.
- ^ Smith, Michael; Jackson A G M (2000). "Dr". South African Institution of Chemical Engineers – Conference 2000: 280–289.
- ^ Balakrishnan, M. R. (1971). "Economics of blending, a case study" (PDF). Bombay, India: Government of India, Atomic Energy Commission. p. 6. Archived (PDF) from the original on 9 October 2022. Retrieved 7 November 2021.
- ^ US Atomic Energy Commission (January 1961). "Costs of nuclear power". Washington DC: Office of Technical Services, Dept of Commerce. p. 29. Retrieved 7 November 2021.
- ^ "Status Report: USEC-DOE Megatons to Megawatts Program". USEC.com. 1 May 2000. Archived from the original on 6 April 2001.
- ^ "Megatons to Megawatts". centrusenergy.com. December 2013.
- ^ Makhijani, Arjun; Chalmers, Lois; Smith, Brice (15 October 2004). Uranium enrichment (PDF). Institute for Energy and Environmental Research. Archived (PDF) from the original on 9 October 2022. Retrieved 21 November 2009.
- ^ Australia's uranium - Greenhouse friendly fuel for an energy hungry world (PDF). Standing Committee on Industry and Resources (Report). The Parliament of the Commonwealth of Australia. November 2006. p. 730. Archived (PDF) from the original on 9 October 2022. Retrieved 3 April 2015.
- ^ "Q&A: Uranium enrichment". BBC News. BBC. 1 September 2006. Retrieved 3 January 2010.
- ^ "Laser enrichment could cut cost of nuclear power". The Sydney Morning Herald. 26 May 2006.
- S2CID 100862135.
- ^ "Israel's Nuclear Weapons Program". Nuclear Weapon Archive. 10 December 1997. Retrieved 7 October 2007.
- ^ Burr, William (22 December 2015). "Strategic Air Command Declassifies Nuclear Target List from 1950s". nsarchive2.gwu.edu. Retrieved 27 November 2020.
Oralloy [Oak Ridge alloy] was a term of art for highly enriched uranium.
External links
- Annotated bibliography on enriched uranium from the Alsos Digital Library for Nuclear Issues
- Silex Systems Ltd
- Uranium Enrichment Archived 2 December 2010 at the Wayback Machine, World Nuclear Association
- Overview and history of U.S. HEU production
- News Resource on Uranium Enrichment
- Nuclear Chemistry-Uranium Enrichment Archived 15 October 2008 at the Wayback Machine
- A busy year for SWU (a 2008 review of the commercial enrichment marketplace), Nuclear Engineering International, 1 September 2008
- Uranium Enrichment and Nuclear Weapon Proliferation, by Allan S. Krass, Peter Boskma, Boelie Elzen and Wim A. Smit, 296 pp., published for SIPRI by Taylor and Francis Ltd, London, 1983
- The Periodic Table of Videos. University of Nottingham.
- Gilinsky, V.; Hoehn, W. (December 1969). "The Military Significance of Small Uranium Enrichment Facilities Fed with Low-Enrichment Uranium (Redacted)". DTIC ADA613260.
