Kidney stone disease
| Kidney stone disease | |
|---|---|
| Other names | Urolithiasis, kidney stone, renal calculus, nephrolith, kidney stone disease, |
| Frequency | 22.1 million (2015)[5] |
| Deaths | 16,100 (2015)[6] |
Kidney stone disease, also known as renal calculus disease, nephrolithiasis or urolithiasis, is a
Most calculi form by a combination of
In those who have had renal calculi, drinking fluids is a way to prevent them. Drinking fluids such that more than two liters of urine are produced per day is recommended.
Renal calculi have affected humans throughout history with a description of surgery to remove them dating from as early as 600 BC in ancient India by Sushruta.[1] Between 1% and 15% of people globally are affected by renal calculi at some point in their lives.[8][11] In 2015, 22.1 million cases occurred,[5] resulting in about 16,100 deaths.[6] They have become more common in the Western world since the 1970s.[8][12] Generally, more men are affected than women.[2][11] The prevalence and incidence of the disease rises worldwide and continues to be challenging for patients, physicians, and healthcare systems alike. In this context, epidemiological studies are striving to elucidate the worldwide changes in the patterns and the burden of the disease and identify modifiable risk factors that contribute to the development of renal calculi.[13]
Signs and symptoms

The hallmark of a stone that obstructs the ureter or renal pelvis is excruciating, intermittent pain that radiates from the flank to the groin or to the inner thigh.
The
Pain in the lower-left quadrant can sometimes be confused with diverticulitis because the sigmoid colon overlaps the ureter, and the exact location of the pain may be difficult to isolate due to the proximity of these two structures.
Risk factors
Dehydration from low fluid intake is a major factor in stone formation.[14][18] Individuals living in warm climates are at higher risk due to increased fluid loss.[19] Obesity, immobility, and sedentary lifestyles are other leading risk factors.[19]
High dietary intake of animal
Kidney stones can result from an underlying metabolic condition, such as distal renal tubular acidosis,[21] Dent's disease,[22] hyperparathyroidism,[23] primary hyperoxaluria,[24] or medullary sponge kidney. 3–20% of people who form kidney stones have medullary sponge kidney.[25][26]
Kidney stones are more common in people with Crohn's disease;[27] Crohn's disease is associated with hyperoxaluria and malabsorption of magnesium.[28]
A person with recurrent kidney stones may be screened for such disorders. This is typically done with a 24-hour urine collection. The urine is analyzed for features that promote stone formation.[17]
Calcium oxalate

Calcium is one component of the most common type of human kidney stones, calcium oxalate. Some studies suggest that people who take calcium or vitamin D as a dietary supplement have a higher risk of developing kidney stones.[29][30] In the United States, kidney stone formation was used as an indicator of excess calcium intake by the Reference Daily Intake committee for calcium in adults.[31]
In the early 1990s, a study conducted for the Women's Health Initiative in the US found that postmenopausal women who consumed 1000 mg of supplemental calcium and 400 international units of vitamin D per day for seven years had a 17% higher risk of developing kidney stones than subjects taking a placebo.[29] The Nurses' Health Study also showed an association between supplemental calcium intake and kidney stone formation.[30]
Unlike supplemental calcium, high intakes of dietary calcium do not appear to cause kidney stones and may actually protect against their development.[30][29] This is perhaps related to the role of calcium in binding ingested oxalate in the gastrointestinal tract. As the amount of calcium intake decreases, the amount of oxalate available for absorption into the bloodstream increases; this oxalate is then excreted in greater amounts into the urine by the kidneys. In the urine, oxalate is a very strong promoter of calcium oxalate precipitation—about 15 times stronger than calcium.
A 2004 study found that diets low in calcium are associated with a higher overall risk for kidney stone formation.[32] For most individuals, other risk factors for kidney stones, such as high intakes of dietary oxalates and low fluid intake, play a greater role than calcium intake.[33]
Other electrolytes
Calcium is not the only electrolyte that influences the formation of kidney stones. For example, by increasing urinary calcium excretion, high dietary sodium may increase the risk of stone formation.[30]
Drinking
Kidney stones are more likely to develop, and to grow larger, if a person has low dietary magnesium. Magnesium inhibits stone formation.[36]
Animal protein
Diets in Western nations typically contain a large proportion of animal protein. Eating animal protein creates an acid load that increases urinary excretion of calcium and uric acid and reduced citrate. Urinary excretion of excess sulfurous amino acids (e.g., cysteine and methionine), uric acid, and other acidic metabolites from animal protein acidifies the urine, which promotes the formation of kidney stones.[37] Low urinary-citrate excretion is also commonly found in those with a high dietary intake of animal protein, whereas vegetarians tend to have higher levels of citrate excretion.[30] Low urinary citrate, too, promotes stone formation.[37]
Vitamins
The evidence linking vitamin C supplements with an increased rate of kidney stones is inconclusive.[38][39] The excess dietary intake of vitamin C might increase the risk of calcium-oxalate stone formation.[40] The link between vitamin D intake and kidney stones is also tenuous.
