Thymidine kinase
| Thymidine kinase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Thymidine kinase | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||
| Symbol | TK | ||||||||||
| Pfam | PF00265 | ||||||||||
| Pfam clan | CL0023 | ||||||||||
| InterPro | IPR001267 | ||||||||||
| PROSITE | PDOC00524 | ||||||||||
| |||||||||||
Chr. 17 q23.2-25.3 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Chr. 16 [1] | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Thymidine kinase is an
- Thd + ATP → TMP + ADP
where Thd is (deoxy)thymidine, ATP is adenosine triphosphate, TMP is (deoxy)thymidine monophosphate and ADP is adenosine diphosphate. Thymidine kinases have a key function in the synthesis of
History
The incorporation of thymidine in DNA was demonstrated around 1950.[4] Somewhat later, it was shown that this incorporation was preceded by phosphorylation,[5] and, around 1960, the enzyme responsible was purified and characterized.[6][7]
Classification
Two different classes of thymidine kinases have been identified
Isozymes
Mammals have two
The viral thymidine kinases differ completely from the mammalian enzymes both structurally and biochemically and are inhibited by
Recombinant TK1 cannot be activated and converted to a tetramer in this way, showing that the enzyme occurring in cells has been modified after synthesis.[20][23][24]
TK1 is synthesized by the cell during the S phase of cell division. After cell division is completed, TK1 is
Genes for virus specific thymidine kinases have been identified in Herpes simplex virus, Varicella zoster virus and Epstein-Barr virus.[37][38][39][40][41][42][43]
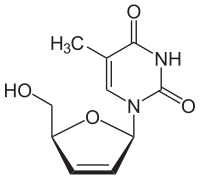
Thymidine reacts with ATP to give thymidine monophosphate and ADP.
Function
Thymidine monophosphate, the product of the reaction catalyzed by thymidine kinase, is in turn phosphorylated to
Thymidine monophosphate is also produced by the cell in a different reaction by
Deficiency
Thymidine kinase 2 is used by the cells for synthesis of mitochondrial DNA. Mutations in the gene for TK2 lead to a myopathic form of mitochondrial DNA depletion syndrome. Another reason for TK 2 deficiency may be oxidative stress induced S-glutathionylation and proteolytic degradation of mitochondrial thymidine kinase 2.[55] No syndrome caused by TK1 deficiency is known, probably as a defective TK1 gene would lead to fetal death.
Thymidine kinase during development
The formation of tetramer after modification of thymidine kinase 1 after synthesis enhances the enzyme activity. It has been suggested that this is a mechanism for regulation of the enzyme activity. The formation of tetramers is observed after the Dictyostelium development stage. Its use for fine regulation of DNA synthesis is suggested to have been established in warm blooded animals after they branched out from the vertebrates.[56] Also the development of thymidine kinase like enzymes in the development has been studied.[57]
Species distribution
Thymidine kinase is present in animals,
Applications
Identification of dividing cells
The first indirect use of thymidine kinase in biochemical research was the identification of dividing cells by incorporation of radiolabeled thymidine and subsequent measurement of the radioactivity or autoradiography to identify the dividing cells. For this purpose tritiated thymidine is included in the growth medium.[79] In spite of errors in the technique, it is still used to determine the growth rate of malignant cells and to study the activation of lymphocytes in immunology.
PET scan of active tumors
Selection of hybridomas
Study of chromosome structure
Molecular combing of DNA fibers can be used to monitor the structure of chromosomes in the budding yeast Saccharomyces cerevisiae. This provides DNA replication profiles of individual molecules. This requires that the yeast strains express thymidine kinase, which wild type yeasts do not, being fungi (see occurrence). Therefore, a gene for thymidine kinase must be incorporated in the genome.[94]
Clinical chemistry
Thymidine kinase is a salvage enzyme that is only present in anticipation of cell division. The enzyme is not set free from cells undergoing normal division where the cells have a special mechanism to degrade the proteins no longer needed after the cell division.[10] In normal subjects, the amount of thymidine kinase in serum or plasma is therefore very low. Tumor cells release enzyme to the circulation, probably in connection with the disruption of dead or dying tumor cells. The thymidine kinase level in serum therefore serves as a measure of malignant proliferation, indirectly as a measure of the aggressivity of the tumor.
