5-HT3 receptor
This article may be too technical for most readers to understand. (March 2024) |
The 5-HT3 receptor belongs to the
As with other ligand gated ion channels, the 5-HT3 receptor consists of five subunits arranged around a central ion conducting pore, which is permeable to
Structure
The 5-HT3 receptor differs markedly in structure and mechanism from the other
Human and mouse genes
The genes encoding human 5-HT3 receptors are located on
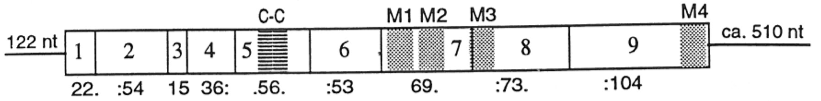
The human 5-HT3A receptor gene is similar in structure to the mouse gene which has 9 exons and is spread over ~13 kb. Four of its introns are exactly in the same position as the introns in the homologous α7-acetylcholine receptor gene, clearly showing their evolutionary relationship.[12][13]

Expression. The 5-HT3C, 5-HT3D and 5-HT3E genes tend to show peripherally restricted pattern of expression, with high levels in the
Polymorphism. In patients treated with
Tissue distribution
The 5-HT3 receptor is expressed throughout the central and peripheral nervous systems and mediates a variety of physiological functions.[14] On a cellular level, it has been shown that postsynaptic 5-HT3 receptors mediate fast excitatory synaptic transmission in rat neocortical interneurons, amygdala, and hippocampus, and in ferret visual cortex.[15][16][17][18] 5-HT3 receptors are also present on presynaptic nerve terminals. There is some evidence for a role in modulation of neurotransmitter release,[19][20] but evidence is inconclusive.[21]
Effects
When the receptor is activated to open the ion channel by
- emesis.[22]
Ligands
Agonists
Agonists for the receptor include:
- Cereulide
- 2-methyl-5-HT
- Alpha-Methyltryptamine
- Bufotenin
- Chlorophenylbiguanide[22]
- Ethanol
- Ibogaine
- Phenylbiguanide
- Quipazine
- RS-56812 – Potent and selective 5-HT3 partial agonist, 1000× selectivity over other serotonin receptors
- SR-57227
- Varenicline[26]
- YM-31636[27]
- S 21007[28] (SAR c.f. CGS-12066A)
Antagonists
Antagonists for the receptor (sorted by their respective therapeutic application) include:
- Antiemetics
- Gastroprokinetics
- Alosetron
- Batanopride
- Metoclopramide (high doses)
- Renzapride
- Zacopride
- M1, the major active metabolite of mosapride
- Antidepressants
- Antipsychotics
- Antimalarials
- Others
- 3-Tropanyl indole-3-carboxylate
- Cannabidiol (CBD)
- Delta-9-Tetrahydrocannabinol
- Lamotrigine (epilepsy and bipolar disorder)
- Memantine (Alzheimer's disease medication)
- Menthol[29]
- Thujone
Positive allosteric modulators
These agents are not agonists at the receptor, but increase the affinity or efficacy of the receptors for an agonist:
Negative allosteric modulators
Negative allostric modulators of the serotonin 5-HT3 receptor include bupropion and hydroxybupropion.[33]
Discovery
Identification of the 5-HT3 receptor did not take place until 1986, lacking selective pharmacological tools.
See also
- 5-HT1 receptor
- 5-HT2 receptor
- 5-HT4 receptor
- 5-HT5 receptor
- 5-HT6 receptor
- 5-HT7 receptor
References
- ^ PMID 18761359.
- ^ PMID 17073663.
- S2CID 36985954.
- S2CID 4401851.
- PMID 10521471.
- S2CID 10168401.
- S2CID 37832903.
- S2CID 40072549.
- PMID 21345729.
- S2CID 25811139.
- PMID 19086255.
- ^ S2CID 28979681.
- ^ Uetz, P. (1992) Das 5HT3-Rezeptorgen der Maus. Diploma Thesis, University of Heidelberg, 143 pp.
- ^ ISBN 3-540-66127-1.
- PMID 12196560.
- PMID 7520482.
- S2CID 22554779.
- PMID 9334409.
- PMID 9489730.
- PMID 9448942.
- S2CID 36074796.
- ^ ISBN 0-443-07145-4., page 187.
- PMID 19942458.
- PMID 30240783.
- S2CID 6060490.
- PMID 20965579.
- PMID 12628674.
- PMID 8982686.
- S2CID 111928.
- PMID 23594147.
- PMID 21342788.
- ^ S2CID 22050514.
- PMID 38931362.
- PMID 17373882.
External links
- 5-HT3+Receptor at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