Excessive vitamin D supplementation may increase the risk of stone formation by increasing the intestinal absorption of calcium; correction of a deficiency does not.[30]
Pathophysiology

Supersaturation of urine
When the urine becomes
Supersaturation of the urine with respect to a calculogenic compound is pH-dependent. For example, at a pH of 7.0, the solubility of uric acid in urine is 158 mg/100 mL. Reducing the pH to 5.0 decreases the solubility of uric acid to less than 8 mg/100 mL. The formation of uric-acid stones requires a combination of hyperuricosuria (high urine uric-acid levels) and low urine pH; hyperuricosuria alone is not associated with uric-acid stone formation if the urine pH is alkaline.[42] Supersaturation of the urine is a necessary, but not a sufficient, condition for the development of any urinary calculus.[25] Supersaturation is likely the underlying cause of uric acid and cystine stones, but calcium-based stones (especially calcium oxalate stones) may have a more complex cause.[43]
Randall's plaque
While supersaturation of urine may lead to
Pathogenic bacteria
Some
Inhibitors of stone formation
Normal
Sufficient dietary intake of
Hypocitraturia
Hypocitraturia or low urinary-citrate excretion (variably defined as less than 320 mg/day) can be a contributing cause of kidney stones in up to 2/3 of cases. The protective role of citrate is linked to several mechanisms; citrate reduces urinary supersaturation of calcium salts by forming soluble complexes with calcium ions and by inhibiting crystal growth and aggregation. Therapy with potassium citrate is commonly prescribed in clinical practice to increase urinary citrate and to reduce stone formation rates. Alkali citrate is also used to increase urine citrate levels. It can be prescribed or found over-the-counter in pill, liquid or powder form.[54][55]
Diagnosis
Diagnosis of kidney stones is made on the basis of information obtained from the history, physical examination,
Imaging studies
Calcium-containing stones are relatively
In people with a history of stones, those who are less than 50 years of age and are presenting with the symptoms of stones without any concerning signs do not require
Otherwise a noncontrast helical CT scan with 5 millimeters (0.2 in) sections is the diagnostic method to use to detect kidney stones and confirm the diagnosis of kidney stone disease.[16][56][60][65][7] Near all stones are detectable on CT scans with the exception of those composed of certain drug residues in the urine,[58] such as from indinavir.
Where a CT scan is unavailable, an
Renal ultrasonography can sometimes be useful, because it gives details about the presence of hydronephrosis, suggesting that the stone is blocking the outflow of urine.[58] Radiolucent stones, which do not appear on KUB, may show up on ultrasound imaging studies. Other advantages of renal ultrasonography include its low cost and absence of radiation exposure. Ultrasound imaging is useful for detecting stones in situations where X-rays or CT scans are discouraged, such as in children or pregnant women.[66] Despite these advantages, renal ultrasonography in 2009 was not considered a substitute for noncontrast helical CT scan in the initial diagnostic evaluation of urolithiasis.[65] The main reason for this is that, compared with CT, renal ultrasonography more often fails to detect small stones (especially ureteral stones) and other serious disorders that could be causing the symptoms.[14]
On the contrary, a 2014 study suggested that ultrasonography should be used as the initial diagnostic imaging test, with further imaging studies be performed at the discretion of the physician on the basis of clinical judgment, and using
-
Bilateral kidney stones can be seen on thisKUB radiograph. There are phleboliths in the pelvis, which can be misinterpreted as bladder stones.
-
CT scan of abdomen without contrast, showing a 3-mm stone (marked by an arrow) in the left proximal ureter
-
Renal ultrasonographof a stone located at the pyeloureteric junction with accompanying hydronephrosis.
-
Measurement of a 5.6 mm large kidney stone in soft tissue versus skeletal CT window.
Laboratory examination
Laboratory investigations typically carried out include:[56][65][58][68]
- leukocytes, urinary casts, and crystals;
- urine culture to identify any infecting organisms present in the urinary tract and sensitivityto determine the susceptibility of these organisms to specific antibiotics;
- neutrophil granulocytecount) suggestive of bacterial infection, as seen in the setting of struvite stones;
- renal function tests to look for abnormally high blood calcium levels (hypercalcemia);
- 24 hour urine collection to measure total daily urinary volume, magnesium, sodium, uric acid, calcium, citrate, oxalate, and phosphate;
- collection of stones (by urinating through a StoneScreen kidney stone collection cup or a simple tea strainer) is useful. Chemical analysis of collected stones can establish their composition, which in turn can help to guide future preventive and therapeutic management.