Therapeutic applications
Some drugs are specifically directed against dividing cells. They can be used against tumors and viral diseases (both against retrovirus and against other virus), as the diseased cells replicate much more frequently than normal cells and also against some non-malignant diseases related to excessively rapid cell replication (for instance psoriasis). It has been suggested that the antiviral and anti-cancer activity of thymidine analogues is, at least partly, achieved by down-regulation of mitochondrial thymidine kinase.[95]
Cytostatics
There are different classes of drugs directed against thymidine metabolism and thereby involving thymidine kinase that are used to control cancer associated cell division.[96][97][98][99][100][101] Chain terminators are thymidine analogues that are included in the growing DNA chain, but modified so that the chain cannot be further elongated. As analogs of thymidine, this type of drugs are readily phosphorylated to 5'-monophosphates. The monophosphate is further phosphorylated to the corresponding triphosphate and incorporated in the growing DNA chain. The analog has been modified so that it does not have the hydroxyl group in the 3'-position that is required for continued chain growth. In zidovudine (AZT; ATC:J05AF01) the 3'-hydroxyl group has been replaced by an azido group,[36][100] in stavudine (ATC: J05AF04) it has been removed without replacement.[102][103] AZT is used as substrate in one of the methods for determination of thymidine kinase in serum.[104] This implies that AZT interferes with this method and may be a limitation: AZT is a standard component of HAART therapy in HIV infection. One common consequence of AIDS is lymphoma and the most important diagnostic application of thymidine kinase determination is for monitoring of lymphoma.
Other thymidine analogues, for instance Idoxuridine (ATC: J05AB02) act by blocking base pairing during subsequent replication cycles, thereby making the resulting DNA chain defective.[105] This may also be combined with radioactivity to achieve apoptosis of malignant cells.[106]
Antivirals
Some antiviral drugs, such as acyclovir (ATC: J05AB01) and ganciclovir (ATC: J05AB06) as well as other nucleoside analogs make use of the substrate specificity of viral thymidine kinase, as opposed to human thymidine kinases.[15] These drugs act as pro-drugs, which in themselves are not toxic, but are converted to toxic drugs by phosphorylation by viral thymidine kinase. Cells infected with the virus therefore produce highly toxic triphosphates that lead to cell death. Human thymidine kinase, in contrast, with its more narrow specificity, is unable to phosphorylate and activate the prodrug. In this way, only cells infected by the virus are susceptible to the drug. Such drugs are effective only against viruses from the herpes group with their specific thymidine kinase.[107][108] In patients treated with this type of drugs, the development of antiviral drug resistance is frequently observed. Sequencing the thymidine kinase gene in Herpes simplex virus and Varicella zoster virus shows the rapid genetic variability and may facilitate the diagnosis of antiviral drug resistance.[16][75]
After smallpox was declared eradicated by WHO in December 1979, vaccination programs were terminated. A re-emergence of the disease either by accident or as a result of biological warfare would meet an unprotected population and could result in an epidemic that could be difficult to control. Mass vaccination to combat a smallpox epidemic could be challenging because the only approved smallpox vaccine, Vaccinia Virus, can have severe side effects. Nevertheless, some governments stockpile Smallpox vaccine to insure against the possibility. However, the development of specific and effective antiviral drugs is prioritized. One possible approach would be to use the specificity of the thymidine kinase of poxvirus for the purpose, in a similar way that it is used for drugs against herpesvirus. One difficulty is that the poxvirus thymidine kinase belongs to the same family of thymidine kinases as the human thymidine kinases and thereby is more similar chemically. The structure of poxvirus thymidine kinases has therefore been determined to find potential antiviral drugs.[70] The search has, however, not yet resulted in a usable antiviral drug against poxviruses.
