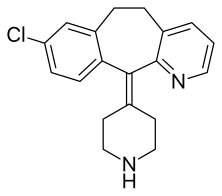
Desloratadine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Clarinex, Aerius, Allex, others[1][2] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602002 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth (tablets, solution) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Rapidly absorbed |
| Protein binding | 83 to 87% |
| Metabolism | UGT2B10, CYP2C8 |
| Metabolites | 3-Hydroxydesloratadine |
| Onset of action | within 1 hour[3] |
| Elimination half-life | 27 hours[3] |
| Duration of action | up to 24 hours[3] |
| Excretion | 40% as conjugated metabolites into urine Similar amount into the feces |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Desloratadine (trade names Clarinex and Aerius) is a tricyclic H1 inverse agonist that is used to treat allergies. It is an active metabolite of loratadine.[3]
It was patented in 1984 and came into medical use in 2001.[4]
Medical uses
Desloratadine is used to treat
An emerging indication for desloratadine is in the treatment of acne, as an inexpensive adjuvant to isotretinoin and possibly as maintenance therapy or monotherapy.[7][8]
Side effects
The most common side-effects are
Interactions
Co-administration with erythromycin, ketoconazole, azithromycin, fluoxetine or cimetidine resulted in elevated blood plasma concentrations of desloratadine and its metabolite 3-hydroxydesloratadine in studies. However, no clinically relevant changes were observed.[10][11]
Pharmacology
Pharmacodynamics
Desloratadine is a selective H1-
At very high doses, is also an antagonist at various subtypes of the muscarinic acetylcholine receptors. This effect is not relevant for the drug's action at therapeutic doses.[13]
Pharmacokinetics
Desloratadine is well absorbed from the gut and reaches highest
Desloratadine is metabolized to 3-hydroxydesloratadine in a three-step sequence in normal metabolizers. First, n-glucuronidation of desloratadine by UGT2B10; then, 3-hydroxylation of desloratadine N-glucuronide by CYP2C8; and finally, a non-enzymatic deconjugation of 3-hydroxydesloratadine N-glucuronide.[14] Both desloratadine and 3-hydroxydesloratadine are eliminated via urine and feces with a half-life of 27 hours in normal metabolizers.[11][15]

It exhibits only peripheral activity since it does not readily cross the
Desloratadine does not have a strong effect on a number of tested enzymes in the
Pharmacogenomics
2% of
See also
References
- S2CID 195689362.
- ^ "Allex EPAR". European Medicines Agency (EMA).
- ^ ISBN 9780323044042.
Desloratadine is a metabolite of loratadine. The onset of action is within 1 hour. Peak serum concentrations of desloratadine appear 3 hours after dosing. The mean elimination half-life of desloratadine is 27 hours and that of its metabolite is 36 hours. The consumption of food does not interfere with the absorption of desloratadine. Wheal inhibition is detected 1 hour after administration and may last 24 hours.
- ISBN 9783527607495.
- ^ PMID 14655812. Archived from the originalon 2005-07-24. Retrieved 2005-08-01.
- ^ Drugs.com Desloratadine entry at drugs.com international Page accessed May 4, 2015
- S2CID 3406128.
- PMID 27015774.
- ^ S2CID 40472187.
- U.S. Food and Drug Administration. Retrieved 2022-01-21.
- ^ a b c d "Aerius: EPAR – Product Information" (PDF). European Medicines Agency. Archived from the original (PDF) on 2019-07-05. Retrieved 2022-01-21.
- PMID 23268457.
- ^ "Aerius: EPAR – Scientific Discussion" (PDF). European Medicines Agency. 2006-04-03. Archived from the original (PDF) on 2018-03-16. Retrieved 2017-10-13.
- ^ PMID 26135009.
- ^ a b Drugs.com: Desloratadine Monograph.
- PMID 10784544.
External links
- "Desloratadine". Drug Information Portal. U.S. National Library of Medicine.