Composition
| Kidney stone type | Relative incidence (adults)[69] | Circumstances | Color and microscopy appearance | Radio-density | Details |
|---|---|---|---|---|---|
| Calcium oxalate | 60% | when urine is acidic (decreased pH)[70] | Black/dark brown  |
Radio-opaque | Some of the oxalate in urine is produced by the body. Calcium and oxalate in the diet play a part but are not the only factors that affect the formation of calcium oxalate stones. Dietary oxalate is found in many vegetables, fruits, and nuts. Calcium from bone may also play a role in kidney stone formation. |
| Calcium phosphate | 10-20% | when urine is alkaline (high pH) | Dirty white  |
Radio-opaque | Tends to grow in alkaline urine especially when Proteus bacteria are present. The most common type among pregnant women.[69] |
| Uric acid | 10-20% | when urine is persistently acidic | Yellow/reddish brown |
Radiolucent | Diets rich in animal proteins and purines: substances found naturally in all food but especially in organ meats, fish, and shellfish. |
| Struvite | 3% | infections in the kidney and when urine is alkaline (high pH) | Dirty white  |
Radio-opaque | Prevention of struvite stones depends on staying infection-free. Diet has not been shown to affect struvite stone formation. |
| Cystine | 1–2%[71] | rare genetic disorder | Pink/yellow  |
Radio-opaque | Cystine, an amino acid (a dimer of cysteine, of the building blocks of protein), leaks through the kidneys and into the urine to form crystals. |
| Xanthine[72] | extremely rare | Brick red | Radiolucent |



Calcium-containing stones
By far, the most common type of kidney stones worldwide contains calcium. For example, calcium-containing stones represent about 80% of all cases in the United States; these typically contain calcium oxalate either alone or in combination with calcium phosphate in the form of apatite or brushite.[25][52] Factors that promote the precipitation of oxalate crystals in the urine, such as primary hyperoxaluria, are associated with the development of calcium oxalate stones.[24] The formation of calcium phosphate stones is associated with conditions such as hyperparathyroidism[23] and renal tubular acidosis.[73]
Calcium oxalate crystals can come in two varieties. Calcium oxalate monohydrate can appear as 'dumbbells' or as long ovals that resemble the individual posts in a picket fence. Calcium oxalate dihydrate have a tetragonal “envelope” appearance.[74]
Struvite stones
About 10–15% of urinary calculi are composed of
Struvite stones (triple-phosphate/magnesium ammonium phosphate) have a 'coffin lid' morphology by microscopy.[74]
Uric acid stones
About 5–10% of all stones are formed from
As noted above (section on calcium oxalate stones), people with inflammatory bowel disease (Crohn's disease, ulcerative colitis) tend to have hyperoxaluria and form oxalate stones. They also have a tendency to form urate stones. Urate stones are especially common after colon resection.
Uric acid stones appear as pleomorphic crystals, usually diamond-shaped. They may also look like squares or rods which are polarizable.[74]
Other types
People with certain rare
Location
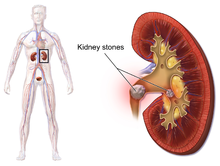
Urolithiasis refers to stones originating anywhere in the urinary system, including the kidneys and bladder.
Size
Stones less than 5 mm (0.2 in) in diameter pass spontaneously in up to 98% of cases, while those measuring 5 to 10 mm (0.2 to 0.4 in) in diameter pass spontaneously in less than 53% of cases.[84]
Stones that are large enough to fill out the renal calyces are called staghorn stones and are composed of struvite in a vast majority of cases, which forms only in the presence of urease-forming bacteria. Other forms that can possibly grow to become staghorn stones are those composed of cystine, calcium oxalate monohydrate, and uric acid.[85]
Prevention
Preventative measures depend on the type of stones. In those with calcium stones, drinking plenty of fluids, thiazide diuretics and citrate are effective as is allopurinol in those with high uric acid levels in urine.[86][87]
Dietary measures
Specific therapy should be tailored to the type of stones involved. Diet can have an effect on the development of kidney stones. Preventive strategies include some combination of dietary modifications and medications with the goal of reducing the excretory load of calculogenic compounds on the kidneys.[32][88][89] Dietary recommendations to minimize the formation of kidney stones include:
- increasing total fluid intake to achieve more than two liters per day of urine output;[90]
- limiting cola, including sugar-sweetened soft drinks;[86][90][91] to less than one liter per week.[92]
- limiting animal protein intake to no more than two meals daily (an association between animal protein and recurrence of kidney stones has been shown in men);[93]
- increasing citrate, or alkali intake, including from lemon and lime juice.[94] Citric acid in its natural form, such as from citrus fruits, "prevents small stones from becoming 'problem stones' by coating them and preventing other material from attaching and building onto the stones."[95];
- reducing sodium intake is associated with a reduction in urine calcium excretion.
Maintenance of dilute urine by means of vigorous fluid therapy is beneficial in all forms of kidney stones, so increasing urine volume is a key principle for the prevention of kidney stones. Fluid intake should be sufficient to maintain a urine output of at least 2
Calcium binds with available oxalate in the gastrointestinal tract, thereby preventing its absorption into the bloodstream. Reducing oxalate absorption decreases kidney stone risk in susceptible people.[96] Because of this, some doctors recommend increasing dairy intake so that its calcium content will serve as an oxalate binder. Taking calcium citrate tablets during or after meals containing high oxalate foods[97] may be useful if dietary calcium cannot be increased by other means as in those with lactose intolerance. The preferred calcium supplement for people at risk of stone formation is calcium citrate, as opposed to calcium carbonate, because it helps to increase urinary citrate excretion.[89]
