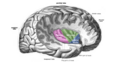
Insular cortex
| Insular cortex | |
|---|---|
 Right insula, exposed by removing the opercula | |
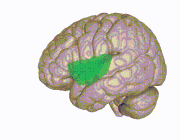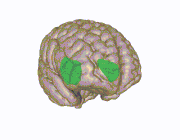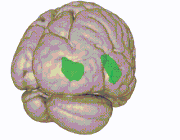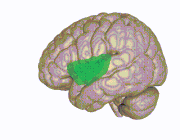 3D view of the insular cortex | |
| Details | |
| Part of | cerebral cortex of brain |
| Artery | Middle cerebral |
| Identifiers | |
| Latin | cortex insularis |
| MeSH | D000087623 |
| NeuroNames | 111 |
| NeuroLex ID | birnlex_1117 |
| TA98 | A14.1.09.149 A12.2.07.053 |
| TA2 | 5502 |
| FMA | 67329 |
| Anatomical terms of neuroanatomy | |
The insular cortex (also insula and insular lobe) is a portion of the
.The insulae are believed to be involved in
The insular cortex is divided into two parts: the anterior insula and the posterior insula in which more than a dozen field areas have been identified. The cortical area overlying the insula toward the lateral surface of the brain is the operculum (meaning lid). The opercula are formed from parts of the enclosing frontal, temporal, and parietal lobes.
Structure
Connections
The anterior part of the insula is subdivided by shallow sulci into three or four short gyri.
The anterior insula receives a direct projection from the basal part of the ventral medial nucleus of the thalamus and a particularly large input from the central nucleus of the amygdala. In addition, the anterior insula itself projects to the amygdala.
One study on rhesus monkeys revealed widespread reciprocal connections between the insular cortex and almost all subnuclei of the amygdaloid complex. The posterior insula projects predominantly to the dorsal aspect of the lateral and to the central amygdaloid nuclei. In contrast, the anterior insula projects to the anterior amygdaloid area as well as the medial, the cortical, the accessory basal magnocellular, the medial basal, and the lateral amygdaloid nuclei.[1]
The posterior part of the insula is formed by a long gyrus.
The posterior insula connects reciprocally with the secondary somatosensory cortex and receives input from spinothalamically activated ventral posterior inferior thalamic nuclei. It has also been shown that this region receives inputs from the ventromedial nucleus (posterior part) of the thalamus that are highly specialized to convey homeostatic information such as pain, temperature, itch, local oxygen status, and sensual touch.[2]
A human neuroimaging study using diffusion tensor imaging revealed that the anterior insula is interconnected to regions in the temporal and occipital lobe, opercular and orbitofrontal cortex, triangular and opercular parts of the inferior frontal gyrus. The same study revealed differences in the anatomical connection patterns between the left and right hemisphere.[3]
The 'circular sulcus of insula' (or sulcus of Reil[4]) is a semi-circular sulcus or fissure[4] that separates the insula from the neighboring gyri of the operculum[5] in the front, above, and behind.[4]
Cytoarchitecture
The insular cortex has regions of variable cell structure or
Development
The insular cortex is considered a separate
Function
Multimodal sensory processing, sensory binding
Functional imaging studies show activation of the insula during audio-visual integration tasks.[9][10]
Taste
The anterior insula is part of the primary gustatory cortex.[11][12] Research in rhesus monkeys has also reported that apart from numerous taste-sensitive neurons, the insular cortex also responds to non-taste properties of oral stimuli related to the texture (viscosity, grittiness) or temperature of food.[13]
Speech
The sensory speech region, Wernicke’s area, and the motor speech region, Broca’s area, are interconnected by a large axonal fiber system known as the arcuate fasciculus which passes directly beneath the insular cortex. On account of this anatomical architecture, ischemic strokes in the insular region can disrupt the arcuate fasciculus.[14] Functional imaging studies on the cerebral correlates of language production also suggest that the anterior insula forms part of the brain network of speech motor control.[15] Moreover, electrical stimulation of the posterior insular can evoke speech disturbances such as speech arrest and reduced voice intensity.[16]
Lesion of the pre-central gyrus of the insula can also cause “pure speech apraxia” (i.e. the inability to speak with no apparent aphasic or orofacial motor impairments).[17] This demonstrates that the insular cortex forms part of a critical circuit for the coordination of complex articulatory movements prior to and during the execution of the motor speech plans.[17] Importantly, this specific cortical circuit is different from those that relate to the cognitive aspects of language production (e.g., Broca’s area on the inferior frontal gyrus).[17] Subvocal, or silent, speech has also been shown to activate right insular cortex, further supporting the theory that the motor control of speech proceeds from the insula.[18]
