Laron syndrome
| Laron syndrome | |
|---|---|
| Other names | genetic pituitary dwarfism (1966), Laron dwarfism (1973), Laron-type dwarfism (1984), growth hormone insensitivity (1994), hereditary somatomedin deficiency, growth hormone receptor deficiency (GHRD)(1999) IGF-1, Mecasermin[3] |
| Frequency | 1–9 / 1,000,000 (approximately 250 known cases worldwide) [4][5] |
Laron syndrome (LS), also known as growth hormone insensitivity or growth hormone receptor deficiency (GHRD), is an
Affected individuals classically present with
LS is a very rare condition with a total of 250 known individuals worldwide.[4][5] The genetic origins of these individuals have been traced back to Mediterranean, South Asian, and Semitic ancestors, with the latter group comprising the majority of cases.[5] Molecular genetic testing for growth hormone receptor gene mutations confirms the diagnosis of LS, but clinical evaluation may include laboratory analysis of basal GH, IGF-1 and IGFBP levels, GH stimulation testing, and/or GH trial therapy.
People with LS are unresponsive to growth hormone therapy; the disease is instead treated mainly with recombinant IGF-1, Mecasermin.[3]
Evidence has suggested that people with Laron syndrome have a reduced risk of developing cancer and
Presentation
Physical features
LS is recognized as being part of a spectrum of conditions that affect the
In addition to short stature, other characteristic physical symptoms of LS include: prominent forehead, depressed
Pathophysiology
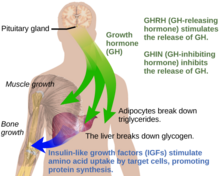

Under normal circumstances in humans, growth hormone (GH) is released in a pulsatile fashion from cells known as
Growth hormone receptor mutations
Molecular genetic investigations have shown that LS is mainly associated with autosomal recessive mutations in the gene for the growth hormone receptor (GHR).[6][17] These can result in defective hormone binding to the ectodomain or reduced efficiency of dimerization of the receptor after hormone occupancy.[18]
LS is generally classified as "primary" GH insensitivity, which is distinguished from "secondary" GH insensitivity.
STAT5B
A related condition involving postreceptor insensitivity to growth hormone has been associated with STAT5B.[19]
Diagnosis
LS should be suspected in children or adults with distinctive physical features listed above, extremely elevated serum hGH concentrations despite low serum IGF-1 levels.[14] A failure of IGF-1 to increase in response to exogenous hGH (IGF-1 stimulation test) is diagnostic for LS.[14] The gold standard for confirming a diagnosis of LS is to perform a genetic analysis with PCR to identify the precise molecular defect in the GH receptor gene.[14] Other laboratory abnormalities include GHBP (growth hormone binding protein) levels being low in cases with mutations in the extracellular domain of the GH receptor and normal in cases with mutations in the intracellular domain. Low serum levels of IGFBP are non-diagnostic for LS.[14]
Treatment
Administration of
The drug product
IPLEX (Mecasermin rinfabate) is composed of recombinant human
Legal battle for access to Medicine in Ecuador
In Ecuador, despite having the highest number of Laron syndrome cases in the world, affected children lacked proper treatment. In 2010 and 2014, a group of parents, led by Santiago Vasco Morales, filed lawsuits requesting the Ecuadorian government to provide the necessary comprehensive treatment. However, due to the lack of response and non-compliance with court rulings, the parents sought assistance from the Inter-American Commission on Human Rights (IACHR). Only after an admissibility report was issued by the IACHR on April 24, 2020 [27] did the Ecuadorian state begin administering the treatment. Unfortunately, many patients who needed IGF-1 treatment were unable to benefit from it, as they reached adulthood without receiving this essential medication. This was due to the unjustified refusal of the Ecuadorian state to provide such treatment for more than a decade.[28]