Aside from vigorous oral hydration and eating more dietary calcium, other prevention strategies include avoidance of higher doses of supplemental vitamin C (since ascorbate is metabolized to oxalate) and restriction of oxalate-rich foods such as
Urine alkalinization
The mainstay for medical management of uric acid stones is
Increasing the urine pH to around 6.5 provides optimal conditions for
Slaked lime
Calcium hydroxide decreases urinary calcium when combined with food rich in oxalic acid such as green leafy vegetables.[100]
Diuretics
One of the recognized medical therapies for prevention of stones is the thiazide and thiazide-like diuretics, such as chlorthalidone or indapamide. These drugs inhibit the formation of calcium-containing stones by reducing urinary calcium excretion.[14] Sodium restriction is necessary for clinical effect of thiazides, as sodium excess promotes calcium excretion. Thiazides work best for renal leak hypercalciuria (high urine calcium levels), a condition in which high urinary calcium levels are caused by a primary kidney defect. Thiazides are useful for treating absorptive hypercalciuria, a condition in which high urinary calcium is a result of excess absorption from the gastrointestinal tract.[52]
Allopurinol
For people with hyperuricosuria and calcium stones, allopurinol is one of the few treatments that have been shown to reduce kidney stone recurrences. Allopurinol interferes with the production of uric acid in the liver. The drug is also used in people with gout or hyperuricemia (high serum uric acid levels).[101] Dosage is adjusted to maintain a reduced urinary excretion of uric acid. Serum uric acid level at or below 6 mg/100 mL is often a therapeutic goal. Hyperuricemia is not necessary for the formation of uric acid stones; hyperuricosuria can occur in the presence of normal or even low serum uric acid. Some practitioners advocate adding allopurinol only in people in whom hyperuricosuria and hyperuricemia persist, despite the use of a urine-alkalinizing agent such as sodium bicarbonate or potassium citrate.[42]
Treatment
Stone size influences the rate of spontaneous stone passage. For example, up to 98% of small stones (less than 5 mm (0.2 in) in diameter) may pass spontaneously through urination within four weeks of the onset of symptoms,[7] but for larger stones (5 to 10 mm (0.2 to 0.4 in) in diameter), the rate of spontaneous passage decreases to less than 53%.[84] Initial stone location also influences the likelihood of spontaneous stone passage. Rates increase from 48% for stones located in the proximal ureter to 79% for stones located at the vesicoureteric junction, regardless of stone size.[84] Assuming no high-grade obstruction or associated infection is found in the urinary tract, and symptoms are relatively mild, various nonsurgical measures can be used to encourage the passage of a stone.[42] Repeat stone formers benefit from more intense management, including proper fluid intake and use of certain medications, as well as careful monitoring.[102]
Pain management
Management of pain often requires intravenous administration of NSAIDs or opioids.[14] NSAIDs appear somewhat better than opioids or paracetamol in those with normal kidney function.[103] Medications by mouth are often effective for less severe discomfort.[66] The use of antispasmodics does not have further benefit.[9]
Medical expulsive therapy
The use of medications to speed the spontaneous passage of stones in the ureter is referred to as medical expulsive therapy.[104][105] Several agents, including alpha adrenergic blockers (such as tamsulosin) and calcium channel blockers (such as nifedipine), may be effective.[104] Alpha-blockers likely result in more people passing their stones, and they may pass their stones in a shorter time.[105] People taking alpha-blockers may also use less pain medication and may not need to visit the hospital.[105] Alpha-blockers appear to be more effective for larger stones (over 5 mm in size) than smaller stones.[105] However, use of alpha-blockers may be associated with a slight increase in serious, unwanted effects from this medication.[105] A combination of tamsulosin and a corticosteroid may be better than tamsulosin alone.[104] These treatments also appear to be useful in addition to lithotripsy.[7]
Lithotripsy
For a stone greater than 10 millimetres (0.39 in), ESWL may not help break the stone in one treatment; instead, two or three treatments may be needed. Some 80-85% of simple renal calculi can be effectively treated with ESWL.[7] A number of factors can influence its efficacy, including chemical composition of the stone, presence of anomalous renal anatomy and the specific location of the stone within the kidney, presence of hydronephrosis, body mass index, and distance of the stone from the surface of the skin.[106]
Common adverse effects of ESWL include acute
In addition to the aforementioned potential for acute kidney injury, animal studies suggest these acute injuries may progress to scar formation, resulting in loss of functional renal volume.
To address these concerns, the
Alpha-blockers are sometimes prescribed after shock wave lithotripsy to help the pieces of the stone leave the person's body.[111] By relaxing muscles and helping to keep blood vessels open, alpha blockers may relax the ureter muscles to allow the kidney stone fragments to pass. When compared to usual care or placebo treatment, alpha blockers may lead to faster clearing of stones, a reduced need for extra treatment and fewer unwanted effects.[111] They may also clear kidney stones in more adults than the standard shock wave lithotripsy procedure. The unwanted effects associated with alpha blockers are hospital emergency visits and return to hospital for stone-related issues, but these effects were more common in adults who did not receive alpha-blockers as a part of their treatment.[111]
