Myristoylation
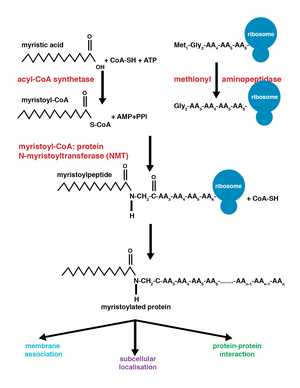
Myristoylation is a lipidation modification where a
Discovery
In 1982, Koiti Titani's lab identified an "N-terminal blocking group" on the catalytic subunit of cyclic AMP-dependent protein kinase in cows as n-tetradecanoyl.[6] Almost simultaneously in Claude B. Klee's lab, this same N-terminal blocking group was further characterized as myristic acid.[7] Both labs made this discovery utilizing similar techniques: mass spectrometry and gas chromatography.[6][7]
N-myristoyltransferase

The enzyme N-myristoyltransferase (NMT) or
Structure
The
Mechanism
The addition of the myristoyl group proceeds via a

Co-translational vs. post-translational addition
Co-translational and post-translational covalent modifications enable proteins to develop higher levels of complexity in cellular function, further adding diversity to the
Functions
Myristoylated proteins
| Protein | Physiological Role | Myristoylation Function |
|---|---|---|
| Actin | Cytoskeleton structural protein | Post-translational myristoylation during apoptosis [8] |
| Bid | Apoptosis promoting protein | Post-translational myristoylation after caspase cleavage targets protein to mitochondrial membrane[8] |
| MARCKS | actin cross-linking when phosphorylated by protein kinase C | Co-translational myristoylation aids in plasma membrane association |
| G-Protein | Signaling GTPase | Co-translational myristoylation aids in plasma membrane association[11] |
| Gelsolin | Actin filament-severing protein | Post-translational myristoylation up-regulates anti-apoptotic properties [8] |
| PAK2 | Serine/threonine kinase cell growth, mobility, survival stimulator | Post-translational myristoylation up-regulates apoptotic properties and induces plasma membrane localization[8] |
| Arf | vesicular trafficking and actin remodeling regulation | N-terminus myristoylation aids in membrane association |
| Hippocalcin | Neuronal calcium sensor | Contains a Ca2+/myristoyl switch |
| FSP1 | Apoptosis-inducing factor mitochondria-associated 2 (AIFM2) | Facilitates the association of FSP1 with the lipid-bilayer which enables ferroptosis resistance.[12] |
Myristoylation molecular switch
Myristoylation not only diversifies the function of a protein, but also adds layers of regulation to it. One of the most common functions of the myristoyl group is in
Both hydrophobic myristoyl groups and "basic patches" (highly positive regions on the protein) characterize myristoyl-electrostatic switches. The basic patch allows for favorable
Myristoyl-conformational switches can come in several forms.
Dual modifications of myristoylated proteins
Further modifications on N-myristoylated proteins can add another level of regulation for myristoylated protein. Dual
Myristoylation and palmitoylation are commonly coupled modifications. Myristoylation alone can promote transient membrane interactions[5] that enable proteins to anchor to membranes but dissociate easily. Further palmitoylation allows for tighter anchoring and slower dissociation from membranes when required by the cell. This specific dual modification is important for G protein-coupled receptor pathways and is referred to as the dual fatty acylation switch.[5][8]
Myristoylation is often followed by
Signal transduction
Myristoylation plays a vital role in membrane targeting and signal transduction[16] in plant responses to environmental stress. In addition, in signal transduction via G protein, palmitoylation of the α subunit, prenylation of the γ subunit, and myristoylation is involved in tethering the G protein to the inner surface of the plasma membrane so that the G protein can interact with its receptor.[17]
Apoptosis
Myristoylation is an integral part of
Impact on human health
Cancer
Viral infectivity

Prokaryotic and eukaryotic infections
Certain NMTs are therapeutic targets for development of drugs against bacterial
See also
- Acylation
- Prenylation
- Palmitoylation
- Palmitoleoylation
- Glycophosphatidylinositol
References
- ^ ISBN 978-0716743392.)
{{cite book}}: CS1 maint: multiple names: authors list (link - ^ ISBN 978-0-12-122722-7.
- PMID 30501005.
- PMID 3278984.
- ^ PMID 11527981.
- ^ PMID 6959104.
- ^ S2CID 40889752.
- ^ PMID 21056615.
- ^ PMID 10570244.
- ^ Snider, Jared. "Overview of Post-Translational Modifications (PTMs)". Thermo Scientific.
- PMID 11313912.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - S2CID 204833583.
- ^ PMID 7667880.
- ^ PMID 19898886.
- PMID 20583817.
- PMID 20467215.
- PMID 8521505.
- ^ PMID 2545855.
- PMID 21376230.
- PMID 17411366.