Prognosis
Cancer and diabetes
It has been reported that people with LS in Ecuador are resistant to cancer and diabetes and are somewhat protected against aging.[29][30][31] This is consistent with findings in mice with a defective growth hormone receptor gene.[22] Among the approximately 100 individuals in this population, there were no reported cases of diabetes and one case of cancer.[32]
A 2019 study of individuals with
Incidence
Numerous Laron syndrome patients are found in Israel among the country's diverse Jewish population composed of Jews from around the world, as well as patients outside Israel originally from communities of the Jewish diaspora, such as Egypt and Iraq. The original "Israeli cohort" of patients referred to Zvi Laron and colleagues beginning in 1958 consisted of 64 patients as of 2009, including 4 deceased patients.[1] The countries of origin of these patients include Israel, Palestine, Jordan, Lebanon, Iran, Malta, Italy, Argentina, Ecuador, and Peru.[1]
A disproportionate number of people with the condition are found in remote villages in the
Other patients include people of other Semitic non-Jewish origins, including from Saudi Arabia, Japan, and China.[1]
Homo floresiensis
Recent publications have proposed that
History
See also
- Hypothalamic–pituitary–somatic axis
References
- ^ ISBN 978-3-642-11183-9. Retrieved 10 November 2020.
- ^ a b c d e f g Hamosh A, O'Neill M, Phillips J, McKusick V. "# 262500 LARON SYNDROME". omim.org. McKusick-Nathans Institute of Genetic Medicine, Johns Hopkins University School of Medicine. Retrieved 10 November 2020.
- ^ S2CID 5798925.
- ^ a b Leger J. "ORPHA:633". orpha.net. Retrieved 30 October 2020.
- ^ a b c d e f Rosenbloom AL (13 November 2019). "Growth Hormone Resistance". Medscape Reference. Retrieved 3 November 2020.
- ^ PMID 15001582.
- ^ S2CID 11524548.
- ^ PMID 25905205. Retrieved 3 November 2020.
- ^ ISBN 978-3-642-11183-9.
- PMID 28528685.
- PMID 28457105.
- PMID 21525302.
- ^ "Laron Syndrome". GARD: Genetics and Rare Disease Information Center. Retrieved 16 November 2020.
- ^ ISBN 978-3-642-11183-9. Retrieved 10 November 2020.
- S2CID 260168044.
- ^ ISBN 978-3319280387. Retrieved 16 November 2020.
- PMID 29748515.
- ISBN 978-3-642-11183-9. Retrieved 12 November 2020.
- S2CID 46405455.
- PMID 6263171.
- PMID 19946434.
- ^ ISSN 0362-4331. Retrieved 17 February 2011.
- ^ "Increlex (mecasermin)". Centerwatch.com. Retrieved 21 November 2014.
- ^ Kemp SF. "Mecasermin rinfabate". Thomson Reuters. Retrieved 5 March 2011.
- ^ Meyer R. "Approval letter (Mecasermin rinfabate)" (PDF). FDA. Retrieved 5 March 2011.
- ^ "Insmed Provides Update on Supply of IPLEX(TM)". Retrieved 25 August 2017.
- ^ "Report No. 75/20 - Petition 1011-11 - Report on Admissibility" (PDF). www.oas.org. Archived from the original (PDF) on 20 October 2020. Retrieved 9 June 2023.
- ISSN 1981-982X.
- ^ PMID 21325617.
- ^ Bai N. "Defective Growth Gene in Rare Dwarfism Disorder Stunts Cancer and Diabetes". Scientific American. Retrieved 17 February 2011.
- PBS. Retrieved 17 February 2011.
- PMID 22261798.
- ^ PMID 30576428.
- PMID 2233903. Retrieved 10 November 2020.
- PMID 17596857.
- S2CID 89469580.
- PMID 20229502.
- ISBN 978-3-642-11183-9. Retrieved 12 November 2020.
- Who Named It?
- PMID 5916640.
External links
- Laron+syndrome at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