Interoceptive awareness
There is evidence that, in addition to its base functions, the insula may play a role in certain higher-level functions that operate only in humans and other
The insular cortex also is where the sensation of
Physiological studies in rhesus monkeys have shown that neurons in the insula respond to skin stimulation.[30] PET studies have also revealed that the human insula can also be activated by vibrational stimulation to the skin.[31]
Another perception of the right anterior insula is the degree of nonpainful
One brain imaging study suggests that the unpleasantness of subjectively perceived
The cerebral cortex processing
Other noninteroceptive perceptions include passive listening to music,[40] laughter and crying,[41] empathy and compassion,[42] and language.[43]
Motor control
In motor control, it contributes to hand-and-eye motor movement,[44][45] swallowing,[46] gastric motility,[47] and speech articulation.[48][49] It has been identified as a "central command” centre that ensures that heart rate and blood pressure increase at the onset of exercise.[50] Research upon conversation links it to the capacity for long and complex spoken sentences.[51] It is also involved in motor learning[52] and has been identified as playing a role in the motor recovery from stroke.[53]
Homeostasis
It plays a role in a variety of homeostatic functions related to basic survival needs, such as taste, visceral sensation, and autonomic control. The insula controls autonomic functions through the regulation of the sympathetic and parasympathetic systems.[54][55] It has a role in regulating the immune system.[56][57][58]
Self
The insula has been identified as playing a role in the experience of bodily self-awareness,[59][60] sense of agency,[61] and sense of body ownership.[62]
Social emotions
The anterior insula processes a person's sense of disgust both to smells[63] and to the sight of contamination and mutilation[64] — even when just imagining the experience.[65] This associates with a mirror neuron-like link between external and internal experiences.
In social experience, it is involved in the processing of norm violations,[66] emotional processing,[67] empathy,[68] and orgasms.[69]
The insula is active during social decision making. Tiziana Quarto et al. measured emotional intelligence (EI) (the ability to identify, regulate, and process emotions of themselves and of others) of sixty-three healthy subjects. Using fMRI EI was measured in correlation with left insular activity. The subjects were shown various pictures of facial expressions and tasked with deciding to approach or avoid the person in the picture. The results of the social decision task yielded that individuals with high EI scores had left insular activation when processing fearful faces. Individuals with low EI scores had left insular activation when processing angry faces.[70]
Emotions
The insular cortex, in particular its most anterior portion, is considered a
In terms of function, the insula is believed to process convergent information to produce an emotionally relevant context for sensory experience. To be specific, the anterior insula is related more to olfactory, gustatory, viscero-autonomic, and limbic function, whereas the posterior insula is related more to auditory-somesthetic-skeletomotor function.
The anterior insular cortex (AIC) is believed to be responsible for emotional feelings, including maternal and romantic love, anger, fear, sadness, happiness, sexual arousal, disgust, aversion, unfairness, inequity, indignation, uncertainty,[72] disbelief, social exclusion, trust, empathy, sculptural beauty, a ‘state of union with God’, and hallucinogenic states.[73]
Functional imaging studies have also implicated the insula in conscious desires, such as food craving and drug craving. What is common to all of these emotional states is that they each change the body in some way and are associated with highly salient subjective qualities. The insula is well-situated for the integration of information relating to bodily states into higher-order cognitive and emotional processes. The insula receives information from "homeostatic afferent" sensory pathways via the thalamus and sends output to a number of other limbic-related structures, such as the
A study using
Another study using voxel-based morphometry and MRI on experienced
The strongest evidence against a causative role for the insula cortex in emotion comes from Damasio et al. (2012) [78] which showed that a patient who suffered bilateral lesions of the insula cortex expressed the full complement of human emotions, and was fully capable of emotional learning.