Surgery
Most stones under 5 mm (0.2 in) pass spontaneously.[32][7] Prompt surgery may, nonetheless, be required in persons with only one working kidney, bilateral obstructing stones, a urinary tract infection and thus, it is presumed, an infected kidney, or intractable pain.[112] Beginning in the mid-1980s, less invasive treatments such as extracorporeal shock wave lithotripsy, ureteroscopy, and percutaneous nephrolithotomy began to replace open surgery as the modalities of choice for the surgical management of urolithiasis.[7] More recently, flexible ureteroscopy has been adapted to facilitate retrograde nephrostomy creation for percutaneous nephrolithotomy. This approach is still under investigation, though early results are favorable.[113] Percutaneous nephrolithotomy or, rarely, anatrophic nephrolithotomy, is the treatment of choice for large or complicated stones (such as calyceal staghorn calculi) or stones that cannot be extracted using less invasive procedures.[56][7]
Ureteroscopic surgery
More definitive ureteroscopic techniques for stone extraction (rather than simply bypassing the obstruction) include basket extraction and ultrasound ureterolithotripsy. Laser lithotripsy is another technique, which involves the use of a holmium:yttrium aluminium garnet (Ho:YAG) laser to fragment stones in the bladder, ureters, and kidneys.[116]
Ureteroscopic techniques are generally more effective than ESWL for treating stones located in the lower ureter, with success rates of 93–100% using Ho:YAG laser lithotripsy.[84] Although ESWL has been traditionally preferred by many practitioners for treating stones located in the upper ureter, more recent experience suggests ureteroscopic techniques offer distinct advantages in the treatment of upper ureteral stones. Specifically, the overall success rate is higher, fewer repeat interventions and postoperative visits are needed, and treatment costs are lower after ureteroscopic treatment when compared with ESWL. These advantages are especially apparent with stones greater than 10 mm (0.4 in) in diameter. However, because ureteroscopy of the upper ureter is much more challenging than ESWL, many urologists still prefer to use ESWL as a first-line treatment for stones of less than 10 mm, and ureteroscopy for those greater than 10 mm in diameter.[84] Ureteroscopy is the preferred treatment in pregnant and morbidly obese people, as well as those with bleeding disorders.[7]
Epidemiology
| Country | Earliest prevalence (years)[117] | Latest prevalence (years)[117] |
|---|---|---|
| United States | 2.6% (1964–1972) | 5.2% (1988–1994) |
| Italy | 1.2% (1983) | 1.7% (1993–1994) |
| Scotland | 3.8% (1977) | 3.5% (1987) |
| Spain | 0.1% (1977) | 10.0% (1991) |
| Turkey | n/a | 14.8% (1989) |
| Country | New cases per 100,000 (year)[117] | Trend |
|---|---|---|
| United States | 116 (2000) | decreasing |
| Germany | 720 (2000) | increasing |
| Japan | 114.3 (2005) | increasing |
| Spain | 270 (1984) | decreasing |
| Sweden | 200 (1969) | increasing |

Kidney stones affect all geographical, cultural, and racial groups. The
In North America and Europe, the annual number of new cases per year of kidney stones is roughly 0.5%. In the United States, the frequency in the population of urolithiasis has increased from 3.2% to 5.2% from the mid-1970s to the mid-1990s.[21] In the United States, about 9% of the population has had a kidney stone.[2]
The total cost for treating urolithiasis was US$2 billion in 2003.
A 2010 review concluded that rates of disease are increasing.[117]
History
The existence of kidney stones was first recorded thousands of years ago, with various explanations given; Joseph Glanville's Saducismus Triumphatus, for example, gives a detailed description of Abraham Mechelburg's voiding of small stones through his penis' virga, attributing the issue to witchcraft.[121]
In 1901, a stone discovered in the pelvis of an ancient Egyptian mummy was dated to 4,800 BC.
Medical texts from ancient Mesopotamia, India, China, Persia, Greece, and Rome all mentioned calculous disease. Part of the Hippocratic Oath suggests there were practicing surgeons in ancient Greece to whom physicians were to defer for lithotomies, or the surgical removal of stones. The Roman medical treatise De Medicina by Aulus Cornelius Celsus contained a description of lithotomy,[122] and this work served as the basis for this procedure until the 18th century.[123]
Examples of people who had kidney stone disease include
New techniques in lithotomy began to emerge starting in 1520, but the operation remained risky. After Henry Jacob Bigelow popularized the technique of litholapaxy in 1878,[125] the mortality rate dropped from about 24% to 2.4%. However, other treatment techniques continued to produce a high level of mortality, especially among inexperienced urologists.[123][124] In 1980, Dornier MedTech introduced extracorporeal shock wave lithotripsy for breaking up stones via acoustical pulses, and this technique has since come into widespread use.[106]
Etymology
The term renal calculus is from the Latin rēnēs, meaning "kidneys", and calculus, meaning "pebble". Lithiasis (stone formation) in the kidneys is called nephrolithiasis (/ˌnɛfroʊlɪˈθaɪəsɪs/), from nephro-, meaning kidney, + -lith, meaning stone, and -iasis, meaning disorder. A distinction between nephrolithiasis and urolithiasis can be made because not all urinary stones (uroliths) form in the kidney; they can also form in the bladder. But the distinction is often clinically irrelevant (with similar disease process and treatment either way) and the words are thus often used loosely as synonyms.