As a "suicide gene" in gene therapy
The herpesvirus thymidine kinase gene has also been used as a "suicide gene" as a safety system in gene therapy experiments, allowing cells expressing the gene to be killed using ganciclovir. This is desirable in case the recombinant gene causes a mutation leading to uncontrolled cell growth (insertional mutagenesis). The cytotoxic products produced by these modified cells may diffuse to neighboring cells, rendering them similarly susceptible to ganciclovir, a phenomenon known as the "bystander effect." This approach has been used to treat cancer in animal models, and is advantageous in that the tumor may be killed with as few as 10% of malignant cells expressing the gene.[109][110][111][112][113][114][115][116][117][118][119][120][121][122] A similar system has been tried using tomato thymidine kinase and AZT.[123][124] In addition, thymidine kinase gene is used as a suicide gene to tackle dangerous graft-versus-host disease in hematopoietic stem cell transplant therapy named Zalmoxis that was conditionally approved in Europe in 2016[125]
Tumor marker genes
A similar use of the thymidine kinase makes use of the presence in some tumor cells of substances not present in normal cells (
Neutron capture therapy for tumors
Incorporation of a thymidine analogue with boron has been suggested and tried in animal models for boron neutron capture therapy of brain tumors. A very extensive number of thymidine derivatives containing boron have been described.[132][133][134][135][136][137][138][139][140][141][142][143][144][145][146][147][148]
Antiparasitics
Introduction of a TK gene in a parasite genome makes it possible to incorporate BrdU and thereby makes the parasite sensitive to treatment with this drug has also been suggested and constitutes a sensitive indicator of replication of the parasite genome.[149]
Measurement
In serum and plasma
Thymidine kinase levels in serum or plasma have been mostly measured using enzyme activity assays. In commercial assays, this is done by incubation of a serum sample with a substrate analog and measurement of the amount of product formed.[71][72][73][104][150][151][152][153][154][155] Direct determination of the thymidine kinase protein by immunoassay has also been used.[156][157][158][159][160] The amounts of thymidine kinase found by this method does not correlate well with the enzyme activities. One reason for this is that a large amount of serum TK1 identified by immunoassay is not enzymatically active.[22][161] This is particularly the case with solid tumors where immunoassays may be more sensitive.[162][163]
In tissue
Thymidine kinase has been determined in tissue samples after extraction of the tissue. No standard method for the extraction or for the assay has been developed and TK determination in extracts from cells and tissues have not been validated in relation to any specific clinical question, see however Romain et al.[164] and Arnér et al.[165] A method has been developed for specific determination of TK2 in cell extracts using the substrate analog 5-Bromovinyl 2'-deoxyuridine.[166] In the studies referred to below the methods used and the way the results are reported are so different that comparisons between different studies are not possible. The TK1 levels in fetal tissues during development are higher than those of the corresponding tissues later.[167][168][169] Certain non-malignant diseases also give rise to dramatic elevation of TK values in cells and tissue: in peripheric lymphocytes during monocytosis[170] and in bone marrow during pernicious anemia.[171][172] As TK1 is present in cells during cell division, it is reasonable to assume that the TK activity in malignant tissue should be higher than in corresponding normal tissue. This is also confirmed in most studies.
Immunohistochemical staining
Antibodies against thymidine kinase are available for immunohistochemical detection.[173] Staining for thymidine kinase was found to be a reliable technique for identification of patients with stage 2 breast carcinoma. The highest number of patients identified was obtained by combination of thymidine kinase and Ki-67 staining.[174][175] The technique has also been validated for lung cancer,[174][176] for colorectal carcinoma,[177] for lung cancer[178] and for renal cell carcinoma.[179]
Fluorescent staining
2'-deoxy-2',2'-difluoro-5-ethynyluridine (dF-EdU) binds to Herpes simplex virus thymidine kinase but, because of sterical hindrance, not to human thymidine kinase. This reagent together with a fluorescent azide cause fluorescence of infected cells but not of uninfected cells. Therefore, this substrate analog makes it possible to specifically stain infected cells.[180]
See also
- Thymidine kinase 1
- Thymidine kinase from herpesvirus
- Thymidylate kinase
- Nucleoside-diphosphate kinase
- Thymidylate synthase
- Thymidine kinase in clinical chemistry
- DiviTum(R) TKa test for Thymidine kinase
References
- S2CID 84259415.
- PMID 3939993.
- PMID 9056888.
- PMID 14824173.
- PMID 13363894.
- PMID 13563524.
- PMID 13784139.
- ^ PMID 3027984.
- ^ PMID 2389555.
- ^ PMID 4223355.
- PMID 4457349.
- PMID 4735344.
- PMID 4632422.
- S2CID 21397238.
- ^ PMID 1707295.
- ^ PMID 26055375.
- S2CID 4207771.
- S2CID 22171412.
- PMID 3301530.
- ^ PMID 15611477.
- S2CID 21351513.
- ^ PMID 22741536.
- ^ PMID 7572355.
- PMID 14697231.
- S2CID 26971963.
- PMID 14046233.
- PMID 3000146.
- PMID 12741827.
- PMID 6727866.
- PMID 3812083.
- PMID 3731105.
- PMID 3355601.
- PMID 2536472.
- PMID 2706629.
- PMID 8016290.
- ^ PMID 25215937.
- ^ PMID 6258156.
- ^ S2CID 13276721.
- ^ PMID 6245273.
- ^ PMID 6317035.
- ^ PMID 3004022.
- ^ PMID 3019675.
- PMID 14033128.
- PMID 6940130.
- PMID 3670299.
- PMID 3561412.
- PMID 6260157.
- PMID 3669741.
- PMID 3372530.
- PMID 3596017.
- PMID 1708095.
- PMID 8676376.
- PMID 25152750.
- PMID 24561807.
- PMID 22661713.
- S2CID 37549615.
- S2CID 12250075.