Salience
Functional neuroimaging research suggests the insula is involved in two types of salience. Interoceptive information processing that links interoception with emotional salience to generate a subjective representation of the body. This involves, first, the anterior insular cortex with the pregenual anterior cingulate cortex (Brodmann area 33) and the anterior and posterior mid-cingulate cortices, and, second, a general salience network concerned with environmental monitoring, response selection, and skeletomotor body orientation that involves all of the insular cortex and the mid-cingulate cortex.[79] A related idea is that the anterior insula, as part of the salience network, interacts with the mid-posterior insula to combine salient stimuli with autonomic information, leading to a high state of physiological awareness of salient stimuli.[80]
An alternative or perhaps complementary proposal is that the right anterior insular regulates the interaction between the salience of the
Decision Making
Studies have shown that damage or dysfunction in the insular cortex can impair decision-making, emotional regulation, and social behavior. The insula is considered a key brain structure in the neural circuitry underlying complex decision-making processes. [82] It plays a significant role in integrating internal and external cues to facilitate adaptive choices.
Auditory perception
Recent research indicates that the insular cortex is involved in
Direct recordings from the posterior part of the insula showed responses to unexpected sounds within regular auditory streams, a process known as auditory deviance detection. Researchers observed a mismatch negativity (MMN) potential, a well known event related potential, as well as the high frequency activity signals originating from local neurons.[86]
Simple auditory illusions and hallucinations were elicited by electrical functional mapping.[87][83]
Clinical significance
Progressive expressive aphasia
Progressive
Addiction
A number of functional brain imaging studies have shown that the insular cortex is activated when drug users are exposed to environmental cues that trigger cravings. This has been shown for a variety of drugs, including
A recent study in rats by Contreras et al.[95] corroborates these findings by showing that reversible inactivation of the insula disrupts amphetamine conditioned place preference, an animal model of cue-induced drug craving. In this study, insula inactivation also disrupted "malaise" responses to lithium chloride injection, suggesting that the representation of negative interoceptive states by the insula plays a role in addiction. However, in this same study, the conditioned place preference took place immediately after the injection of amphetamine, suggesting that it is the immediate, pleasurable interoceptive effects of amphetamine administration, rather than the delayed, aversive effects of amphetamine withdrawal that are represented within the insula.
A model proposed by Naqvi et al. (see above) is that the insula stores a representation of the pleasurable interoceptive effects of drug use (e.g., the airway sensory effects of nicotine, the cardiovascular effects of amphetamine), and that this representation is activated by exposure to cues that have previously been associated with drug use. A number of functional imaging studies have shown the insula to be activated during the administration of addictive psychoactive drugs. Several functional imaging studies have also shown that the insula is activated when drug users are exposed to drug cues, and that this activity is correlated with subjective urges. In the cue-exposure studies, insula activity is elicited when there is no actual change in the level of drug in the body. Therefore, rather than merely representing the interoceptive effects of drug use as it occurs, the insula may play a role in memory for the pleasurable interoceptive effects of past drug use, anticipation of these effects in the future, or both. Such a representation may give rise to conscious urges that feel as if they arise from within the body. This may make addicts feel as if their bodies need to use a drug, and may result in persons with lesions in the insula reporting that their bodies have forgotten the urge to use, according to this study.
Subjective certainty in ecstatic seizures
A common quality in mystical experiences is a strong feeling of certainty which cannot be expressed in words. Fabienne Picard proposes a neurological explanation for this subjective certainty, based on clinical research of epilepsy.[96][97] According to Picard, this feeling of certainty may be caused by a dysfunction of the anterior insula, a part of the brain which is involved in
Other clinical conditions
The insular cortex has been suggested to have a role in anxiety disorders,[102] emotion dysregulation,[103] and anorexia nervosa.[104]
History
The insula was first described by
Additional images
-
Location and structure of the insular cortex
-
Coronal section of brain immediately in front of pons (Insula labeled at upper right)
-
Horizontal section of left cerebral hemisphere
-
Insular cortex highlighted in green on coronal T1 MRI images
-
Insular cortex highlighted in green on sagittal T1 MRI images
-
Insular cortex highlighted in green on transversal T1 MRI images
See also
References
- S2CID 46366616.
- S2CID 7077496.
- S2CID 12293575.
- ^ a b c Johannes Sobotta. "Sobotta's Atlas and Text-book of human anatomy 1909". p. 145. Retrieved November 10, 2013.
- ^ "Definition: 'Circular Sulcus Of Insula'". MediLexicon. Archived from the original on 2013-06-04. Retrieved 2012-03-30.