Children
Although kidney stones do not often occur in children, the incidence is increasing.[126] These stones are in the kidney in two thirds of reported cases, and in the ureter in the remaining cases. Older children are at greater risk independent of whether or not they are male or female.[127]
As with adults, most pediatric kidney stones are predominantly composed of calcium oxalate; struvite and calcium phosphate stones are less common. Calcium oxalate stones in children are associated with high amounts of calcium, oxalate, and magnesium in acidic urine.[128]
Treatment of kidney stones in children is similar to treatments for adults, including shock wave lithotripsy, medication, and treatment using scope through the bladder, kidney or skin.[129] Of these treatments, research is uncertain if shock waves are more effective than medication or a scope through the bladder, but it is likely less successful than a scope through skin into the kidney.[129] When going in with a scope through the kidney, a regular and a mini-sized scope likely have similar success rates of stone removal. Alpha-blockers, a type of medication, may increase the successful removal of kidney stones when compared with a placebo and without ibuprofen.[129]
Research
Metabolic syndrome and its associated diseases of obesity and diabetes as general risk factors for kidney stone disease are under research to determine if urinary excretion of calcium, oxalate and urate are higher than in people with normal weight or underweight, and if diet and physical activity have roles.[130][131] Dietary, fluid intake, and lifestyle factors remain major topics for research on prevention of kidney stones, as of 2017.[132]
Gut microbiota
The gut microbiota has been explored as a contributing factor for stone disease, indicating that some bacteria may be different in people forming kidney stones.[133] One bacterium, Oxalobacter formigenes, is potentially beneficial for mitigating calcium oxalate stones because of its ability to metabolize oxalate as its sole carbon source,[134] but 2018 research suggests that it is instead part of a network of oxalate degrading bacteria.[135] Additionally, one study found that oral antibiotic use, which alters the gut microbiota,[136] can increase the odds of a person developing a kidney stone.[137]
In animals
Among ruminants, uroliths more commonly cause problems in males than in females; the sigmoid flexure of the ruminant male urinary tract is more likely to obstruct passage. Early-castrated males are at greater risk, because of lesser urethral diameter.[138]
Low Ca:P intake ratio is conducive to phosphatic (e.g. struvite) urolith formation.[138] Incidence among wether lambs can be minimized by maintaining a dietary Ca:P intake ratio of 2:1.[138][139]
Alkaline (higher) pH favors formation of carbonate and phosphate calculi. For domestic ruminants, dietary cation: anion balance is sometimes adjusted to assure a slightly acidic urine pH, for prevention of calculus formation.[138]
Differing generalizations regarding effects of pH on formation of silicate uroliths may be found.[138][140] In this connection, it may be noted that under some circumstances, calcium carbonate accompanies silica in siliceous uroliths.[141]
Pelleted feeds may be conducive to formation of phosphate uroliths, because of increased urinary phosphorus excretion. This is attributable to lower saliva production where pelleted rations containing finely ground constituents are fed. With less blood phosphate partitioned into saliva, more tends to be excreted in urine.[142] (Most saliva phosphate is fecally excreted.[143])
Oxalate uroliths can occur in ruminants, although such problems from oxalate ingestion may be relatively uncommon. Ruminant urolithiasis associated with oxalate ingestion has been reported.[144] However, no renal tubular damage or visible deposition of calcium oxalate crystals in kidneys was found in yearling wether sheep fed diets containing soluble oxalate at 6.5 percent of dietary dry matter for about 100 days.[145]
Conditions limiting water intake can result in stone formation.[146]
Various surgical interventions, e.g. amputation of the urethral process at its base near the glans penis in male ruminants, perineal urethrostomy, or tube cystostomy may be considered for relief of obstructive urolithiasis.[146]
See also
References
- ^ ISBN 978-3-319-12105-5. Archivedfrom the original on 8 September 2017.
- ^ a b c d e f g h i j k l m n o p q r s "Kidney Stones in Adults". February 2013. Archived from the original on 11 May 2015. Retrieved 22 May 2015.
- ISBN 978-3-642-28732-9. Archivedfrom the original on 8 September 2017.
- ^ PMID 25364887.
- ^ PMID 27733282.
- ^ PMID 27733281.
- ^ PMID 17332586.
- ^ S2CID 28313474.
- ^ PMID 26120804.
- PMID 27616037.
- ^ S2CID 221572651.
- PMID 12675858.
- license.
- ^ a b c d e f g Preminger GM (2007). "Chapter 148: Stones in the Urinary Tract". In Cutler RE (ed.). The Merck Manual of Medical Information Home Edition (3rd ed.). Whitehouse Station, New Jersey: Merck Sharp and Dohme Corporation. Archived from the original on 8 December 2014. Retrieved 7 August 2011.
- ^ Nephrolithiasis~Overview at eMedicine § Background.
- ^ a b c Pearle MS, Calhoun EA, Curhan GC (2007). "Ch. 8: Urolithiasis" (PDF). In Litwin MS, Saigal CS (eds.). Urologic Diseases in America (NIH Publication No. 07–5512). Bethesda, Maryland: National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, United States Public Health Service, United States Department of Health and Human Services. pp. 283–319. Archived (PDF) from the original on 18 October 2011.
- ^ ISBN 978-0-7614-7772-3. Archivedfrom the original on 14 April 2021. Retrieved 6 November 2020.
- ^ PMID 8561157.
- ^ OCLC 944472408.
- PMID 20842614.
- ^ (PDF) from the original on 15 August 2011.
- (PDF) from the original on 5 November 2012.
- ^ a b National Endocrine and Metabolic Diseases Information Service (2006). "Hyperparathyroidism (NIH Publication No. 6–3425)". Information about Endocrine and Metabolic Diseases: A-Z list of Topics and Titles. Bethesda, Maryland: National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Public Health Service, US Department of Health and Human Services. Archived from the original on 24 May 2011. Retrieved 27 July 2011.
- ^ S2CID 23503869.
- ^ a b c d e Reilly RF, Ch. 13: "Nephrolithiasis". In Reilly & Perazella 2005, pp. 192–207.
- ^ National Kidney and Urologic Diseases Information Clearinghouse (2008). "Medullary Sponge Kidney (NIH Publication No. 08–6235)". Kidney & Urologic Diseases: A-Z list of Topics and Titles. Bethesda, Maryland: National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Public Health Service, US Department of Health and Human Services. Archived from the original on 7 August 2011. Retrieved 27 July 2011.
- ^ National Digestive Diseases Information Clearinghouse (2006). "Crohn's Disease (NIH Publication No. 06–3410)". Digestive Diseases: A-Z List of Topics and Titles. Bethesda, Maryland: National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, United States Public Health Service, United States Department of Health and Human Services. Archived from the original on 9 June 2014. Retrieved 27 July 2011.
