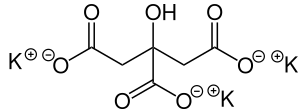
Potassium citrate
| |
| Names | |
|---|---|
| Preferred IUPAC name
Tripotassium 2-hydroxypropane-1,2,3-tricarboxylate | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.011.596 |
| E number | E332(ii) (antioxidants, ...) |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| K3C6H5O7 | |
| Molar mass | 306.395 g/mol |
| Appearance | white powder hygroscopic
|
| Odor | odorless |
| Density | 1.98 g/cm3 |
| Melting point | 180 °C (356 °F; 453 K)[1] |
| Boiling point | 230 °C (446 °F; 503 K)[1] |
| soluble | |
| Solubility | soluble in glycerin (95%)
insoluble in ethanol |
| Acidity (pKa) | 8.5 |
| Pharmacology | |
| A12BA02 (WHO) | |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
170 mg/kg (IV, dog) 5400mg/kg (oral, rat) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Potassium citrate (also known as tripotassium citrate) is a
As a
.In 2020, it was the 297th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[2][3]
Synthesis
Potassium citrate can be synthesized by the neutralization of citric acid which is achieved by the addition of potassium bicarbonate, potassium carbonate or potassium hydroxide to it. The solution can then be filtered and the solvent can be evaporated till granulation.
Uses
Potassium citrate is rapidly absorbed when given by mouth, and is excreted in the urine.[4] Since it is an alkaline salt, it is effective in reducing the pain and frequency of urination when these are caused by highly acidic urine.[5] It is used for this purpose in dogs and cats, but is chiefly employed as a non-irritating diuretic.
Potassium citrate is an effective way to treat/manage
It is widely used to treat urinary calculi (
It is also used as an
It is also used in many
Frequently used in an aqueous solution with other potassium salts, it is a
Administration
Potassium citrate liquid is usually administered by mouth in a diluted aqueous solution, because of its somewhat caustic effect on the stomach lining, and the potential for other mild health hazards. Pill tablets also exist in normal, and extended-release formulations.
References
- ^ a b "Potassium Citrate". hazard.com. Archived from the original on 2017-08-15.
{{cite web}}: CS1 maint: unfit URL (link) - ^ "The Top 300 of 2020". ClinCalc. Retrieved 7 October 2022.
- ^ "Potassium Citrate - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.
- ^ Medscape on hypocitraturia
- ^ Potassium Citrate for Kidney Stones
- PMID 26439475.
- ^ "Potassium citrate for cystitis". patient.info.
- ^ "Soft Drinks with Potassium Citrate".
- ^ Fire, Impact. "What is a Class K Fire Extinguisher Used For?". resources.impactfireservices.com. Retrieved 2022-08-18.
- ^ US Fire extinguishing composition and method for fire extinguishing 5945025A, James A. Cunningham
- ^ Xiaofang Wangy et al 2019 IOP Conf. Ser.: Mater. Sci. Eng. 490 022047
External links
- Tanner, G.A. "Potassium citrate improves renal function in rats with polycystic kidney disease". Journal of the American Society of Nephrology. Retrieved December 17, 2016.