- PMID 25744802.
- PMID 25293656.
- PMID 23859156.
- PMID 23628971.
- PMID 23102844.
- S2CID 9465188.
- PMID 22236202.
- S2CID 18112471.
- S2CID 35388801.
- PMID 25978379.
- ^ PMID 5729618.
- PMID 22462611.
- ^ PMID 2114104.
- ^ PMID 6260651.
- ^ S2CID 43720702.
- ^ PMID 6307593.
- PMID 6339548.
- ^ PMID 25712361.
- PMID 25916707.
- PMID 19563126.
- PMID 15157888.
- PMID 14407455.
- PMID 12839975.
- PMID 17178290.
- S2CID 10592042.
- S2CID 23952279.
- PMID 18628460.
- PMID 26778585.
- PMID 25737423.
- ^ "Methotrexate". PubChem. U.S. National Library of Medicine.
- ^ "Aminopterin". PubChem. U.S. National Library of Medicine.
- S2CID 4161444.
- S2CID 19360350.
- S2CID 27198746.
- S2CID 36092417.
- PMID 107455.
- PMID 26832684.
- PMID 24976398.
- PMID 1691243.
- PMID 177781.
- PMID 6996606.
- PMID 7001501.
- ^ PMID 25901475.
- PMID 25182642.
- PMID 3039911.
- PMID 3028398.
- ^ S2CID 18361311.
- PMID 13628760.
- S2CID 37766626.
- PMID 2983088.
- PMID 26125628.
- PMID 12647801.
- S2CID 22554802.
- PMID 25505885.
- PMID 26025964.
- PMID 26004498.
- PMID 25999859.
- PMID 25966361.
- PMID 25623859.
- PMID 25623749.
- PMID 25596375.
- PMID 23582229.
- S2CID 29527107.
- PMID 26836371.
- PMID 26711045.
- PMID 26061968.
- S2CID 5086235.
- ^ "Zalmoxis". European Medicines Agency. 2016.
- PMID 8607025.
- PMID 8528962.
- PMID 7541712.
- S2CID 24945709.
- PMID 7584059.
- PMID 8815006.
- PMID 16942024.
- S2CID 41765189.
- PMID 16831554.
- PMID 16529536.
- PMID 15715485.
- S2CID 30489989.
- S2CID 15932412.
- PMID 15336255.
- PMID 15308203.
- PMID 12127539.
- PMID 18981415.
- PMID 23318906.
- PMID 22889558.
- PMID 24006340.
- PMID 26087030.
- PMID 26282567.
- PMID 23617430.
- PMID 26630917.
- ^ WO application 2006000246, Gronowitz JS, "A method and kit for determination of thymidine kinase activity and use thereof", published 2006-02-24, assigned to Gronowitz JS
- PMID 16140350.
- PMID 25486911.
- PMID 22902741.
- PMID 22516918.
- S2CID 7615993.
- PMID 22247653.
- S2CID 25940455.
- PMID 6286702.
- PMID 21545220.
- PMID 27079872.
- PMID 23831216.
- PMID 25881026.
- PMID 16142366.
- PMID 7857717.
- PMID 1359886.
- S2CID 3134631.
- S2CID 20904034.
- PMID 4660462.
- S2CID 30647108.
- S2CID 39093011.
- PMID 5649653.
- PMID 1059244.
- PMID 11384664.
- ^ S2CID 242285275.)
{{cite journal}}: CS1 maint: DOI inactive as of February 2024 (link - S2CID 23072666.
- PMID 15809747.
- PMID 11205225.
- PMID 15583816.
- S2CID 205545163.
- PMID 25974835.
Further reading
- Garone C, Garcia-Diaz B, Emmanuele V, Lopez LC, Tadesse S, Akman HO, et al. (August 2014). "Deoxypyrimidine monophosphate bypass therapy for thymidine kinase 2 deficiency". EMBO Molecular Medicine. 6 (8): 1016–27. PMID 24968719.
- O'Neill KL, Buckwalter MR, Murray BK (November 2001). "Thymidine kinase: diagnostic and prognostic potential". Expert Review of Molecular Diagnostics. 1 (4): 428–33. S2CID 12790327.
- Topolcan O, Holubec L (February 2008). "The role of thymidine kinase in cancer diseases". Expert Opinion on Medical Diagnostics. 2 (2): 129–41. PMID 23485133.
- Jagarlamudi KK, Shaw M (September 2018). "Thymidine kinase 1 as a tumor biomarker: technical advances offer new potential to an old biomarker". Biomarkers in Medicine. 12 (9): 1035–1048. S2CID 51713461.
External links
- Thymidine+kinase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)