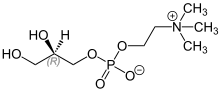
Glycerophosphorylcholine
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
L-α-Glycerophosphorylcholine (alpha-GPC, choline alfoscerate, sn-glycero-3-phosphocholine) is a natural
parasympathomimetic acetylcholine precursor[1] which has been investigated for its potential for the treatment of Alzheimer's disease[2] and other dementias.[3]
Alpha-GPC rapidly delivers choline to the brain across the blood–brain barrier and is a biosynthetic precursor of acetylcholine.[2] It is a non-prescription drug in most countries. The FDA determined that intake of no more than 196.2 mg/person/day is considered generally recognized as safe (GRAS).[4]
Production
Industrially, alpha-GPC is produced by the chemical or enzymatic deacylation of
phospholipids followed by chromatographic purification. Alpha-GPC may also be derived in small amounts from highly purified soy lecithin as well as from purified sunflower lecithin.[5][6]
Safety
Alpha-GPC metabolizes to
trimethylamine n-oxide in the gastrointestinal tract, which has implications for cardiovascular health. In one study, risk of stroke over a ten-year period was increased by about 40% in users of alpha-GPC.[7]
References
- PMID 12637119.
- ^ S2CID 34661218.
- S2CID 46175609.
- ^ "Generally Recognized as Safe (GRAS) Determination for the Use of AlphaSize® Alpha-Glycerylphosphoryl Choline" (PDF). United States Food and Drug Administration. 25 January 2012. Archived from the original (PDF) on 24 December 2013.
- PMID 24156263.
- PMID 23387341.
- PMID 34817582.
