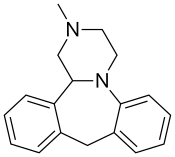
Mianserin
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Tolvon, others |
| Other names | Mianserin hydrochloride; Org GB 94[1][2] |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
oxidation, N-demethylation)[4] | |
| Elimination half-life | 21–61 hours[5] |
| Excretion | Urine: 4–7%[4] Feces: 14–28%[4] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Mianserin, sold under the brand name Tolvon among others, is an atypical antidepressant that is used primarily in the treatment of depression in Europe and elsewhere in the world.[6] It is a tetracyclic antidepressant (TeCA). Mianserin is closely related to mirtazapine, both chemically and in terms of its actions and effects, although there are significant differences between the two drugs.[7]
Medical uses
Mianserin at higher doses (30–90mg/day) is used for the treatment of major depressive disorder.[6]
It can also be used at lower doses (around 10mg/day) to treat insomnia.[8][9]
Contraindications
It should not be given, except if based on clinical need and under strict medical supervision, to people younger than 18 years old, as it can increase the risk of suicide attempts and suicidal thinking, and it can increase aggressiveness.[6]
While there is no evidence that it can harm a fetus from animal models, there are no data showing it safe for pregnant women to take.[6]
People with severe liver disease should not take mianserin, and it should be used with caution for people with epilepsy or who are at risk for seizures, as it can lower the threshold for seizures. If based on clinical decision, normal precautions should be exercised and the dosages of mianserin and any concurrent therapy kept under review and adjusted as needed.[6]
Side effects
Very common (incidence > 10%) adverse effects include constipation, dry mouth, and drowsiness at the beginning of treatment.[5][6]
Common (1% < incidence ≤ 10%) adverse effects include drowsiness during maintenance therapy, tremor, headache, dizziness, vertigo, and weakness.[5]
Uncommon (0.1% < incidence ≤ 1%) adverse effects include weight gain.[5]
Withdrawal
Abrupt or rapid discontinuation of mianserin may provoke a
Overdose
Overdose of mianserin is known to produce sedation, coma, hypotension or hypertension, tachycardia, and QT interval prolongation.[11]
Interactions
Mianserin may enhance the sedative effects of drugs such as alcohol, anxiolytics, hypnotics, or antipsychotics when co-administered. It may decrease the efficacy of
Pharmacology
Pharmacodynamics
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| SERT | 4,000 | Human | [13] |
| NET | 71 | Human | [13] |
| DAT | 9,400 | Human | [13] |
| 5-HT1A | 400–2,600 | Human | [14][15] |
| 5-HT1B | ≥2,800 | Rat | [16] |
| 5-HT1D | 220–400 | Human | [17][18] |
| 5-HT1E | ND | ND | ND |
| 5-HT1F | 13 | Human | [14] |
| 5-HT2A | 1.6–55 | Human | [19][20] |
| 5-HT2B | 1.6–20 | Human | [21][22] |
| 5-HT2C | 0.63–6.5 | Human | [19][23] |
| 5-HT3 | 5.8–300 | Rodent | [24][15] |
| 5-HT4 | ND | ND | ND |
| 5-HT5A | ND | ND | ND |
| 5-HT6 | 55–81 | Human | [25][26] |
| 5-HT7 | 48–56 | Human | [27][28][29] |
| α1 | 34 | Human | [30] |
| α2 | 73 | Human | [30] |
| α2A | 4.8 | Human | [27] |
| α2B | 27 | Human | [31] |
| α2C | 3.8 | Human | [27] |
D1 |
426–1,420 | Human | [15][27] |
D2 |
2,100–2,700 | Human | [30][32] |
D3 |
2,840 | Human | [30] |
D4 |
ND | ND | ND |
D5 |
ND | ND | ND |
| H1 | 0.30–1.7 | Human | [33][30][27] |
| H2 | 437 | Human | [34] |
| H3 | 95,500 | Human | [34] |
| H4 | >100,000 | Human | [34][35] |
| mACh | 820 | Human | [30] |
MOR |
21,000 | Human | [36] |
DOR |
30,200 | Human | [36] |
KOR |
530 ( EC50 ) |
Human | [36] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. | |||
Mianserin appears to exert its effects via antagonism of
Blockade of the H1 and possibly α1-adrenergic receptors has
Along with mirtazapine, although to a lesser extent in comparison, mianserin has sometimes been described as a noradrenergic and specific serotonergic antidepressant (NaSSA).[41] However, the actual evidence in support of this label has been regarded as poor.[42]
