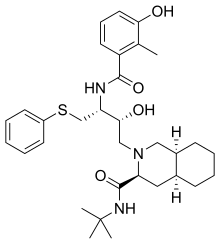
Nelfinavir
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Viracept |
| Other names | NFV |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697034 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Uncertain; increases when taking with food[1] |
| Protein binding | >98% |
| Metabolism | Liver by CYP including CYP3A4 and CYP2C19 |
| Elimination half-life | 3.5–5 hours |
| Excretion | feces (87%), urine (1–2%) |
| Identifiers | |
| |
JSmol) | |
| Melting point | 349.94 °C (661.89 °F) |
| |
| |
| | |
Nelfinavir, sold under the brand name Viracept, is an
Nelfinavir is an orally bioavailable human immunodeficiency virus HIV-1 protease inhibitor (Ki = 2 nM) and is widely prescribed in combination with HIV reverse transcriptase inhibitors for the treatment of HIV infection.[2]
It was patented in 1992 and approved for medical use in 1997.[3]
Toxicity
Common (>1%) side effects include insulin resistance, hyperglycemia and lipodystrophy.[4]
Nelfinavir can produce a range of adverse side effects. Flatulence,
reactions may occur, but are rare (less than one in one thousand patients) .Other bioactivity
Antiviral
Nelfinavir inhibits maturation and export of the
Anti-virulence activity
Nelfinavir and simple derivatives have been found to inhibit the production of the virulence factor streptolysin S, a cytolysin produced by the human pathogen Streptococcus pyogenes.[7] Nelfinavir and these related molecules did not exhibit detectable antibiotic activity, but did also inhibit the production of other biologically active molecules, including plantazolicin (antibiotic), listeriolysin S (cytolysin), and clostridiolysin S (cytolysin), by other bacteria.[7]
Interactions
Nelfinavir's interaction profile is similar to that of other
, by which nelfinavir is metabolised.Pharmacology
Nelfinavir should be taken with food. Taking the drug with food decreases the risk of diarrhea as a side effect.
Mechanism of action
Nelfinavir is a
History
Nelfinavir was developed by Agouron Pharmaceuticals as part of a joint venture with Eli Lilly and Company.
The US Food and Drug Administration (FDA) approved it for therapeutic use in March 1997,[citation needed] making it the twelfth[citation needed] approved antiretroviral. The initial product launched proved to be the largest[citation needed] "biotech launch" in the history of the pharmaceutical industry, achieving first full year sales exceeding $US335M.[citation needed] Agouron's patent on the drug expired in 2014.
In June 2007, both the Medicines and Healthcare products Regulatory Agency and the European Medicines Agency[9] put out an alert requesting the recall of any of the drug in circulation, because some batches may have been contaminated with potentially cancer-causing chemicals.
Research
Since 2009, nelfinavir has been under investigation for potential use as an anti-cancer agent.[10] When applied to cancer cells in culture (in vitro), it can inhibit the growth of a variety of cancer types and can trigger cell death (apoptosis).[11] When Nelfinavir was given to laboratory mice with tumors of the prostate or of the brain, it could suppress tumor growth in these animals.[12][13] At the cellular level, nelfinavir exerts multiple effects to inhibit cancer growth; the two main ones appear to be inhibition of the Akt/PKB signaling pathway and activation of endoplasmic reticulum stress with subsequent unfolded protein response.[14]
In the United States, about three dozen clinical trials are being conducted (or have been completed) in order to determine whether nelfinavir is effective as a cancer therapeutic agent in humans.
As of April 2022[update], nelfinavir is being studied as a radiosensitizing agent as part of treatment of advanced cervical cancer.[16]
References
- ^ "Viracept (nelfinavir mesylate) Tablets and Oral Powder, for Oral Use. Full Prescribing Information" (PDF). ViiV Healthcare Company. Research Triangle Park, NC 27709. Retrieved 23 November 2015.
- PMID 11257019.
- ISBN 9783527607495.
- ^ PMID 25013375.
- PMID 24574416.
- PMID 21402841.
- ^ PMID 25668590.
- PMID 9397180.
- ^ Press release from the European Medicines Agency regarding possible genotoxic ethyl mesylate contamination
- PMID 19111246.
- PMID 17785575.
- PMID 18006837.
- S2CID 44702882.
- PMID 26097685.
- ^ "Search of: Nelfinavir cancer - List Results - ClinicalTrials.gov". clinicaltrials.gov.
- ^ Clinical trial number NCT03256916 for "A Phase III Randomized Clinical Trial to Study the Radiosensitizing Effect of Nelfinavir in Locally Advanced Carcinoma of Uterine Cervix." at ClinicalTrials.gov
Further reading
- Pai VB, Nahata MC (March 1999). "Nelfinavir mesylate: a protease inhibitor". The Annals of Pharmacotherapy. 33 (3): 325–339. S2CID 24066955.
- Bardsley-Elliot A, Plosker GL (March 2000). "Nelfinavir: an update on its use in HIV infection". Drugs. 59 (3): 581–620. PMID 10776836.