- from the original on 20 March 2020. Retrieved 1 November 2013.
- ^ a b c "Tolerable upper intake levels: Calcium and vitamin D". In Committee to Review Dietary Reference Intakes for Vitamin D and Calcium 2011, pp. 403–56.
- ^ from the original on 31 January 2021. Retrieved 18 May 2019.
- ^ "Summary". In Committee to Review Dietary Reference Intakes for Vitamin D and Calcium 2011, pp. 1–14.
- ^ PMID 15191979.
- (PDF) from the original on 16 January 2016.
- ISBN 978-0-309-65799-0. Archivedfrom the original on 30 July 2011.
- PMID 27445166.
- ^ PMID 24127630.
- ^ PMID 23732207.
- PMID 9818798.
- PMID 15000301.
- PMID 26463139.
- ^ Perazella MA, Ch. 14: "Urinalysis". In Reilly & Perazella 2005, pp. 209–26.
- ^ a b c d e f Knudsen BE, Beiko DT, Denstedt JD, Ch. 16: "Uric Acid Urolithiasis". In Stoller & Meng 2007, pp. 299–308.
- ^ Nephrolithiasis~Overview at eMedicine § Pathophysiology.
- PMID 4182793.
- PMID 7359628.
- PMID 17856988.
- PMID 22229020.
- PMID 25383321.
- PMID 2841283.
- S2CID 245012979.
- PMID 31749785.
- ^ PMID 16200192.
- PMID 23924538.
- ^ "Educate Your Patients about Kidney Stones" (PDF). kidney.org. Archived (PDF) from the original on 26 October 2020.
- PMID 19911682.
- ^ ISBN 978-0-7817-9141-0. Archivedfrom the original on 14 April 2021. Retrieved 6 November 2020.
- ISBN 978-1-58255-082-4.
- ^ (PDF) from the original on 23 November 2011.
- PMID 27578040.
- ^ S2CID 73387308.
- ISBN 978-1-4160-3105-5. Archived from the originalon 8 October 2011.
- PMID 10494278.
- ^ American College of Emergency Physicians (27 October 2014). "Ten Things Physicians and Patients Should Question". Choosing Wisely. Archived from the original on 7 March 2014. Retrieved 14 January 2015.
- ^ "American Urological Association | Choosing Wisely". www.choosingwisely.org. Archived from the original on 23 February 2017. Retrieved 28 May 2017.
- ^ ISBN 978-0-7817-7513-7. Archivedfrom the original on 21 March 2021. Retrieved 6 November 2020.
- ^ PMID 24109196.
- (PDF) from the original on 14 March 2020. Retrieved 25 September 2019.
- ^ National Kidney and Urologic Diseases Information Clearinghouse (2007). "Kidney Stones in Adults (NIH Publication No. 08–2495)". Kidney & Urologic Diseases: A-Z list of Topics and Titles. Bethesda, Maryland: National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Public Health Service, US Department of Health and Human Services. Archived from the original on 26 July 2011. Retrieved 27 July 2011.
- ^ PMID 22150656.
Including cystine stone incidence of 1% - ISBN 978-0-7817-1750-2. Archivedfrom the original on 8 September 2017.
- ^ "Cystine stones". UpToDate. Archived from the original on 26 February 2014. Retrieved 20 February 2014.
- ^ Bailey & Love's/25th/1296
- ^ National Endocrine and Metabolic Diseases Information Service (2008). "Renal Tubular Acidosis (NIH Publication No. 09–4696)". Kidney & Urologic Diseases: A-Z list of Topics and Titles. Bethesda, Maryland: National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Public Health Service, US Department of Health and Human Services. Archived from the original on 28 July 2011. Retrieved 27 July 2011.
- ^ a b c d De Mais D (2009). ASCP Quick Compendium of Clinical Pathology (2nd ed.). Chicago: ASCP Press.
- ^ ISBN 978-0-7817-4750-9. Archivedfrom the original on 20 March 2021. Retrieved 6 November 2020.
- PMID 7783706.
- PMID 8976113.
- PMID 5652624.
- PMID 7504361.
- ^ "Patient Information about Crixivan for HIV (Human Immunodeficiency Virus) Infection" (PDF). Crixivan® (indinavir sulfate) Capsules. Whitehouse Station, New Jersey: Merck Sharp & Dohme Corporation. 2010. Archived (PDF) from the original on 15 August 2011. Retrieved 27 July 2011.
- ISBN 978-1-60795-084-4. Archivedfrom the original on 20 March 2021. Retrieved 25 August 2020.
- PMID 2231920.
- ^ McNutt WF (1893). "Section IV: Diseases of the Bladder, Chapter VII: Vesical Calculi (Cysto-lithiasis)". Diseases of the Kidneys and Bladder: A Text-Book for Students of Medicine. Philadelphia: J.B. Lippincott Company. pp. 185–6. Archived from the original on 14 April 2021. Retrieved 6 November 2020.
- ^ S2CID 36265416.
- PMID 9048853.
- ^ PMID 23546565.
- ^ PMID 25364887.
- from the original on 22 August 2005.
- ^ from the original on 15 October 2008.
- ^ PMID 22896859.
- PMID 23676355.
- ^ "What are kidney stones?". kidney.org. Archived from the original on 14 May 2013. Retrieved 19 August 2013.
- PMID 16837923.
- PMID 25512810.
- ^ "Citric Acid and Kidney Stones" (PDF). uwhealth.org. Archived (PDF) from the original on 5 July 2010.