Pharmacokinetics
The
Chemistry

Mianserin is a tetracyclic piperazinoazepine. Mirtazapine was developed by the same team of organic chemists and differs via addition of a nitrogen atom in one of the rings.[43][44] (S)-(+)-Mianserin is approximately 200–300 times more active than its enantiomer (R)-(−)-mianserin; hence, the activity of mianserin lies in the (S)-(+) isomer.[citation needed]
History
It was
Mianserin was one of the first antidepressants to reach the UK market that was less dangerous than the tricyclic antidepressants in overdose; as of 2012 it was not prescribed much in the UK.[47]
Society and culture

Generic names
Mianserin is the
Brand names
Mianserin is marketed in many countries mainly under the brand name Tolvon. It is also available throughout the world under a variety of other brand names including Athymil, Bonserin, Bolvidon, Deprevon, Lantanon, Lerivon, Lumin, Miansan, Serelan, Tetramide, and Tolvin among others.[1][2][48]
Availability
Mianserin is not approved for use in the United States, but is available in the United Kingdom and other European countries.[50][51] A mianserin generic drug received TGA approval in May 1996 and is available in Australia.[52]
Research
The use of mianserin to help people with schizophrenia who are being treated with antipsychotics has been studied in clinical trials; the outcome is unclear.[53][54]
References
- ^ ISBN 978-3-88763-075-1.
- ^ a b c "International brands for mianserin". Drugs.com. Retrieved 20 August 2017.
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b c d e f g h i j Truven Health Analytics, Inc. Drugdex System (Internet) [cited 2013 Sep 29]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- ^ a b c d e "Tolvon Product Information" (PDF). Medicines. AU: Merck Sharp & Dohme. Archived from the original (PDF) on 2016-04-02. Retrieved 2013-10-05 – via GuildLink.
- ^ a b c d e f g "Mianserin 30 mg film-coated tablets". UK Electronic Medicines Compendium. January 2014. Retrieved 20 August 2017.
- ^ "A Comparison of the Physicochemical and Biological Properties of Mirtazapine and Mianserin". Journal of pharmacy and pharmacology. Oxford University Press. April 2011. Retrieved 29 January 2022.
- ^ "Que faire devant une insomnie" [What to do when facing insomnia]. Sommeil (in French). Lyon, FR: University of Lyon.
- ^ "Traitement des troubles du sommeil" [Treatment of the troubles of sleep]. Research gate (in French). Archived from the original on 2019-03-30.
- S2CID 527031.
- ^ Taylor D, Paton C, Kapur S, Taylor D (2012). The Maudsley Prescribing Guidelines in Psychiatry (11th ed.). Chichester, West Sussex: John Wiley & Sons.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ PMID 9537821.
- ^ S2CID 35553281.
- ^ PMID 9686407.
- PMID 1738111.
- S2CID 23503235.
- S2CID 21344978.
- ^ S2CID 20221398.
- S2CID 25820829.
- PMID 10455251.
- PMID 7582481.
- PMID 8632342.
- PMID 2543418.
- S2CID 35874409.
- PMID 14645659.
- ^ PMID 15771415.
- PMID 9298538.
- PMID 9149537.
- ^ PMID 6086881.
- PMID 2172775.
- PMID 2532362.
- PMID 16782354.
- ^ S2CID 14274150.
- PMID 11179435.
- ^ PMID 22708686.
- ISBN 978-0-7506-4096-1.
- ISBN 978-3-527-60402-9.
- S2CID 18893305.
- S2CID 22105260.
- PMID 23823741.
- S2CID 23442056.
- ^ "Mirtazapine label – Australia". GuildLink, Pharmacy Guild of Australia. 27 May 2016. Archived from the original on 21 November 2018. Retrieved 21 June 2017.
- S2CID 12270528.
- ISBN 978-0-19-517668-1.
- ISBN 978-1-10702598-1.
- ISBN 978-0-70204293-5.
- ^ ISBN 978-1-4757-2085-3.
- ISBN 978-0-7514-0499-9.
- ISBN 978-1-4757-1137-0.
- ISBN 978-1-135-06284-2.
- ^ "Lumin Mianserin hydrochloride product information" (PDF). Medicines. AlphaPharm – via GuildLink.
- PMID 25991654.
- PMID 25240772.
Further reading
- Peet M, Behagel H (1978). "Mianserin: a decade of scientific development". British Journal of Clinical Pharmacology. 5 (Suppl 1): 5S–9S. PMID 623702.