- PMID 23466178.
- ^ Brain, MSN Encarta. Archived 2009-10-31.
- ISBN 978-0-7167-5300-1.
- PMID 11150347.
- S2CID 1098979.
- ISBN 978-0-8053-0094-9.
- PMID 10495075.
- PMID 15331650.
- ^ https://academic.oup.com/brain/article-abstract/103/2/337/378338. Retrieved 2023-12-11.
{{cite web}}: Missing or empty|title=(help) - S2CID 9909543.
- PMID 20946128.
- ^ a b c https://academic.oup.com/cercor/article/31/8/3723/6213947. Retrieved 2023-12-11.
{{cite web}}: Missing or empty|title=(help) - S2CID 2040545.
- PMID 16888142.
- PMID 20045470.
- PMID 21750565.
- S2CID 13344271.
- ^ PMID 17414805.
- S2CID 8653997.
- S2CID 1078691.
- PMID 19073802.
- PMID 16275823.
- PMID 16855007.
- S2CID 21437784.
- S2CID 20207990.
- PMID 8237217.
- S2CID 20068852.
- S2CID 7077496.
- PMID 11159877.
- ]
- PMID 12394703.
- PMID 18263796.[permanent dead link]
- S2CID 42990194.
- PMID 16918202.
- S2CID 12308683.
- S2CID 9509954.
- ^ "Interview with Tania Singer | the Center for Compassion and Altruism Research and Education". Archived from the original on 2010-07-14. Retrieved 2010-07-04.
- S2CID 22339177.
- PMID 7953589.
- S2CID 15881491.
- S2CID 15438676.
- PMID 13293263.
- S2CID 4305696.
- S2CID 36867434.
- PMID 15849712.
- PMID 17499317.
- PMID 17327919.
- S2CID 25131597.
- S2CID 32371468.
- S2CID 32616395.
- PMID 15745959.
- S2CID 24813018.
- S2CID 20527835.
- PMID 16079395.
- S2CID 2340032.
- S2CID 768408.
- PMID 17138596.
- S2CID 766157.
- S2CID 6864309.
- PMID 18698355.
- S2CID 7111382.
- S2CID 7150871.
- S2CID 15411628.
- S2CID 3377994.
- PMID 26859495.
- S2CID 7150871.
- PMID 22840519.
- S2CID 2340032. Archived from the original(PDF) on 2013-01-07.
- S2CID 19794544. Archived from the original(PDF) on 2010-06-22. Retrieved 2009-09-03.
- PMID 16272874.
- S2CID 207090878.
- PMID 19015095.
- PMID 22473895.
- S2CID 12917288.
- PMID 20512370.
- ^ PMID 19072895.
- .
- ^ S2CID 36927038.
- PMID 22230626, retrieved 2023-12-11
- S2CID 22339177.
- S2CID 202749677.
- S2CID 2506125.
- PMID 12902311.
- PMID 14991811.
- PMID 17255515.
- S2CID 8917168.
- PMID 22052507.
- PMID 24169814.
- PMID 16055762.
- S2CID 23499558.
- S2CID 206984751
- PMID 26924970
- ^ Picard 2013, p.2496-2498
- ^ Picard 2013, p.2497-2498
- ^ See also satori in Japanese Zen
- ^ a b Picard 2013, p.2498
- S2CID 17889111.
- PMID 11163422.
- S2CID 16526824.
- ^ S2CID 8152708.
External links
- Insular cortex in the Brede Database at the Technical University of Denmark. Location and literature citations for the insula
- synd/1212 at Who Named It?
- "Anatomy diagram: 13048.000-1". Roche Lexicon - illustrated navigator. Elsevier. Archived from the original on 2014-11-07.
- Stained brain slice images which include the "insular cortex" at the BrainMaps project
- Thomas P. Naidicha; et al. (1 February 2004). "The Insula: Anatomic Study and MR Imaging Display at 1.5 T". American Journal of Neuroradiology. 25 (2): 222–32. PMID 14970021.
- Kakigia R, Nakataa H, Inuia K, Hiroea N, et al. (October 2005). "Intracerebral pain processing in a Yoga Master who claims not to feel pain during meditation". Eur J Pain. 9 (5): 581–9. S2CID 17598717.
As for fMRI recording, there were remarkable changes in levels of activity in the...SII-insula (mainly the insula)