- PMID 16529131.
- ^ S2CID 28256459.
- ^ (PDF) from the original on 21 September 2017. Retrieved 3 June 2011.
- PMID 21121431.
- ^ "Effect of addition of calcium hydroxide to foods rich in oxalic acid on calcium and oxalic acid metabolism | Request PDF". Archived from the original on 7 November 2021. Retrieved 6 March 2021.
- PMID 3607420.
- PMID 8863542.
- PMID 29174580.
- ^ PMID 19560860.
- ^ PMID 29620795.
- ^ a b c d e f g h i Shock Wave Lithotripsy Task Force (2009). "Current Perspective on Adverse Effects in Shock Wave Lithotripsy" (PDF). Clinical Guidelines. Linthicum, Maryland: American Urological Association. Archived from the original (PDF) on 18 July 2013. Retrieved 13 October 2015.
- ^ Lingeman JE, Matlaga BR, Evan AP (2007). "Surgical Management of Urinary Lithiasis". In Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA (eds.). Campbell-Walsh Urology. Philadelphia: W. B. Saunders. pp. 1431–1507.
- PMID 17993340.
- ^ ISBN 978-0-7817-0263-8.
- ^ a b c Evan AP, Willis LR (2007). "Ch. 41: Extracorporeal Shock Wave Lithotripsy: Complications". In Smith AD, Badlani GH, Bagley DH, Clayman RV, Docimo SG (eds.). Smith's Textbook on Endourology. Hamilton, Ontario, Canada: B C Decker, Inc. pp. 353–65.
- ^ PMID 33179245.
- ^ Young JG, Keeley FX, Ch. 38: "Indications for Surgical Removal, Including Asymptomatic Stones" in Rao, Preminger & Kavanagh 2011, pp. 441–454.
- PMID 22563900.
- ^ Lam JS, Gupta M, Ch. 25: "Ureteral Stents". In Stoller & Meng 2007, pp. 465–83.
- PMID 30726554.
- ISBN 978-1-84800-362-0, archived from the original on 20 February 2021, retrieved 6 November 2020 in Rao, Preminger & Kavanagh 2011, pp. 301–310.
- ^ PMID 20811557.
- ISBN 978-1-58829-109-7.
- S2CID 1541253. Archived from the originalon 19 May 2020.
- ISBN 978-0-7817-9149-6. Archivedfrom the original on 9 September 2016.
- OCLC 943229347.
- ^ Celsus AC (1831). "Book VII, Chapter XXVI: Of the operation necessary in a suppression of urine, and lithotomy". In Collier GF (ed.). A translation of the eight books of Aul. Corn. Celsus on medicine (2nd ed.). London: Simpkin and Marshall. pp. 306–14. Archived from the original on 8 July 2014.
- ^ S2CID 44311421.
- ^ ISBN 978-0-632-06140-2.
- ^ Bigelow HJ (1878). Litholapaxy or rapid lithotrity with evacuation. Boston: A. Williams and Company. p. 29. Archived from the original on 14 April 2021. Retrieved 25 August 2020.
- PMID 22595060.
- ^ "Diet and Definition of Kidney Stones, Renal Calculi". Archived from the original on 17 November 2007. Retrieved 11 October 2013.
- PMID 23953243.
- ^ PMID 29859007.
- PMID 30066054.
- S2CID 4585476.
- PMID 28830863.
- PMID 32503496.
- PMID 12147479.
- S2CID 14055215.
- PMID 33330122.
- PMID 29748329.
- ^ ISBN 978-1-4377-2354-0. Archivedfrom the original on 14 April 2021. Retrieved 17 June 2018.
- PMID 5841867.
- S2CID 10130833. Archived from the original(PDF) on 2 March 2019.
- PMID 17649146.
- PMID 3399614.
- (PDF) from the original on 16 April 2021. Retrieved 29 August 2019.
- ^ Waltner-Toews, D. and D. H. Meadows. 1980. Case report: Urolithiasis in a herd of beef cattle associated with oxalate ingestion. Can. Vet. J. 21: 61-62
- PMID 4647453.
- ^ a b Kahn CM, ed. (2005). Merck veterinary manual (9th ed.). Whitehouse Station: Merck & Co., Inc.
Notes
- Committee to Review Dietary Reference Intakes for Vitamin D and Calcium, Institute of Medicine of the National Academies (2011). Ross AC, Taylor CL, Yaktine AL, Del HB (eds.). Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: The National Academies Press. from the original on 9 September 2014. Retrieved 21 June 2011.
- Rao PN, Preminger GM, Kavanagh JP, eds. (2011). Urinary Tract Stone Disease (1st ed.). London: Springer-Verlag. ISBN 978-1-84800-361-3.
- Reilly RF, Perazella MA, eds. (2005). Nephrology in 30 Days (1st ed.). New York: The McGraw-Hill Companies, Inc. ISBN 978-0-07-143701-1. Archivedfrom the original on 14 April 2021. Retrieved 25 August 2020.
- Stoller ML, Meng MV, eds. (2007). Urinary stone disease: the practical guide to medical and surgical management (1st ed.). Totowa, New Jersey: Humana Press. ISBN 978-1-59259-972-1.
External links
- Kidney stone disease at Curlie
- Information from the European Urological Association
- Kidney Stone Guide Book Archived 3 August 2020 at the Wayback Machine – University of Chicago Kidney Stone Program






